Biomaterials Translational ›› 2021, Vol. 2 ›› Issue (3): 214-235.doi: 10.12336/biomatertransl.2021.03.005
• REVIEW • Previous Articles Next Articles
Yu Lu1, Subodh Deshmukh2, Ian Jones1, Yu-Lung Chiu1,*( )
)
Received:2021-06-01
Revised:2021-07-14
Accepted:2021-08-16
Online:2021-09-28
Published:2021-09-28
Contact:
Yu-Lung Chiu
E-mail:y.chiu@bham.ac.uk
About author:Yu-Lung Chiu, y.chiu@bham.ac.uk.Lu, Y.; Deshmukh, S.; Jones, I.; Chiu, Y. Biodegradable magnesium alloys for orthopaedic applications. Biomater Transl. 2021, 2(3), 214-235.
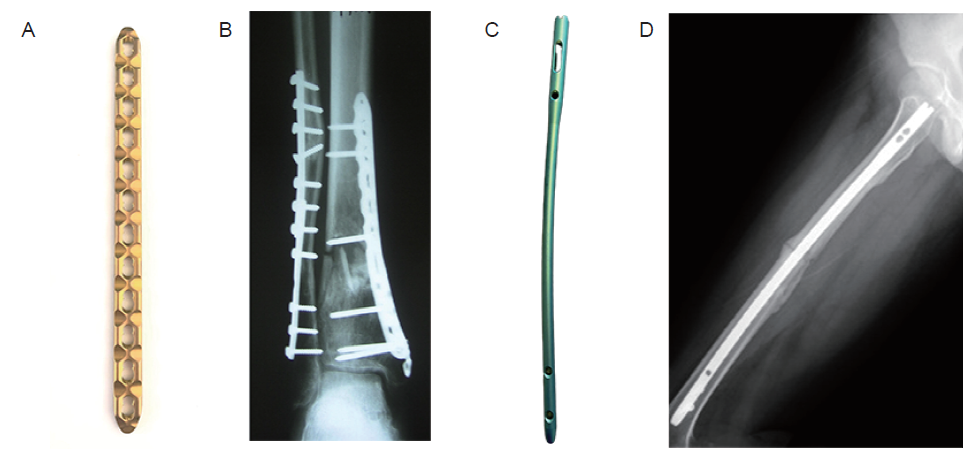
Figure 1. (A) Locking compression plate. (B) Radiograph of distal tibial fracture treated with a locking plate. Reprinted from Bastias et al.11 Copyright 2014 European Foot and Ankle Society. (C) Interlocking nail. (D) Radiograph of femoral fracture treated with locking nail. Reprinted from Hsu et al.12 Copyright 2019, with permission from Elsevier.
| Yield strength (MPa) | Ultimate tensile strength (MPa) | Elongation (%) | |
|---|---|---|---|
| CP Ti | |||
| Grade 1 | 170 | 240 | 24 |
| Grade 2 | 275 | 345 | 20 |
| Grade 3 | 380 | 450 | 18 |
| Grade 4 | 483 | 550 | 15 |
| Stainless steel | |||
| 18Cr-14Ni-2.5Mo | |||
| Annealed | 190 | 490 | 40 |
| Cold-worked | 690 | 860 | 12 |
| Co-28Cr-6Mo | |||
| Annealed | 517 | 897 | 20 |
| Hot-worked | 700 | 1000 | 12 |
Table 1 Mechanical performance of commercial pure (CP) titanium, stainless steel and cobalt-chromium alloys for surgical implant application.
| Yield strength (MPa) | Ultimate tensile strength (MPa) | Elongation (%) | |
|---|---|---|---|
| CP Ti | |||
| Grade 1 | 170 | 240 | 24 |
| Grade 2 | 275 | 345 | 20 |
| Grade 3 | 380 | 450 | 18 |
| Grade 4 | 483 | 550 | 15 |
| Stainless steel | |||
| 18Cr-14Ni-2.5Mo | |||
| Annealed | 190 | 490 | 40 |
| Cold-worked | 690 | 860 | 12 |
| Co-28Cr-6Mo | |||
| Annealed | 517 | 897 | 20 |
| Hot-worked | 700 | 1000 | 12 |
| Density (g/cm3) | Elastic modulus (GPa) | Tensile strength (MPa) | Fracture toughness (MPa1/2) | Total elongation (%) | |
|---|---|---|---|---|---|
| Cortical bone | 1.8﹣2.1 | 3﹣20 | 35﹣283 | 3﹣6 | 1.07﹣2.10 |
| Cancellous bone | 1.0﹣1.4 | NA | 1.5﹣38 | NA | NA |
| Magnesium alloys | 1.74﹣2.0 | 41﹣45 | 150﹣400 | 15﹣40 | 2﹣20 |
Table 2 Summary of the physical and mechanical properties of magnesium alloys in comparison with human bone.
| Density (g/cm3) | Elastic modulus (GPa) | Tensile strength (MPa) | Fracture toughness (MPa1/2) | Total elongation (%) | |
|---|---|---|---|---|---|
| Cortical bone | 1.8﹣2.1 | 3﹣20 | 35﹣283 | 3﹣6 | 1.07﹣2.10 |
| Cancellous bone | 1.0﹣1.4 | NA | 1.5﹣38 | NA | NA |
| Magnesium alloys | 1.74﹣2.0 | 41﹣45 | 150﹣400 | 15﹣40 | 2﹣20 |
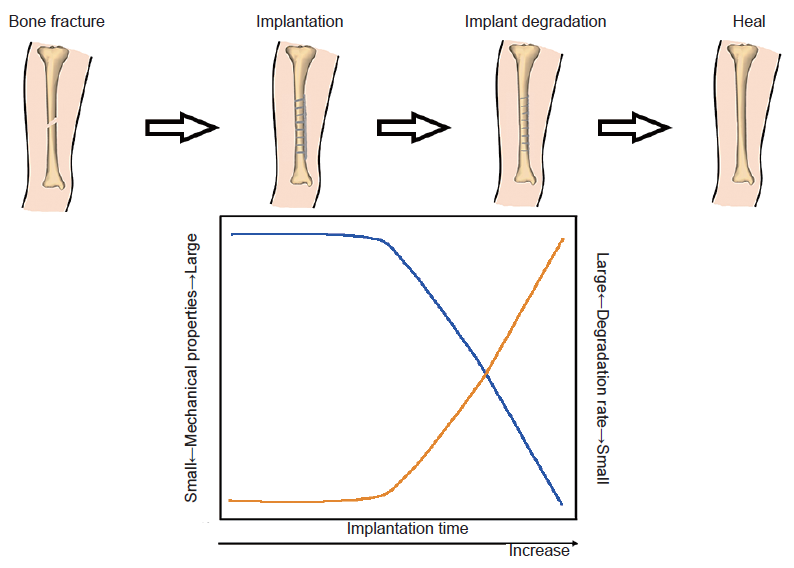
Figure 3. Optimal degradation behaviour of a magnesium-based implant in bone fracture healing. The blue and yellow lines indicate the mechanical integrity and biodegradation rate, respectively.
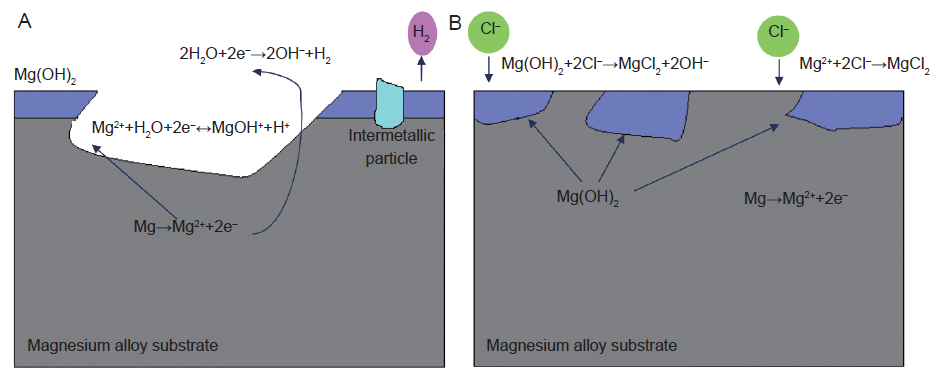
Figure 4. Schematic illustration of the corrosion of magnesium in an aqueous environment:45 (A) The dissolution of magnesium via the anodic reaction. The cathodic reaction increases the pH and produces H2, while hydrolysis reduces the pH. Intermetallic particles act as cathodic sites and consume the electrons produced by the anodic reaction. (B) Chloride ions in the solution attack and dissolve the Mg(OH)2 film.
| Recommended dietary allowances for magnesium (mg) | Tolerable upper intake levels for supplemental magnesium (mg) | ||||
|---|---|---|---|---|---|
| Age (year) | Male | Female | Male | Female | |
| 19﹣30 | 400 | 310 | 350 | 350 | |
| 31﹣50 | 420 | 320 | 350 | 350 | |
| 51 or more | 420 | 320 | 350 | 350 | |
Table 3 Intake allowances for magnesium.
| Recommended dietary allowances for magnesium (mg) | Tolerable upper intake levels for supplemental magnesium (mg) | ||||
|---|---|---|---|---|---|
| Age (year) | Male | Female | Male | Female | |
| 19﹣30 | 400 | 310 | 350 | 350 | |
| 31﹣50 | 420 | 320 | 350 | 350 | |
| 51 or more | 420 | 320 | 350 | 350 | |
| Element | Solubility limits (wt%) | Growth restriction factor | Effects on mechanical properties | Effects on corrosion behaviour | Biological impact | Maximum daily allowable dose (mg) |
|---|---|---|---|---|---|---|
| Aluminium (Al) | 12.7 | 4.32 | Improves strength and ductility Grain refinement Increases castability | Decreases corrosion rate | Neurotoxic; Decreases osteoclast viability | 14 |
| Calcium (Ca) | 1.34 | 11.94 | Improves strength Grain refinement Increases castability | Decreases corrosion resistance | Most abundant mineral in the human body; Is tightly regulated by homeostasis | 1400 |
| Zinc (Zn) | 6.2 | 5.31 | Improves strength Reduces ductility at high concentration | Essential trace element (immune system, co-factor); Neurotoxic at higher concentration | 15 | |
| Manganese (Mn) | 2.2 | 0.15 | Improves strength and ductility Grain refinement | Decreases corrosion rate by removing iron and other heavy metal elements into relatively harmless compounds | Essential trace element; Important role in metabolic cycle and for the immune system; Neurotoxic at higher concentration | 5 |
| Lithium (Li) | 5.5 | Reduces strength Improves ductility | Reduces corrosion resistance | Possible teratogenic effects | ||
| Zirconium (Zr) | 3.8 | 38.29 | Improves strength and ductility Grain refinement | |||
| Silicon (Si) | ~0 | 9.25 | Reduces ductility and castability | Reduces corrosion resistance | Essential mineral in human body Helps to build the immune system | |
| Strontium (Sr) | 0.11 | 3.51 | Improves strength and ductility; Grain refinement | Deceases corrosion rate | Trace element in human body; Stimulates bone formation | 5 |
| Yttrium (Y) | 12.4 | 1.7 | Improves strength | Same standard electrochemical potential as Mg; Decreases corrosion rate by purifying the alloy and forming a passive film | May exhibit anti-carcinogenic properties | 0.016a |
| Neodymium (Nd) | 3.6 | Improves strength | Decreases corrosion rate by creating less noble intermetallic phase (‘scavenger effect’ on impurities) | May exhibit anti-carcinogenic properties | 4.2a | |
| Copper (Cu) | Increases strength | Causes hypotension, jaundice, etc. |
Table 4 The physical properties and influence of alloying elements on properties and biological impact of magnesium-based alloys.
| Element | Solubility limits (wt%) | Growth restriction factor | Effects on mechanical properties | Effects on corrosion behaviour | Biological impact | Maximum daily allowable dose (mg) |
|---|---|---|---|---|---|---|
| Aluminium (Al) | 12.7 | 4.32 | Improves strength and ductility Grain refinement Increases castability | Decreases corrosion rate | Neurotoxic; Decreases osteoclast viability | 14 |
| Calcium (Ca) | 1.34 | 11.94 | Improves strength Grain refinement Increases castability | Decreases corrosion resistance | Most abundant mineral in the human body; Is tightly regulated by homeostasis | 1400 |
| Zinc (Zn) | 6.2 | 5.31 | Improves strength Reduces ductility at high concentration | Essential trace element (immune system, co-factor); Neurotoxic at higher concentration | 15 | |
| Manganese (Mn) | 2.2 | 0.15 | Improves strength and ductility Grain refinement | Decreases corrosion rate by removing iron and other heavy metal elements into relatively harmless compounds | Essential trace element; Important role in metabolic cycle and for the immune system; Neurotoxic at higher concentration | 5 |
| Lithium (Li) | 5.5 | Reduces strength Improves ductility | Reduces corrosion resistance | Possible teratogenic effects | ||
| Zirconium (Zr) | 3.8 | 38.29 | Improves strength and ductility Grain refinement | |||
| Silicon (Si) | ~0 | 9.25 | Reduces ductility and castability | Reduces corrosion resistance | Essential mineral in human body Helps to build the immune system | |
| Strontium (Sr) | 0.11 | 3.51 | Improves strength and ductility; Grain refinement | Deceases corrosion rate | Trace element in human body; Stimulates bone formation | 5 |
| Yttrium (Y) | 12.4 | 1.7 | Improves strength | Same standard electrochemical potential as Mg; Decreases corrosion rate by purifying the alloy and forming a passive film | May exhibit anti-carcinogenic properties | 0.016a |
| Neodymium (Nd) | 3.6 | Improves strength | Decreases corrosion rate by creating less noble intermetallic phase (‘scavenger effect’ on impurities) | May exhibit anti-carcinogenic properties | 4.2a | |
| Copper (Cu) | Increases strength | Causes hypotension, jaundice, etc. |
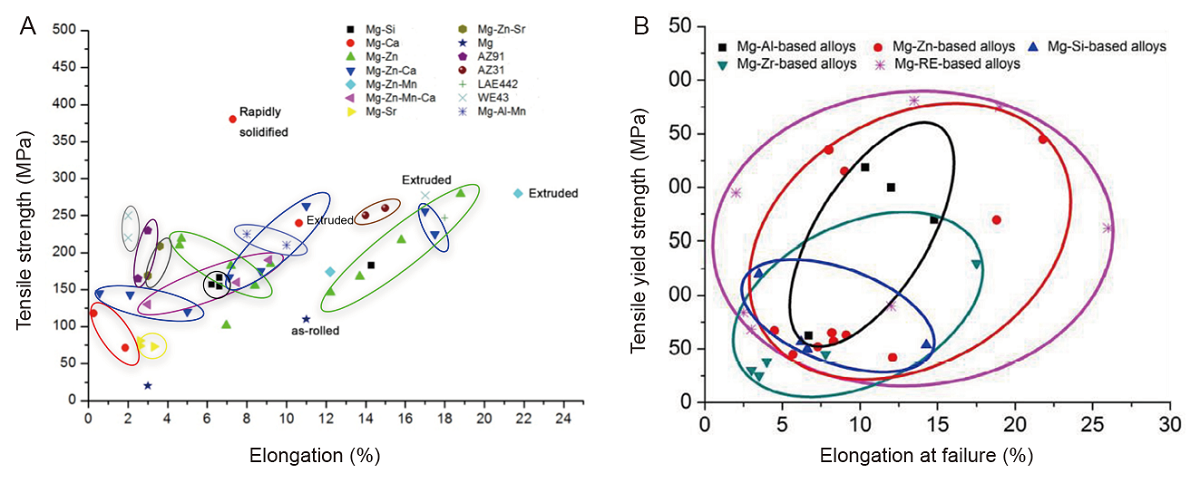
Figure 5. The tensile strength and elongation of various magnesium alloys. (A) Reprinted from Lu.45 (B) Reprinted from Chen et al.84 Copyright 2014, with permission from Acta Materialia Inc.
| Alloy | Condition | In vitro corrosion rate (mm/year) | In vivo corrosion rate (mm/year) | Reference |
|---|---|---|---|---|
| Mg-0.8Ca | As-extruded | - | 0.5 | |
| Mg-1Ca | As-cast | ﹣ | 1.27 | |
| Mg-1Al | As-cast | 2.07 | ﹣ | |
| Mg-1Zn | As-cast | 1.52 | ﹣ | |
| Mg-1Zn | As-rolled | 0.92 | ﹣ | |
| Mg-1Zr | As-cast | 2.2 | ﹣ | |
| Mg-1Zr | As-rolled | 0.91 | ﹣ | |
| Mg-1Sn | As-cast | 2.45 | ﹣ | |
| Mg-2Sr | As-rolled | 0.37 | 1.01 | |
| Mg-3Zn | Solution treated | 1.53 | ﹣ | |
| Mg-6Zn | As-extruded | 0.07 | 2.32 | |
| Mg-8Y | As-cast | 2.17 | ﹣ | |
| AZ31 | As-cast | 2 | 1.17 | |
| AZ31 | As-extruded | 0.21 | ﹣ | |
| AZ61 | As-cast | 0.73 | ﹣ | |
| AZ91D | As-cast | 2.8 | 1.38 | |
| WE43 | As-cast | 0.26 | 1.56 | |
| Mg-1.2Mn-1Zn | As-cast | ﹣ | 0.45 | |
| Mg-1Zn-1Ca | As-cast | 2.13 | ﹣ | |
| Mg-3Zn-0.3Ca | Solution treated | 0.81 | ﹣ | |
| Mg-6Zn-1Ca | As-cast | 9.21 | ﹣ | |
| Mg-4Zn-0.5Ca-0.4Mn | As-cast | 0.25 | ﹣ | |
| Mg-3.09Nd-0.22Zn-0.44Zr | As-extruded | 0.13 | ﹣ | |
| Mg-2Zn-1.53Y | As-extruded | 0.7 | ﹣ | |
| Mg-11.3Gd-2.5Zn-0.7Zr | As-extruded | 0.17 | ﹣ |
Table 5 Bio-corrosion properties of magnesium-based alloys.
| Alloy | Condition | In vitro corrosion rate (mm/year) | In vivo corrosion rate (mm/year) | Reference |
|---|---|---|---|---|
| Mg-0.8Ca | As-extruded | - | 0.5 | |
| Mg-1Ca | As-cast | ﹣ | 1.27 | |
| Mg-1Al | As-cast | 2.07 | ﹣ | |
| Mg-1Zn | As-cast | 1.52 | ﹣ | |
| Mg-1Zn | As-rolled | 0.92 | ﹣ | |
| Mg-1Zr | As-cast | 2.2 | ﹣ | |
| Mg-1Zr | As-rolled | 0.91 | ﹣ | |
| Mg-1Sn | As-cast | 2.45 | ﹣ | |
| Mg-2Sr | As-rolled | 0.37 | 1.01 | |
| Mg-3Zn | Solution treated | 1.53 | ﹣ | |
| Mg-6Zn | As-extruded | 0.07 | 2.32 | |
| Mg-8Y | As-cast | 2.17 | ﹣ | |
| AZ31 | As-cast | 2 | 1.17 | |
| AZ31 | As-extruded | 0.21 | ﹣ | |
| AZ61 | As-cast | 0.73 | ﹣ | |
| AZ91D | As-cast | 2.8 | 1.38 | |
| WE43 | As-cast | 0.26 | 1.56 | |
| Mg-1.2Mn-1Zn | As-cast | ﹣ | 0.45 | |
| Mg-1Zn-1Ca | As-cast | 2.13 | ﹣ | |
| Mg-3Zn-0.3Ca | Solution treated | 0.81 | ﹣ | |
| Mg-6Zn-1Ca | As-cast | 9.21 | ﹣ | |
| Mg-4Zn-0.5Ca-0.4Mn | As-cast | 0.25 | ﹣ | |
| Mg-3.09Nd-0.22Zn-0.44Zr | As-extruded | 0.13 | ﹣ | |
| Mg-2Zn-1.53Y | As-extruded | 0.7 | ﹣ | |
| Mg-11.3Gd-2.5Zn-0.7Zr | As-extruded | 0.17 | ﹣ |

Figure 6. (A) As-built WE43 scaffold with diamond lattice fabricated by selective laser melting. Reprinted from Li et al.40 Copyright 2017, with permission from Acta Materialia Inc. (B) Honeycomb-structured magnesium scaffold produced by laser perforation. Reprinted from Tan et al.167 Copyright IOP Publishing. Reproduced with permission. All rights reserved. Scale bars: 1 mm.
| Ion | Blood plasma | Ringer’s solution | Earle’s balanced salt solution | Hank’s balanced salt solution | Kokubo’s simulated body fluid |
|---|---|---|---|---|---|
| Na+ (mM) | 142.00 | 130.00 | 143.60 | 138.00 | 142.00 |
| K+ (mM) | 5.00 | 4.00 | 5.37 | 6.14 | 5.00 |
| Ca2+ (mM) | 2.50 | 1.40 | 1.80 | 1.26 | 2.50 |
| Mg2+ (mM) | 1.50 | NA | 0.81 | 0.81 | 1.50 |
| Cl﹣ (mM) | 103.00 | 109.00 | 125.30 | 144.8 | 147.80 |
| HCO3﹣ (mM) | 27.00 | NA | 26.2 | 4.2 | 4.20 |
| HPO42- (mM) | 1.00 | NA | 1.00 | 0.78 | 1.00 |
| SO42- (mM) | 0.50 | NA | 0.81 | 0.81 | 0.50 |
| Ca/P (mM) | 2.50 | NA | 1.80 | 1.62 | 2.50 |
| Buffer (mM) | NA | NA | NA | NA | Tris |
| pH | 7.4 | 6.5 | 6.7-6.9 | 6.7-6.9 | 7.4 |
Table 6 The concentration of ions and pH values in blood plasma and in different solutions.
| Ion | Blood plasma | Ringer’s solution | Earle’s balanced salt solution | Hank’s balanced salt solution | Kokubo’s simulated body fluid |
|---|---|---|---|---|---|
| Na+ (mM) | 142.00 | 130.00 | 143.60 | 138.00 | 142.00 |
| K+ (mM) | 5.00 | 4.00 | 5.37 | 6.14 | 5.00 |
| Ca2+ (mM) | 2.50 | 1.40 | 1.80 | 1.26 | 2.50 |
| Mg2+ (mM) | 1.50 | NA | 0.81 | 0.81 | 1.50 |
| Cl﹣ (mM) | 103.00 | 109.00 | 125.30 | 144.8 | 147.80 |
| HCO3﹣ (mM) | 27.00 | NA | 26.2 | 4.2 | 4.20 |
| HPO42- (mM) | 1.00 | NA | 1.00 | 0.78 | 1.00 |
| SO42- (mM) | 0.50 | NA | 0.81 | 0.81 | 0.50 |
| Ca/P (mM) | 2.50 | NA | 1.80 | 1.62 | 2.50 |
| Buffer (mM) | NA | NA | NA | NA | Tris |
| pH | 7.4 | 6.5 | 6.7-6.9 | 6.7-6.9 | 7.4 |

Figure 7. Micro-computed tomographic three-dimensional images of as-cast Mg-3Zn-0.3Ca alloy. (A) Before immersion test. (B) After immersion test. Scale bars: 2 mm. Reprinted from Lu et al.105 Copyright 2018, with permission from Acta Materialia Inc.
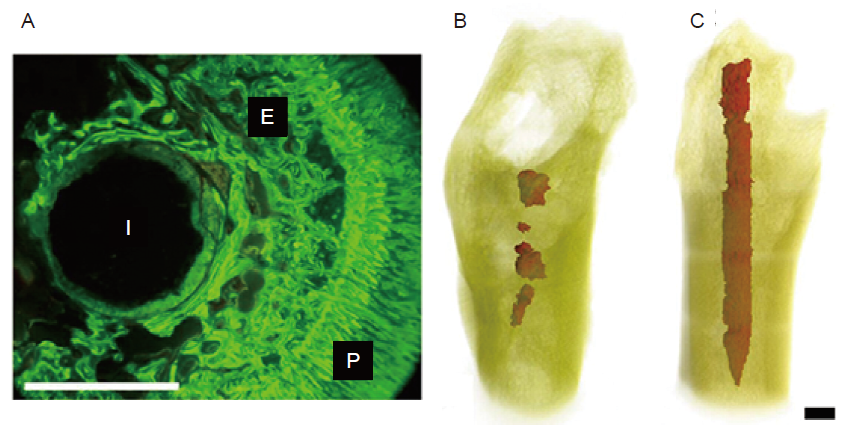
Figure 8. (A) Fluoroscopic image of cross-section of magnesium rod in a guinea pig femur. E: endosteal bone formation; I: implant residual; P: periosteal bone formation. (B) Three-dimensional reconstruction of remaining AZ91D in the femur of a guinea pig. (C) Three-dimensional reconstruction of remaining LAE442 in the femur of a guinea pig. Scale bars: 1.5 mm. A-C were reprinted from Witte et al.39, 109 Copyright 2005 & 2006, with permission from Elsevier Ltd.
| [1] | Ratner, B. D.; Hoffman, A. S.; Schoen, F. J.; Lemons, J. E. Biomaterial sciences: an introduction to materials in medicine. Academic Press: San Diego. 1996. |
| [2] | Temenoff, J. S.; Mikos, A. G. Biomaterials: the intersection of biology and materials sceince. Pearson. 2008. |
| [3] | Zheng, Y. F.; Gu, X. N.; Witte, F. Biodegradable metals. Mater Sci Eng RRep. 2014, 77, 1-34. |
| [4] | B., Ermanno. Basic Composition and Structure of Bone. In Mechanical testing of bone and the bone-implant interface, An, Y. H.; Draughn, R. A., eds.; CRC Press: Boca Raton, 1999; pp 3-22. |
| [5] | Suzuki, H.; He, J. Evaluation on bending properties of biomaterial GUM Metal meshed plates for bone graft applications. In IOP Conference Series: Materials Science and Engineering, IOP Publishing: Tianjin. 2017; Vol. 269, p 012078. |
| [6] | Augat, P.; von Rüden, C. Evolution of fracture treatment with bone plates. Injury. 2018, 49 Suppl 1, S2-S7. |
| [7] |
Nana, A. D.; Joshi, A.; Lichtman, D. M. Plating of the distal radius. J Am Acad Orthop Surg. 2005, 13, 159-171.
doi: 10.5435/00124635-200505000-00003 URL |
| [8] | AO Foundation. AO Foundation Surgery Reference. https://surgeryreference.aofoundation.org/ . Accessed April 13, 2021. |
| [9] |
Gueorguiev, B.; Lenz, M. Why and how do locking plates fail? Injury. 2018, 49 Suppl 1, S56-S60.
doi: 10.1016/S0020-1383(18)30305-X URL |
| [10] |
Perren, S. M. Evolution of the internal fixation of long bone fractures. The scientific basis of biological internal fixation: choosing a new balance between stability and biology. J Bone Joint Surg Br. 2002, 84, 1093-1110.
doi: 10.1302/0301-620X.84B8.0841093 URL |
| [11] |
Bastias, C.; Henríquez, H.; Pellegrini, M.; Rammelt, S.; Cuchacovich, N.; Lagos, L.; Carcuro, G. Are locking plates better than non-locking plates for treating distal tibial fractures? Foot Ankle Surg. 2014, 20, 115-119.
doi: 10.1016/j.fas.2013.12.004 URL |
| [12] |
Hsu, K. L.; Kuan, F. C.; Chang, W. L.; Liu, Y. F.; Hong, C. K.; Yeh, M. L.; Su, W. R. Interlocking nailing of femoral shaft fractures with an extremely narrow medullary canal is associated with iatrogenic fractures. Injury. 2019, 50, 2306-2311.
doi: 10.1016/j.injury.2019.10.010 URL |
| [13] |
Cronier, P.; Pietu, G.; Dujardin, C.; Bigorre, N.; Ducellier, F.; Gerard, R. The concept of locking plates. Orthop Traumatol Surg Res. 2010. doi: 10.1016/j.otsr.2010.03.008.
doi: 10.1016/j.otsr.2010.03.008 URL |
| [14] | McRae, R.; Esser, M. Practical Fracture Treatment. 5th ed. Churchill Livingstone: 2008. |
| [15] | Elias, C. N.; Lima, J. H. C.; Valiev, R.; Meyers, M. A. Biomedical applications of titanium and its alloys. JOM. 2008, 60, 46-49. |
| [16] | ASTM F67. Standard specification for unalloyed titanium, for surgical implant applications. 2006. |
| [17] | ASTM F138-19. Standard specification for wrought 18chromium-14nickel-2.5molybdenum stainless steel bar and wire for surgical implants. 2020. |
| [18] | ASTM F1537-20. Standard specification for wrought cobalt-28chromium-6molybdenum alloys for surgical implants. 2020. |
| [19] |
Disegi, J. A.; Eschbach, L. Stainless steel in bone surgery. Injury. 2000, 31 Suppl 4, 2-6.
doi: 10.1016/S0020-1383(99)00256-9 URL |
| [20] | Disegi, J. A. Titanium alloys for fracture fixation implants. Injury. 2000, 31 Suppl 4, 14-17. |
| [21] | Golish, S. R.; Mihalko, W. M. Principles of biomechanics and biomaterials in orthopaedic surgery. Instr Course Lect. 2011, 60, 575-581. |
| [22] |
Bayraktar, H. H.; Morgan, E. F.; Niebur, G. L.; Morris, G. E.; Wong, E. K.; Keaveny, T. M. Comparison of the elastic and yield properties of human femoral trabecular and cortical bone tissue. J Biomech. 2004, 37, 27-35.
doi: 10.1016/S0021-9290(03)00257-4 URL |
| [23] |
Ryan, G.; Pandit, A.; Apatsidis, D. P. Fabrication methods of porous metals for use in orthopaedic applications. Biomaterials. 2006, 27, 2651-2670.
doi: 10.1016/j.biomaterials.2005.12.002 URL |
| [24] | Vamsi Krishna, B.; Xue, W.; Bose, S.; Bandyopadhyay, A. Engineered porous metals for implants. JOM. 2008, 60, 45-48. |
| [25] |
Lhotka, C.; Szekeres, T.; Steffan, I.; Zhuber, K.; Zweymüller, K. Four-year study of cobalt and chromium blood levels in patients managed with two different metal-on-metal total hip replacements. J Orthop Res. 2003, 21, 189-195.
doi: 10.1016/S0736-0266(02)00152-3 URL |
| [26] |
Maehara, T.; Moritani, S.; Ikuma, H.; Shinohara, K.; Yokoyama, Y. Difficulties in removal of the titanium locking plate in Japan. Injury. 2013, 44, 1122-1126.
doi: 10.1016/j.injury.2013.01.042 URL |
| [27] |
Morgan, E. F.; Unnikrisnan, G. U.; Hussein, A. I. Bone mechanical properties in healthy and diseased states. Annu Rev Biomed Eng. 2018, 20, 119-143.
doi: 10.1146/bioeng.2018.20.issue-1 URL |
| [28] | Friedrich, H. E.; Mordike, B. L. Magnesium technology: metallurgy, design data, applications. Springer, Berlin,Heidelberg, 2006. |
| [29] | Schweitzer, P. E. Metallic materials: physical, mechanical, and corrosion properties. CRC Press: 2020. |
| [30] |
Staiger, M. P.; Pietak, A. M.; Huadmai, J.; Dias, G. Magnesium and its alloys as orthopedic biomaterials: a review. Biomaterials. 2006, 27, 1728-1734.
doi: 10.1016/j.biomaterials.2005.10.003 URL |
| [31] | Park, J. B. Biomaterials science and engineering. Plenum Press: New York, 1984. |
| [32] |
Hartwig, A. Role of magnesium in genomic stability. Mutat Res. 2001, 475, 113-121.
doi: 10.1016/S0027-5107(01)00074-4 URL |
| [33] |
Okuma, T. Magnesium and bone strength. Nutrition. 2001, 17, 679-680.
doi: 10.1016/S0899-9007(01)00551-2 URL |
| [34] |
Song, G.; Song, S. A possible biodegradable magnesium implant material. Adv Eng Mater. 2007, 9, 298-302.
doi: 10.1002/(ISSN)1527-2648 URL |
| [35] |
Rodríguez-Sánchez, J.; Pacha-Olivenza, M. Á.; González-Martín, M. L. Bactericidal effect of magnesium ions over planktonic and sessile Staphylococcus epidermidis and Escherichia coli. Mater Chem Phys. 2019, 221, 342-348.
doi: 10.1016/j.matchemphys.2018.09.050 URL |
| [36] | Xu, L.; Yu, G.; Zhang, E.; Pan, F.; Yang, K. In vivo corrosion behavior of Mg-Mn-Zn alloy for bone implant application. J Biomed Mater Res A. 2007, 83, 703-711. |
| [37] |
Saris, N. E.; Mervaala, E.; Karppanen, H.; Khawaja, J. A.; Lewenstam, A. An update on physiological, clinical and analytical aspects. Clin Chim Acta. 2000, 294, 1-26.
doi: 10.1016/S0009-8981(99)00258-2 URL |
| [38] |
Gu, X.; Zheng, Y.; Cheng, Y.; Zhong, S.; Xi, T. In vitro corrosion and biocompatibility of binary magnesium alloys. Biomaterials. 2009, 30, 484-498.
doi: 10.1016/j.biomaterials.2008.10.021 URL |
| [39] |
Witte, F.; Kaese, V.; Haferkamp, H.; Switzer, E.; Meyer-Lindenberg, A.; Wirth, C. J.; Windhagen, H. In vivo corrosion of four magnesium alloys and the associated bone response. Biomaterials. 2005, 26, 3557-3563.
doi: 10.1016/j.biomaterials.2004.09.049 URL |
| [40] | Witte, F.; Ulrich, H.; Rudert, M.; Willbold, E. Biodegradable magnesium scaffolds: Part 1: appropriate inflammatory response. J Biomed Mater Res A. 2007, 81, 748-756. |
| [41] |
Claes, L. E. Mechanical characterization of biodegradable implants. Clin Mater. 1992, 10, 41-46.
doi: 10.1016/0267-6605(92)90083-6 URL |
| [42] | Ruedi, T. P.; Buckley, R. E.; Moran, C. G. Principles of fracture management. 2nd ed. Thieme: 2007. |
| [43] | Chalisgaonkar, R. Insight in applications, manufacturing and corrosion behaviour of magnesium and its alloys - A review. Mater Today Proc. 2020, 26, 1060-1071. |
| [44] | Scarritt, M. E.; Londono, R.; Badylak, S. F. Host response to implanted materials and devices: an overview. In The immune response to implanted materials and devices: the impact of the immune system on the success of an implant, Corradetti, B., ed. Springer International Publishing: Cham, 2017; pp 1-14. |
| [45] | Lu, Y. Microstructure and degradation behaviour of Mg-Zn(-Ca) alloys. University of Birmingham: Birmingham, 2014. |
| [46] |
Song, G.; Atrens, A. Understanding magnesium corrosion—a framework for improved alloy performance. Adv Eng Mater. 2003, 5, 837-858.
doi: 10.1002/(ISSN)1527-2648 URL |
| [47] |
Ambat, R.; Aung, N. N.; Zhou, W. Studies on the influence of chloride ion and pH on the corrosion and electrochemical behaviour of AZ91D magnesium alloy. J Appl Electrochem. 2000, 30, 865-874.
doi: 10.1023/A:1004011916609 URL |
| [48] |
Ambat, R.; Aung, N. N.; Zhou, W. Evaluation of microstructural effects on corrosion behaviour of AZ91D magnesium alloy. Corros Sci. 2000, 42, 1433-1455.
doi: 10.1016/S0010-938X(99)00143-2 URL |
| [49] |
Kraus, T.; Fischerauer, S. F.; Hänzi, A. C.; Uggowitzer, P. J.; Löffler, J. F.; Weinberg, A. M. Magnesium alloys for temporary implants in osteosynthesis: in vivo studies of their degradation and interaction with bone. Acta Biomater. 2012, 8, 1230-1238.
doi: 10.1016/j.actbio.2011.11.008 URL |
| [50] |
Xin, Y.; Hu, T.; Chu, P. K. In vitro studies of biomedical magnesium alloys in a simulated physiological environment: a review. Acta Biomater. 2011, 7, 1452-1459.
doi: 10.1016/j.actbio.2010.12.004 URL |
| [51] |
Holweg, P.; Berger, L.; Cihova, M.; Donohue, N.; Clement, B.; Schwarze, U.; Sommer, N. G.; Hohenberger, G.; van den Beucken, J.; Seibert, F.; Leithner, A.; Löffler, J. F.; Weinberg, A. M. A lean magnesium-zinc-calcium alloy ZX00 used for bone fracture stabilization in a large growing-animal model. Acta Biomater. 2020, 113, 646-659.
doi: 10.1016/j.actbio.2020.06.013 URL |
| [52] |
Jahnen-Dechent, W.; Ketteler, M. Magnesium basics. Clin Kidney J. 2012, 5, i3-i14.
doi: 10.1093/ndtplus/sfr163 URL |
| [53] |
Shahi, A.; Aslani, S.; Ataollahi, M.; Mahmoudi, M. The role of magnesium in different inflammatory diseases. Inflammopharmacology. 2019, 27, 649-661.
doi: 10.1007/s10787-019-00603-7 URL |
| [54] |
al-Ghamdi, S. M.; Cameron, E. C.; Sutton, R. A. Magnesium deficiency: pathophysiologic and clinical overview. Am J Kidney Dis. 1994, 24, 737-752.
doi: 10.1016/S0272-6386(12)80667-6 URL |
| [55] | Ding, S.; Zhang, J.; Tian, Y.; Huang, B.; Yuan, Y.; Liu, C. Magnesium modification up-regulates the bioactivity of bone morphogenetic protein-2 upon calcium phosphate cement via enhanced BMP receptor recognition and Smad signaling pathway. Colloids Surf BBiointerfaces. 2016, 145, 140-151. |
| [56] |
Zhou, H.; Liang, B.; Jiang, H.; Deng, Z.; Yu, K. Magnesium-based biomaterials as emerging agents for bone repair and regeneration: from mechanism to application. J Magnes Alloys. 2021, 9, 779-804.
doi: 10.1016/j.jma.2021.03.004 URL |
| [57] |
Qiao, W.; Wong, K. H. M.; Shen, J.; Wang, W.; Wu, J.; Li, J.; Lin, Z.; Chen, Z.; Matinlinna, J. P.; Zheng, Y.; Wu, S.; Liu, X.; Lai, K. P.; Chen, Z.; Lam, Y. W.; Cheung, K. M. C.; Yeung, K. W. K. TRPM7 kinase-mediated immunomodulation in macrophage plays a central role in magnesium ion-induced bone regeneration. Nat Commun. 2021, 12, 2885.
doi: 10.1038/s41467-021-23005-2 URL |
| [58] |
Zhang, Y.; Xu, J.; Ruan, Y. C.; Yu, M. K.; O’Laughlin, M.; Wise, H.; Chen, D.; Tian, L.; Shi, D.; Wang, J.; Chen, S.; Feng, J. Q.; Chow, D. H.; Xie, X.; Zheng, L.; Huang, L.; Huang, S.; Leung, K.; Lu, N.; Zhao, L.; Li, H.; Zhao, D.; Guo, X.; Chan, K.; Witte, F.; Chan, H. C.; Zheng, Y.; Qin, L. Implant-derived magnesium induces local neuronal production of CGRP to improve bone-fracture healing in rats. Nat Med. 2016, 22, 1160-1169.
doi: 10.1038/nm.4162 URL |
| [59] | Cheng, S.; Zhang, D.; Li, M.; Liu, X.; Zhang, Y.; Qian, S.; Peng, F. Osteogenesis, angiogenesis and immune response of Mg-Al layered double hydroxide coating on pure Mg. Bioact Mater. 2021, 6, 91-105. |
| [60] |
Yoshizawa, S.; Brown, A.; Barchowsky, A.; Sfeir, C. Magnesium ion stimulation of bone marrow stromal cells enhances osteogenic activity, simulating the effect of magnesium alloy degradation. Acta Biomater. 2014, 10, 2834-2842.
doi: 10.1016/j.actbio.2014.02.002 URL |
| [61] |
Salimi, M. H.; Heughebaert, J. C.; Nancollas, G. H. Crystal growth of calcium phosphates in the presence of magnesium ions. Langmuir. 1985, 1, 119-122.
doi: 10.1021/la00061a019 URL |
| [62] |
Fox, C.; Ramsoomair, D.; Carter, C. Magnesium: its proven and potential clinical significance. South Med J. 2001, 94, 1195-1201.
doi: 10.1097/00007611-200194120-00014 URL |
| [63] |
Musso, C. G. Magnesium metabolism in health and disease. Int Urol Nephrol. 2009, 41, 357-362.
doi: 10.1007/s11255-009-9548-7 URL |
| [64] | Institute of Medicine Standing Committee on the Scientific Evaluation of Dietary Reference, I. The National Academies Collection: Reports funded by National Institutes of Health. In Dietary Reference Intakes for Calcium, Phosphorus, Magnesium, Vitamin D, and Fluoride, National Academies Press (US): Washington (DC), 1997. |
| [65] | Office of Dietary Supplements, National Institutes of Health. Magnesium: fact sheet for health professionals. https://ods.od.nih.gov/factsheets/Magnesium-HealthProfessional/ . Accessed May 1, 2021. |
| [66] |
Windhagen, H.; Radtke, K.; Weizbauer, A.; Diekmann, J.; Noll, Y.; Kreimeyer, U.; Schavan, R.; Stukenborg-Colsman, C.; Waizy, H. Biodegradable magnesium-based screw clinically equivalent to titanium screw in hallux valgus surgery: short term results of the first prospective, randomized, controlled clinical pilot study. Biomed Eng Online. 2013, 12, 62.
doi: 10.1186/1475-925X-12-62 URL |
| [67] |
Willbold, E.; Kaya, A. A.; Kaya, R. A.; Beckmann, F.; Witte, F. Corrosion of magnesium alloy AZ31 screws is dependent on the implantation site. Mater Sci Eng B. 2011, 176, 1835-1840.
doi: 10.1016/j.mseb.2011.02.010 URL |
| [68] |
Thomann, M.; Krause, C.; Bormann, D.; von der Höh, N.; Windhagen, H.; Meyer-Lindenberg, A. Comparison of the resorbable magnesium. alloys LAE442 und MgCa0.8 concerning their mechanical properties, their progress of degradation and the bone-implant-contact after 12 months implantation duration in a rabbit model. Materialwiss Werkstofftech. 2009, 40, 82-87.
doi: 10.1002/mawe.v40:1/2 URL |
| [69] | Gilbert, S. G. A small dose of toxicology: the health effects of common chemicals. CRC Press: 2004. |
| [70] | World Health Organization, International Atomic Energy Agency & Food and Agriculture Organization of the United Nations. Trace elements in human nutrition and health. World Health Organization: Geneva, 1996. |
| [71] | Alzheimer’s Society. Aluminium, metals and dementia. https://www.alzheimers.org.uk/about-dementia/risk-factors-and-prevention/metals-and-dementia . Accessed May 1, 2021. |
| [72] |
Exley, C. The coordination chemistry of aluminium in neurodegenerative disease. Coord Chem Rev. 2012, 256, 2142-2146.
doi: 10.1016/j.ccr.2012.02.020 URL |
| [73] |
Exley, C. What is the risk of aluminium as a neurotoxin? Expert Rev Neurother. 2014, 14, 589-591.
doi: 10.1586/14737175.2014.915745 URL |
| [74] |
Liu, X.; Shan, D.; Song, Y.; Han, E.H. Influence of yttrium element on the corrosion behaviors of Mg-Y binary magnesium alloy. J Magnes Alloys. 2017, 5, 26-34.
doi: 10.1016/j.jma.2016.12.002 URL |
| [75] |
Miller, P. L.; Shaw, B. A.; Wendt, R. G.; Moshier, W. C. Assessing the corrosion resistance of nonequilibrium magnesium-yttrium alloys. Corrosion. 1995, 51, 922-931.
doi: 10.5006/1.3293568 URL |
| [76] |
McCarty, M. F. Reported antiatherosclerotic activity of silicon may reflect increased endothelial synthesis of heparan sulfate proteoglycans. Med Hypotheses. 1997, 49, 175-176.
doi: 10.1016/S0306-9877(97)90224-3 URL |
| [77] |
Pors Nielsen, S. The biological role of strontium. Bone. 2004, 35, 583-588.
doi: 10.1016/j.bone.2004.04.026 URL |
| [78] | Avedesian, M.; Baker, H. ASM specialty handbook: magnesium and magnesium alloys. ASM International: 1999. |
| [79] | Aa, N.; Clark, J. Phase diagram of binary magnesium alloys. ASM International Monograph Series onAlloy Phase Diagrams. 1988. |
| [80] | Purcell, F. K.; Kotz, C. J. 4.3: Electrochemical potentials. https://chem.libretexts.org/Bookshelves/Inorganic_Chemistry/Book%3A_Introduction_to_Inorganic_Chemistry/04%3A_Redox_Stability_and_Redox_Reactions/4.03%3A_Electrochemical_Potentials . Accessed June 4, 2021. |
| [81] | CrystalMaker Software Ltd. Elements, atomic radii and the periodic table: How big is an atom? Why does its size vary? How can we show this in CrystalMaker? http://crystalmaker.com/support/tutorials/atomic-radii/index.html . Accessed May 2, 2021. |
| [82] |
Witte, F.; Hort, N.; Vogt, C.; Cohen, S.; Kainer, K. U.; Willumeit, R.; Feyerabend, F. Degradable biomaterials based on magnesium corrosion. Curr Opin Solid State Mater Sci. 2008, 12, 63-72.
doi: 10.1016/j.cossms.2009.04.001 URL |
| [83] | Kirkland, N. T.; Staiger, M. P.; Nisbet, D.; Davies, C. H. J.; Birbilis, N. Performance-driven design of Biocompatible Mg alloys. JOM. 2011, 63, 28-34. |
| [84] |
Chen, Y.; Xu, Z.; Smith, C.; Sankar, J. Recent advances on the development of magnesium alloys for biodegradable implants. Acta Biomater. 2014, 10, 4561-4573.
doi: 10.1016/j.actbio.2014.07.005 URL |
| [85] | Lenntech. Recommended daily intake of vitamins and minerals. https://www.lenntech.com/recommended-daily-intake.htm . Accessed May 2, 2021. |
| [86] |
Easton, M. A.; StJohn, D. H. A model of grain refinement incorporating alloy constitution and potency of heterogeneous nucleant particles. Acta Mater. 2001, 49, 1867-1878.
doi: 10.1016/S1359-6454(00)00368-2 URL |
| [87] |
Easton, M.; StJohn, D. Grain refinement of aluminum alloys: Part I. the nucleant and solute paradigms—a review of the literature. Metall Mater Trans A. 1999, 30, 1613-1623.
doi: 10.1007/s11661-999-0098-5 URL |
| [88] |
Easton, M.; StJohn, D. Grain refinement of aluminum alloys: Part II. Confirmation of, and a mechanism for, the solute paradigm. Metall Mater Trans A. 1999, 30, 1625-1633.
doi: 10.1007/s11661-999-0099-4 URL |
| [89] |
StJohn, D. H.; Qian, M.; Easton, M. A.; Cao, P.; Hildebrand, Z. Grain refinement of magnesium alloys. Metall Mater Trans A. 2005, 36, 1669-1679.
doi: 10.1007/s11661-005-0030-6 URL |
| [90] |
Lee, Y. C.; Dahle, A. K.; StJohn, D. H. The role of solute in grain refinement of magnesium. Metall Mater Trans A. 2000, 31, 2895-2906.
doi: 10.1007/BF02830349 URL |
| [91] |
Song, G. L.; Atrens, A. Corrosion mechanisms of magnesium alloys. Adv Eng Mater. 1999, 1, 11-33.
doi: 10.1002/(ISSN)1527-2648 URL |
| [92] |
Velikokhatnyi, O. I.; Kumta, P. N. First-principles studies on alloying and simplified thermodynamic aqueous chemical stability of calcium-, zinc-, aluminum-, yttrium- and iron-doped magnesium alloys. Acta Biomater. 2010, 6, 1698-1704.
doi: 10.1016/j.actbio.2009.08.016 URL |
| [93] |
Li, H.; Wang, P.; Lin, G.; Huang, J. The role of rare earth elements in biodegradable metals: A review. Acta Biomater. 2021, 129, 33-42.
doi: 10.1016/j.actbio.2021.05.014 URL |
| [94] |
Zhang, X.; Yuan, G.; Mao, L.; Niu, J.; Ding, W. Biocorrosion properties of as-extruded Mg-Nd-Zn-Zr alloy compared with commercial AZ31 and WE43 alloys. Mater Lett. 2012, 66, 209-211.
doi: 10.1016/j.matlet.2011.08.079 URL |
| [95] |
Wang, L.; Li, J. B.; Li, L.; Nie, K. B.; Zhang, J. S.; Yang, C. W.; Yan, P. W.; Liu, Y. P.; Xu, C. X. Microstructure, mechanical and bio-corrosion properties of Mg-Zn-Zr alloys with minor Ca addition. Mater Sci Technol. 2017, 33, 9-16.
doi: 10.1080/02670836.2016.1152348 URL |
| [96] |
Yin, D. S.; Zhang, E. L.; Zeng, S. Y. Effect of Zn on mechanical property and corrosion property of extruded Mg-Zn-Mn alloy. Trans Nonferrous Metals Soc China. 2008, 18, 763-768.
doi: 10.1016/S1003-6326(08)60131-4 URL |
| [97] |
Kang, Y. H.; Wu, D.; Chen, R. S.; Han, E. H. Microstructures and mechanical properties of the age hardened Mg-4.2Y-2.5Nd-1Gd-0.6Zr (WE43) microalloyed with Zn. J Magnes Alloys. 2014, 2, 109-115.
doi: 10.1016/j.jma.2014.01.010 URL |
| [98] |
Hänzi, A. C.; Gerber, I.; Schinhammer, M.; Löffler, J. F.; Uggowitzer, P. J. On the in vitro and in vivo degradation performance and biological response of new biodegradable Mg-Y-Zn alloys. Acta Biomater. 2010, 6, 1824-1833.
doi: 10.1016/j.actbio.2009.10.008 URL |
| [99] |
Peng, Q.; Huang, Y.; Zhou, L.; Hort, N.; Kainer, K. U. Preparation and properties of high purity Mg-Y biomaterials. Biomaterials. 2010, 31, 398-403.
doi: 10.1016/j.biomaterials.2009.09.065 URL |
| [100] |
Lu, Y.; Bradshaw, A. R.; Chiu, Y. L.; Jones, I. P. The role of β1’ precipitates in the bio-corrosion performance of Mg-3Zn in simulated body fluid. J Alloys Compd. 2014, 614, 345-352.
doi: 10.1016/j.jallcom.2014.06.078 URL |
| [101] |
Cheng, M.; Chen, J.; Yan, H.; Su, B.; Yu, Z.; Xia, W.; Gong, X. Effects of minor Sr addition on microstructure, mechanical and bio-corrosion properties of the Mg-5Zn based alloy system. J Alloys Compd. 2017, 691, 95-102.
doi: 10.1016/j.jallcom.2016.08.164 URL |
| [102] |
Lu, Y.; Chiu, Y. L.; Jones, I. P. Three-dimensional analysis of the microstructure and bio-corrosion of Mg-Zn and Mg-Zn-Ca alloys. Mater Charact. 2016, 112, 113-121.
doi: 10.1016/j.matchar.2015.12.008 URL |
| [103] |
Li, Z.; Gu, X.; Lou, S.; Zheng, Y. The development of binary Mg-Ca alloys for use as biodegradable materials within bone. Biomaterials. 2008, 29, 1329-1344.
doi: 10.1016/j.biomaterials.2007.12.021 URL |
| [104] |
Sun, Y.; Zhang, B.; Wang, Y.; Geng, L.; Jiao, X. Preparation and characterization of a new biomedical Mg-Zn-Ca alloy. Mater Des. 2012, 34, 58-64.
doi: 10.1016/j.matdes.2011.07.058 URL |
| [105] |
Lu, Y.; Ding, R. G.; Chiu, Y. L.; Jones, I. P. Tomographic investigation of the effects of second phases on the biodegradation and nano-mechanical performance of a Mg-Zn-Ca alloy. Materialia. 2018, 4, 1-9.
doi: 10.1016/j.mtla.2018.08.035 URL |
| [106] |
Zhang, E.; Yang, L.; Xu, J.; Chen, H. Microstructure, mechanical properties and bio-corrosion properties of Mg-Si(-Ca, Zn) alloy for biomedical application. Acta Biomater. 2010, 6, 1756-1762.
doi: 10.1016/j.actbio.2009.11.024 URL |
| [107] | Zhao, C.; Pan, F.; Zhang, L.; Pan, H.; Song, K.; Tang, A. Microstructure, mechanical properties, bio-corrosion properties and cytotoxicity of as-extruded Mg-Sr alloys. Mater Sci Eng CMater Biol Appl. 2017, 70, 1081-1088. |
| [108] |
Wen, Z.; Wu, C.; Dai, C.; Yang, F. Corrosion behaviors of Mg and its alloys with different Al contents in a modified simulated body fluid. J Alloys Compd. 2009, 488, 392-399.
doi: 10.1016/j.jallcom.2009.08.147 URL |
| [109] |
Witte, F.; Fischer, J.; Nellesen, J.; Crostack, H. A.; Kaese, V.; Pisch, A.; Beckmann, F.; Windhagen, H. In vitro and in vivo corrosion measurements of magnesium alloys. Biomaterials. 2006, 27, 1013-1018.
doi: 10.1016/j.biomaterials.2005.07.037 URL |
| [110] |
Zhang, B.; Hou, Y.; Wang, X.; Wang, Y.; Geng, L. Mechanical properties, degradation performance and cytotoxicity of Mg-Zn-Ca biomedical alloys with different compositions. Mater Sci Eng C. 2011, 31, 1667-1673.
doi: 10.1016/j.msec.2011.07.015 URL |
| [111] | Lu, Y.; Bradshaw, A. R.; Chiu, Y. L.; Jones, I. P. Effects of secondary phase and grain size on the corrosion of biodegradable Mg-Zn-Ca alloys. Mater Sci Eng CMater Biol Appl. 2015, 48, 480-486. |
| [112] |
Cho, D. H.; Lee, B. W.; Park, J. Y.; Cho, K. M.; Park, I. M. Effect of Mn addition on corrosion properties of biodegradable Mg-4Zn-0.5Ca-xMn alloys. J Alloys Compd. 2017, 695, 1166-1174.
doi: 10.1016/j.jallcom.2016.10.244 URL |
| [113] |
Zhang, E.; He, W.; Du, H.; Yang, K. Microstructure, mechanical properties and corrosion properties of Mg-Zn-Y alloys with low Zn content. Mater Sci Eng A. 2008, 488, 102-111.
doi: 10.1016/j.msea.2007.10.056 URL |
| [114] |
Zhang, X.; Wu, Y.; Xue, Y.; Wang, Z.; Yang, L. Biocorrosion behavior and cytotoxicity of a Mg-Gd-Zn-Zr alloy with long period stacking ordered structure. Mater Lett. 2012, 86, 42-45.
doi: 10.1016/j.matlet.2012.07.030 URL |
| [115] |
Krause, A.; von der Höh, N.; Bormann, D.; Krause, C.; Bach, F. W.; Windhagen, H.; Meyer-Lindenberg, A. Degradation behaviour and mechanical properties of magnesium implants in rabbit tibiae. J Mater Sci. 2010, 45, 624-632.
doi: 10.1007/s10853-009-3936-3 URL |
| [116] |
Gu, X. N.; Xie, X. H.; Li, N.; Zheng, Y. F.; Qin, L. In vitro and in vivo studies on a Mg-Sr binary alloy system developed as a new kind of biodegradable metal. Acta Biomater. 2012, 8, 2360-2374.
doi: 10.1016/j.actbio.2012.02.018 URL |
| [117] |
Zhang, S.; Zhang, X.; Zhao, C.; Li, J.; Song, Y.; Xie, C.; Tao, H.; Zhang, Y.; He, Y.; Jiang, Y.; Bian, Y. Research on an Mg-Zn alloy as a degradable biomaterial. Acta Biomater. 2010, 6, 626-640.
doi: 10.1016/j.actbio.2009.06.028 URL |
| [118] |
Li, N.; Zheng, Y. Novel magnesium alloys developed for biomedical application: a review. J Mater Sci Technol. 2013, 29, 489-502.
doi: 10.1016/j.jmst.2013.02.005 URL |
| [119] |
Song, G.; Atrens, A.; Dargusch, M. Influence of microstructure on the corrosion of diecast AZ91D. Corros Sci. 1998, 41, 249-273.
doi: 10.1016/S0010-938X(98)00121-8 URL |
| [120] |
Aung, N. N.; Zhou, W. Effect of grain size and twins on corrosion behaviour of AZ31B magnesium alloy. Corros Sci. 2010, 52, 589-594.
doi: 10.1016/j.corsci.2009.10.018 URL |
| [121] |
Hamu, G. B.; Eliezer, D.; Wagner, L. The relation between severe plastic deformation microstructure and corrosion behavior of AZ31 magnesium alloy. J Alloys Compd. 2009, 468, 222-229.
doi: 10.1016/j.jallcom.2008.01.084 URL |
| [122] |
Birbilis, N.; Ralston, K. D.; Virtanen, S.; Fraser, H. L.; Davies, C. H. J. Grain character influences on corrosion of ECAPed pure magnesium. Corros Eng Sci Technol. 2010, 45, 224-230.
doi: 10.1179/147842209X12559428167805 URL |
| [123] |
Ralston, K. D.; Birbilis, N.; Davies, C. H. J. Revealing the relationship between grain size and corrosion rate of metals. Scripta Mater. 2010, 63, 1201-1204.
doi: 10.1016/j.scriptamat.2010.08.035 URL |
| [124] |
op’t Hoog, C.; Birbilis, N.; Estrin, Y. Corrosion of pure Mg as a function of grain size and processing route. Adv Eng Mater. 2008, 10, 579-582.
doi: 10.1002/(ISSN)1527-2648 URL |
| [125] |
Jang, Y. H.; Kim, S. S.; Yim, C. D.; Lee, C. G.; Kim, S. J. Corrosion behaviour of friction stir welded AZ31B Mg in 3•5%NaCl solution. Corros Eng Sci Technol. 2007, 42, 119-122.
doi: 10.1179/174327807X196834 URL |
| [126] |
Song, G.; StJohn, D. The effect of zirconium grain refinement on the corrosion behaviour of magnesium-rare earth alloy MEZ. J Light Metals. 2002, 2, 1-16.
doi: 10.1016/S1471-5317(02)00008-1 URL |
| [127] |
Song, G. Recent progress in corrosion and protection of magnesium alloys. Adv Eng Mater. 2005, 7, 563-586.
doi: 10.1002/(ISSN)1527-2648 URL |
| [128] |
Lunder, O.; Lein, J. E.; Aune, T. K.; Nisancioglu, K. The role of Mg17Al12 phase in the corrosion of Mg alloy AZ91. Corrosion. 1989, 45, 741-748.
doi: 10.5006/1.3585029 URL |
| [129] |
Beldjoudi, T.; Fiaud, C.; Robbiola, L. Influence of homogenization and artificial aging heat treatments on corrosion behavior of Mg-Al alloys. Corrosion. 1993, 49, 738-745.
doi: 10.5006/1.3316126 URL |
| [130] |
Pardo, A.; Merino, M. C.; Coy, A. E.; Arrabal, R.; Viejo, F.; Matykina, E. Corrosion behaviour of magnesium/aluminium alloys in 3.5wt.% NaCl. Corros Sci. 2008, 50, 823-834.
doi: 10.1016/j.corsci.2007.11.005 URL |
| [131] | Lunder, O.; Videm, M.; Nisancioglu, K. Corrosion resistant magnesium alloys. SAE Trans. 1995, 104, 352-357. |
| [132] |
Srinivasan, A.; Ningshen, S.; Kamachi Mudali, U.; Pillai, U. T. S.; Pai, B. C. Influence of Si and Sb additions on the corrosion behavior of AZ91 magnesium alloy. Intermetallics. 2007, 15, 1511-1517.
doi: 10.1016/j.intermet.2007.05.012 URL |
| [133] |
Mao, L.; Shen, L.; Chen, J.; Zhang, X.; Kwak, M.; Wu, Y.; Fan, R.; Zhang, L.; Pei, J.; Yuan, G.; Song, C.; Ge, J.; Ding, W. A promising biodegradable magnesium alloy suitable for clinical vascular stent application. Sci Rep. 2017, 7, 46343.
doi: 10.1038/srep46343 URL |
| [134] |
Zhang, X.; Yuan, G.; Mao, L.; Niu, J.; Fu, P.; Ding, W. Effects of extrusion and heat treatment on the mechanical properties and biocorrosion behaviors of a Mg-Nd-Zn-Zr alloy. J Mech Behav Biomed Mater. 2012, 7, 77-86.
doi: 10.1016/j.jmbbm.2011.05.026 URL |
| [135] |
Zong, Y.; Yuan, G.; Zhang, X.; Mao, L.; Niu, J.; Ding, W. Comparison of biodegradable behaviors of AZ31 and Mg-Nd-Zn-Zr alloys in Hank’s physiological solution. Mater Sci Eng B. 2012, 177, 395-401.
doi: 10.1016/j.mseb.2011.09.042 URL |
| [136] |
Andrei, M.; Eliezer, A.; Bonora, P. L.; Gutman, E. M. DC and AC polarisation study on magnesium alloys Influence of the mechanical deformation. Mater Corros. 2002, 53, 455-461.
doi: 10.1002/(ISSN)1521-4176 URL |
| [137] | Xin, R. L.; Wang, M. Y.; Gao, J. C.; Liu, P.; Liu, Q. Effect of microstructure and texture on corrosion resistance of magnesium alloy. Mater Sci Forum. 2009,610-613,1160-1163. |
| [138] |
Schmutz, P.; Guillaumin, V.; Lillard, R. S.; Lillard, J. A.; Frankel, G. S. Influence of dichromate ions on corrosion processes on pure magnesium. J Electrochem Soc. 2003, 150, B99.
doi: 10.1149/1.1554721 URL |
| [139] |
Gu, X.; Zheng, Y.; Zhong, S.; Xi, T.; Wang, J.; Wang, W. Corrosion of, and cellular responses to Mg-Zn-Ca bulk metallic glasses. Biomaterials. 2010, 31, 1093-1103.
doi: 10.1016/j.biomaterials.2009.11.015 URL |
| [140] |
Zberg, B.; Uggowitzer, P. J.; Löffler, J. F. MgZnCa glasses without clinically observable hydrogen evolution for biodegradable implants. Nat Mater. 2009, 8, 887-891.
doi: 10.1038/nmat2542 URL |
| [141] |
Zhang, X. L.; Chen, G.; Bauer, T. Mg-based bulk metallic glass composite with high bio-corrosion resistance and excellent mechanical properties. Intermetallics. 2012, 29, 56-60.
doi: 10.1016/j.intermet.2012.04.018 URL |
| [142] |
Zberg, B.; Arata, E. R.; Uggowitzer, P. J.; Löffler, J. F. Tensile properties of glassy MgZnCa wires and reliability analysis using Weibull statistics. Acta Mater. 2009, 57, 3223-3231.
doi: 10.1016/j.actamat.2009.03.028 URL |
| [143] |
Park, E. S.; Kim, W. T.; Kim, D. H. Bulk glass formation in Mg-Cu-Ag-Y-Gd alloy. Mater Trans. 2004, 45, 2474-2477.
doi: 10.2320/matertrans.45.2474 URL |
| [144] |
Sun, Y.; Zhang, H. F.; Fu, H. M.; Wang, A. M.; Hu, Z. Q. Mg-Cu-Ag-Er bulk metallic glasses with high glass forming ability and compressive strength. Mater Sci Eng A. 2009, 502, 148-152.
doi: 10.1016/j.msea.2008.10.008 URL |
| [145] |
Wessels, V.; Le Mené, G.; Fischerauer, S. F.; Kraus, T.; Weinberg, A. M.; Uggowitzer, P. J.; Löffler, J. F. In vivo performance and structural relaxation of biodegradable bone implants made from Mg Zn Ca bulk metallic glasses. Adv Eng Mater. 2012, 14, B357-B364.
doi: 10.1002/adem.201180200 URL |
| [146] | Yu, H. J.; Wang, J. Q.; Shi, X. T.; Louzguine-Luzgin, D. V.; Wu, H.-K.; Perepezko, J. H. Ductile biodegradable Mg-based metallic glasses with excellent biocompatibility. Adv Funct Mater. 2013, 23, 4793-4800. |
| [147] |
Ashby, M. F.; Greer, A. L. Metallic glasses as structural materials. Scripta Mater. 2006, 54, 321-326.
doi: 10.1016/j.scriptamat.2005.09.051 URL |
| [148] |
Razavi, M.; Fathi, M. H.; Meratian, M. Microstructure, mechanical properties and bio-corrosion evaluation of biodegradable AZ91-FA nanocomposites for biomedical applications. Mater Sci Eng A. 2010, 527, 6938-6944.
doi: 10.1016/j.msea.2010.07.063 URL |
| [149] |
Lei, T.; Tang, W.; Cai, S. H.; Feng, F. F.; Li, N. F. On the corrosion behaviour of newly developed biodegradable Mg-based metal matrix composites produced by in situ reaction. Corros Sci. 2012, 54, 270-277.
doi: 10.1016/j.corsci.2011.09.027 URL |
| [150] |
Feng, A.; Han, Y. The microstructure, mechanical and corrosion properties of calcium polyphosphate reinforced ZK60A magnesium alloy composites. J Alloys Compd. 2010, 504, 585-593.
doi: 10.1016/j.jallcom.2010.06.013 URL |
| [151] |
Gu, X.; Zhou, W.; Zheng, Y.; Dong, L.; Xi, Y.; Chai, D. Microstructure, mechanical property, bio-corrosion and cytotoxicity evaluations of Mg/HA composites. Mater Sci Eng C. 2010, 30, 827-832.
doi: 10.1016/j.msec.2010.03.016 URL |
| [152] |
Ye, X.; Chen, M.; Yang, M.; Wei, J.; Liu, D. In vitro corrosion resistance and cytocompatibility of nano-hydroxyapatite reinforced Mg-Zn-Zr composites. J Mater Sci Mater Med. 2010, 21, 1321-1328.
doi: 10.1007/s10856-009-3954-3 URL |
| [153] |
Liu, D.; Zuo, Y.; Meng, W.; Chen, M.; Fan, Z. Fabrication of biodegradable nano-sized β-TCP/Mg composite by a novel melt shearing technology. Mater Sci Eng C. 2012, 32, 1253-1258.
doi: 10.1016/j.msec.2012.03.017 URL |
| [154] |
Wang, T.; Lin, C.; Batalu, D.; Zhang, L.; Hu, J.; Lu, W. In vitro study of the PLLA-Mg65Zn30Ca5 composites as potential biodegradable materials for bone implants. J Magnes Alloys. 2021. doi: 10.1016/j.jma.2020.12.014.
doi: 10.1016/j.jma.2020.12.014 URL |
| [155] | Antoniac, I. V.; Antoniac, A.; Vasile, E.; Tecu, C.; Fosca, M.; Yankova, V. G.; Rau, J. V. In vitro characterization of novel nanostructured collagen-hydroxyapatite composite scaffolds doped with magnesium with improved biodegradation rate for hard tissue regeneration. Bioact Mater. 2021, 6, 3383-3395. |
| [156] | Shen, J.; Chen, B.; Zhai, X.; Qiao, W.; Wu, S.; Liu, X.; Zhao, Y.; Ruan, C.; Pan, H.; Chu, P. K.; Cheung, K. M. C.; Yeung, K. W. K. Stepwise 3D-spatio-temporal magnesium cationic niche: Nanocomposite scaffold mediated microenvironment for modulating intramembranous ossification. Bioact Mater. 2021, 6, 503-519. |
| [157] | Shen, J.; Wang, W.; Zhai, X.; Chen, B.; Qiao, W.; Li, W.; Li, P.; Zhao, Y.; Meng, Y.; Qian, S.; Liu, X.; Chu, P. K.; Yeung, K. W. K. 3D-printed nanocomposite scaffolds with tunable magnesium ionic microenvironment induce in situ bone tissue regeneration. Appl Mater Today. 2019, 16, 493-507. |
| [158] |
Wang, X.; Xu, S.; Zhou, S.; Xu, W.; Leary, M.; Choong, P.; Qian, M.; Brandt, M.; Xie, Y. M. Topological design and additive manufacturing of porous metals for bone scaffolds and orthopaedic implants: A review. Biomaterials. 2016, 83, 127-141.
doi: 10.1016/j.biomaterials.2016.01.012 URL |
| [159] |
Zou, X.; Li, H.; Bünger, M.; Egund, N.; Lind, M.; Bünger, C. Bone ingrowth characteristics of porous tantalum and carbon fiber interbody devices: an experimental study in pigs. Spine J. 2004, 4, 99-105.
doi: 10.1016/S1529-9430(03)00407-8 URL |
| [160] |
Pamula, E.; Bacakova, L.; Filova, E.; Buczynska, J.; Dobrzynski, P.; Noskova, L.; Grausova, L. The influence of pore size on colonization of poly(L-lactide-glycolide) scaffolds with human osteoblast-like MG 63 cells in vitro. J Mater Sci Mater Med. 2008, 19, 425-435.
doi: 10.1007/s10856-007-3001-1 URL |
| [161] | Pamula, E.; Filová, E.; Bacáková, L.; Lisá, V.; Adamczyk, D. Resorbable polymeric scaffolds for bone tissue engineering: the influence of their microstructure on the growth of human osteoblast-like MG 63 cells. J Biomed Mater Res A. 2009, 89, 432-443. |
| [162] |
Lefebvre, L. P.; Banhart, J.; Dunand, D. C. Porous metals and metallic foams: current status and recent developments. Adv Eng Mater. 2008, 10, 775-787.
doi: 10.1002/adem.v10:9 URL |
| [163] |
Cheng, M. Q.; Wahafu, T.; Jiang, G. F.; Liu, W.; Qiao, Y. Q.; Peng, X. C.; Cheng, T.; Zhang, X. L.; He, G.; Liu, X. Y. A novel open-porous magnesium scaffold with controllable microstructures and properties for bone regeneration. Sci Rep. 2016, 6, 24134.
doi: 10.1038/srep24134 URL |
| [164] | Razavi, M.; Fathi, M.; Savabi, O.; Vashaee, D.; Tayebi, L. In vitro study of nanostructured diopside coating on Mg alloy orthopedic implants. Mater Sci Eng CMater Biol Appl. 2014, 41, 168-177. |
| [165] |
Li, Y.; Zhou, J.; Pavanram, P.; Leeflang, M. A.; Fockaert, L. I.; Pouran, B.; Tümer, N.; Schröder, K. U.; Mol, J. M. C.; Weinans, H.; Jahr, H.; Zadpoor, A. A. Additively manufactured biodegradable porous magnesium. Acta Biomater. 2018, 67, 378-392.
doi: 10.1016/j.actbio.2017.12.008 URL |
| [166] | Geng, F.; Tan, L.; Zhang, B.; Wu, C.; He, Y.; Yang, J.; Yang, K. Study on β-TCP coated porous Mg as a bone tissue engineering scaffold material. J Mater Sci Technol. 2009, 25, 123-129. |
| [167] |
Tan, L.; Gong, M.; Zheng, F.; Zhang, B.; Yang, K. Study on compression behavior of porous magnesium used as bone tissue engineering scaffolds. Biomed Mater. 2009, 4, 015016.
doi: 10.1088/1748-6041/4/1/015016 URL |
| [168] | Witte, F.; Ulrich, H.; Palm, C.; Willbold, E. Biodegradable magnesium scaffolds: Part II: peri-implant bone remodeling. J Biomed Mater Res A. 2007, 81, 757-765. |
| [169] |
Gu, X. N.; Zhou, W. R.; Zheng, Y. F.; Liu, Y.; Li, Y. X. Degradation and cytotoxicity of lotus-type porous pure magnesium as potential tissue engineering scaffold material. Mater Lett. 2010, 64, 1871-1874.
doi: 10.1016/j.matlet.2010.06.015 URL |
| [170] |
Badekila, A. K.; Kini, S.; Jaiswal, A. K. Fabrication techniques of biomimetic scaffolds in three-dimensional cell culture: A review. J Cell Physiol. 2021, 236, 741-762.
doi: 10.1002/jcp.v236.2 URL |
| [171] |
Zhao, D.; Witte, F.; Lu, F.; Wang, J.; Li, J.; Qin, L. Current status on clinical applications of magnesium-based orthopaedic implants: A review from clinical translational perspective. Biomaterials. 2017, 112, 287-302.
doi: 10.1016/j.biomaterials.2016.10.017 URL |
| [172] | ASTM B557. Standard test methods for tension testing wrought and castaluminum- and magnesium-alloy products. 2015. |
| [173] | International Organization of Standards. ISO 6892-1:Metallic materials-Tensile testing-Part1: Method of test at room temperature. 2019. |
| [174] | ASTM WK61103. New guide for corrosion fatigueevaluation of absorbablemetals. 2017. |
| [175] |
Jalota, S.; Bhaduri, S. B.; Tas, A. C. Using a synthetic body fluid (SBF) solution of 27 mM HCO3 - to make bone substitutes more osteointegrative. Mater Sci Eng C. 2008, 28, 129-140.
doi: 10.1016/j.msec.2007.10.058 URL |
| [176] |
Zhang, X.; Yuan, G.; Niu, J.; Fu, P.; Ding, W. Microstructure, mechanical properties, biocorrosion behavior, and cytotoxicity of as-extruded Mg-Nd-Zn-Zr alloy with different extrusion ratios. J Mech Behav Biomed Mater. 2012, 9, 153-162.
doi: 10.1016/j.jmbbm.2012.02.002 URL |
| [177] |
Wang, H.; Estrin, Y.; Fu, H.; Song, G.; Zúberová, Z. The Effect of pre-processing and grain structure on the bio-corrosion and fatigue resistance of magnesium alloy AZ31. Adv Eng Mater. 2007, 9, 967-972.
doi: 10.1002/(ISSN)1527-2648 URL |
| [178] |
Witte, F.; Feyerabend, F.; Maier, P.; Fischer, J.; Störmer, M.; Blawert, C.; Dietzel, W.; Hort, N. Biodegradable magnesium-hydroxyapatite metal matrix composites. Biomaterials. 2007, 28, 2163-2174.
doi: 10.1016/j.biomaterials.2006.12.027 URL |
| [179] |
Wang, Y.; Wei, M.; Gao, J. Improve corrosion resistance of magnesium in simulated body fluid by dicalcium phosphate dihydrate coating. Mater Sci Eng C. 2009, 29, 1311-1316.
doi: 10.1016/j.msec.2008.09.051 URL |
| [180] |
Kirkland, N. T.; Lespagnol, J.; Birbilis, N.; Staiger, M. P. A survey of bio-corrosion rates of magnesium alloys. Corros Sci. 2010, 52, 287-291.
doi: 10.1016/j.corsci.2009.09.033 URL |
| [181] |
Hort, N.; Huang, Y.; Fechner, D.; Störmer, M.; Blawert, C.; Witte, F.; Vogt, C.; Drücker, H.; Willumeit, R.; Kainer, K. U.; Feyerabend, F. Magnesium alloys as implant materials principles of property design for Mg-RE alloys. Acta Biomater. 2010, 6, 1714-1725.
doi: 10.1016/j.actbio.2009.09.010 URL |
| [182] | Song, G. L.; Atrens, A.; St.John, D. H. Magnesium technology. New Orleans, 2001. |
| [183] |
Alvarez-Lopez, M.; Pereda, M. D.; del Valle, J. A.; Fernandez-Lorenzo, M.; Garcia-Alonso, M. C.; Ruano, O. A.; Escudero, M. L. Corrosion behaviour of AZ31 magnesium alloy with different grain sizes in simulated biological fluids. Acta Biomater. 2010, 6, 1763-1771.
doi: 10.1016/j.actbio.2009.04.041 URL |
| [184] |
He, W.; Zhang, E.; Yang, K. Effect of Y on the bio-corrosion behavior of extruded Mg-Zn-Mn alloy in Hank’s solution. Mater Sci Eng C. 2010, 30, 167-174.
doi: 10.1016/j.msec.2009.09.014 URL |
| [185] |
Kannan, M. B. Influence of microstructure on the in-vitro degradation behaviour of magnesium alloys. Mater Lett. 2010, 64, 739-742.
doi: 10.1016/j.matlet.2010.01.022 URL |
| [186] |
Kirkland, N. T.; Birbilis, N.; Staiger, M. P. Assessing the corrosion of biodegradable magnesium implants: a critical review of current methodologies and their limitations. Acta Biomater. 2012, 8, 925-936.
doi: 10.1016/j.actbio.2011.11.014 URL |
| [187] |
Song, G.; Atrens, A.; Wu, X.; Zhang, B. Corrosion behaviour of AZ21, AZ501 and AZ91 in sodium chloride. Corros Sci. 1998, 40, 1769-1791.
doi: 10.1016/S0010-938X(98)00078-X URL |
| [188] |
Parai, R.; Bandyopadhyay-Ghosh, R. S. Engineered bio-nanocomposite magnesium scaffold for bone tissue regeneration. J Mech Behav Biomed Mater. 2019, 96, 45-52.
doi: 10.1016/j.jmbbm.2019.04.019 URL |
| [189] | Dutta, S.; Devi, K. B.; Gupta, S.; Kundu, B.; Balla, V. K.; Roy, M. Mechanical and in vitro degradation behavior of magnesium-bioactive glass composites prepared by SPS for biomedical applications. J Biomed Mater Res BAppl Biomater. 2019, 107, 352-365. |
| [190] | Chen, K.; Xie, X.; Tang, H.; Sun, H.; Qin, L.; Zheng, Y.; Gu, X.; Fan, Y. In vitro and in vivo degradation behavior of Mg-2Sr-Ca and Mg-2Sr-Zn alloys. Bioact Mater. 2020, 5, 275-285. |
| [191] |
Heublein, B.; Rohde, R.; Kaese, V.; Niemeyer, M.; Hartung, W.; Haverich, A. Biocorrosion of magnesium alloys: a new principle in cardiovascular implant technology? Heart. 2003, 89, 651-656.
doi: 10.1136/heart.89.6.651 URL |
| [192] |
Kaya, R. A.; Cavuşoğlu, H.; Tanik, C.; Kaya, A. A.; Duygulu, O.; Mutlu, Z.; Zengin, E.; Aydin, Y. The effects of magnesium particles in posterolateral spinal fusion: an experimental in vivo study in a sheep model. J Neurosurg Spine. 2007, 6, 141-149.
doi: 10.3171/spi.2007.6.2.141 URL |
| [193] |
Huang, S.; Wang, B.; Zhang, X.; Lu, F.; Wang, Z.; Tian, S.; Li, D.; Yang, J.; Cao, F.; Cheng, L.; Gao, Z.; Li, Y.; Qin, K.; Zhao, D. High-purity weight-bearing magnesium screw: Translational application in the healing of femoral neck fracture. Biomaterials. 2020, 238, 119829.
doi: 10.1016/j.biomaterials.2020.119829 URL |
| [194] |
Grün, N. G.; Holweg, P.; Tangl, S.; Eichler, J.; Berger, L.; van den Beucken, J.; Löffler, J. F.; Klestil, T.; Weinberg, A. M. Comparison of a resorbable magnesium implant in small and large growing-animal models. Acta Biomater. 2018, 78, 378-386.
doi: 10.1016/j.actbio.2018.07.044 URL |
| [195] | Huang, Q.; Liu, L.; Wu, H.; Li, K.; Li, N.; Liu, Y. The design, development, and in vivo performance of intestinal anastomosis ring fabricated by magnesium-zinc-strontium alloy. Mater Sci Eng CMater Biol Appl. 2020, 106, 110158. |
| [196] | Imwinkelried, T.; Beck, S.; Schaller, B. Pre-clinical testing of human size magnesium implants in miniature pigs: Implant degradation and bone fracture healing at multiple implantation sites. Mater Sci Eng CMater Biol Appl. 2020, 108, 110389. |
| [197] |
Li, W.; Zhou, J.; Xu, Y. Study of the in vitro cytotoxicity testing of medical devices. Biomed Rep. 2015, 3, 617-620.
doi: 10.3892/br.2015.481 URL |
| [198] | International Organization of Standards. ISO 10993-5: biological evaluation of medical devices: tests for in vitro cytotoxicity. |
| [199] |
De Melo, W. M.; Maximiano, W. M.; Antunes, A. A.; Beloti, M. M.; Rosa, A. L.; de Oliveira, P. T. Cytotoxicity testing of methyl and ethyl 2-cyanoacrylate using direct contact assay on osteoblast cell cultures. J Oral Maxillofac Surg. 2013, 71, 35-41.
doi: 10.1016/j.joms.2012.09.002 URL |
| [200] |
Sjögren, G.; Sletten, G.; Dahl, J. E. Cytotoxicity of dental alloys, metals, and ceramics assessed by millipore filter, agar overlay, and MTT tests. J Prosthet Dent. 2000, 84, 229-236.
doi: 10.1067/mpr.2000.107227 URL |
| [201] |
Jin, C. Y.; Zhu, B. S.; Wang, X. F.; Lu, Q. H. Cytotoxicity of titanium dioxide nanoparticles in mouse fibroblast cells. Chem Res Toxicol. 2008, 21, 1871-1877.
doi: 10.1021/tx800179f URL |
| [202] | Liu, L.; Huang, B.; Liu, X.; Yuan, W.; Zheng, Y.; Li, Z.; Yeung, K. W. K.; Zhu, S.; Liang, Y.; Cui, Z.; Wu, S. Photo-controlled degradation of PLGA/Ti(3)C(2) hybrid coating on Mg-Sr alloy using near infrared light. Bioact Mater. 2021, 6, 568-578. |
| [203] |
Han, D.; Li, Y.; Liu, X.; Yeung, K. W. K.; Zheng, Y.; Cui, Z.; Liang, Y.; Li, Z.; Zhu, S.; Wang, X.; Wu, S. Photothermy-strengthened photocatalytic activity of polydopamine-modified metal-organic frameworks for rapid therapy of bacteria-infected wounds. J Mater Sci Technol. 2021, 62, 83-95.
doi: 10.1016/j.jmst.2020.05.055 URL |
| [204] |
Sarkar, K.; Rahaman, M.; Agarwal, S.; Bodhak, S.; Halder, S.; Nandi, S. K.; Roy, M. Degradability and in vivo biocompatibility of doped magnesium phosphate bioceramic scaffolds. Mater Lett. 2020, 259, 126892.
doi: 10.1016/j.matlet.2019.126892 URL |
| [205] |
Kumar, V.; Sarkar, K.; Bavya Devi, K.; Ghosh, D.; Nandi, S. K.; Roy, M. Quantitative assessment of degradation, cytocompatibility, and in vivo bone regeneration of silicon-incorporated magnesium phosphate bioceramics. J Mater Res. 2019, 34, 4024-4036.
doi: 10.1557/jmr.2019.367 URL |
| [206] |
Wang, J.; Cui, L.; Ren, Y.; Zou, Y.; Ma, J.; Wang, C.; Zheng, Z.; Chen, X.; Zeng, R.; Zheng, Y. In vitro and in vivo biodegradation and biocompatibility of an MMT/BSA composite coating upon magnesium alloy AZ31. J Mater Sci Technol. 2020, 47, 52-67.
doi: 10.1016/j.jmst.2020.02.006 URL |
| [207] |
Lin, Z.; Zhao, Y.; Chu, P. K.; Wang, L.; Pan, H.; Zheng, Y.; Wu, S.; Liu, X.; Cheung, T.; Wong, K. M. C.; Yeung, K. W. K. A functionalized TiO(2)/Mg(2)TiO(4) nano-layer on biodegradable magnesium implant enables superior bone-implant integration and bacterial disinfection. Biomaterials. 2019, 219, 119372.
doi: 10.1016/j.biomaterials.2019.119372 URL |
| [208] |
Makkar, P.; Kang, H. J.; Padalhin, A. R.; Faruq, O.; Lee, B. In-vitro and in-vivo evaluation of strontium doped calcium phosphate coatings on biodegradable magnesium alloy for bone applications. Appl Surf Sci. 2020, 510, 145333.
doi: 10.1016/j.apsusc.2020.145333 URL |
| [209] | International Organization of Standards. ISO 10993-12: biological evaluation of medical devices: sample preparation and reference materials. |
| [210] |
Yamamoto, A.; Hiromoto, S. Effect of inorganic salts, amino acids and proteins on the degradation of pure magnesium in vitro. Mater Sci Eng C. 2009, 29, 1559-1568.
doi: 10.1016/j.msec.2008.12.015 URL |
| [211] |
Fischer, J.; Pröfrock, D.; Hort, N.; Willumeit, R.; Feyerabend, F. Improved cytotoxicity testing of magnesium materials. Mater Sci Eng B. 2011, 176, 830-834.
doi: 10.1016/j.mseb.2011.04.008 URL |
| [212] |
Wang, J.; Witte, F.; Xi, T.; Zheng, Y.; Yang, K.; Yang, Y.; Zhao, D.; Meng, J.; Li, Y.; Li, W.; Chan, K.; Qin, L. Recommendation for modifying current cytotoxicity testing standards for biodegradable magnesium-based materials. Acta Biomater. 2015, 21, 237-249.
doi: 10.1016/j.actbio.2015.04.011 URL |
| [213] |
Jablonská, E.; Kubásek, J.; Vojtěch, D.; Ruml, T.; Lipov, J. Test conditions can significantly affect the results of in vitro cytotoxicity testing of degradable metallic biomaterials. Sci Rep. 2021, 11, 6628.
doi: 10.1038/s41598-021-85019-6 URL |
| [214] |
Razavi, M.; Fathi, M.; Savabi, O.; Razavi, S. M.; Heidari, F.; Manshaei, M.; Vashaee, D.; Tayebi, L. In vivo study of nanostructured diopside (CaMgSi2O6) coating on magnesium alloy as biodegradable orthopedic implants. Appl Surf Sci. 2014, 313, 60-66.
doi: 10.1016/j.apsusc.2014.05.130 URL |
| [215] | Mahamood, R. M.; Akinlabi, E. T. Types of functionally graded materials and their areas of application. In Functionally Graded Materials, Mahamood, R. M.; Akinlabi, E. T., eds.; Springer International Publishing: Cham, 2017; pp 9-21. |
| [216] |
Shuai, C.; Cheng, Y.; Yang, Y.; Peng, S.; Yang, W.; Qi, F. Laser additive manufacturing of Zn-2Al part for bone repair: Formability, microstructure and properties. J Alloys Compd. 2019, 798, 606-615.
doi: 10.1016/j.jallcom.2019.05.278 URL |
| [217] |
Ilich, J. Z.; Kerstetter, J. E. Nutrition in bone health revisited: a story beyond calcium. J Am Coll Nutr. 2000, 19, 715-737.
doi: 10.1080/07315724.2000.10718070 URL |
| [218] |
Atkins, G. J.; Welldon, K. J.; Halbout, P.; Findlay, D. M. Strontium ranelate treatment of human primary osteoblasts promotes an osteocyte-like phenotype while eliciting an osteoprotegerin response. Osteoporos Int. 2009, 20, 653-664.
doi: 10.1007/s00198-008-0728-6 URL |
| [219] |
Qin, H.; Zhao, Y.; An, Z.; Cheng, M.; Wang, Q.; Cheng, T.; Wang, Q.; Wang, J.; Jiang, Y.; Zhang, X.; Yuan, G. Enhanced antibacterial properties, biocompatibility, and corrosion resistance of degradable Mg-Nd-Zn-Zr alloy. Biomaterials. 2015, 53, 211-220.
doi: 10.1016/j.biomaterials.2015.02.096 URL |
| [220] | Medical Devices Business Services, I. 3.5mm, 4.5mm Locking Compression Plate (LCP®). https://www.jnjmedicaldevices.com/en-US/product/35mm-45mm-locking-compression-plate-lcpr . Accessed May 10, 2021. |
| [221] |
Buzolin, R. H.; Mohedano, M.; Mendis, C. L.; Mingo, B.; Tolnai, D.; Blawert, C.; Kainer, K. U.; Pinto, H.; Hort, N. As cast microstructures on the mechanical and corrosion behaviour of ZK40 modified with Gd and Nd additions. Mater Sci Eng A. 2017, 682, 238-247.
doi: 10.1016/j.msea.2016.11.022 URL |
| [222] | Zhao, X.; Shi, L. L.; Xu, J. A Comparison of corrosion behavior in saline environment: rare earth metals (Y, Nd, Gd, Dy) for alloying of biodegradable magnesium alloys. J Mater Sci Technol. 2013, 29, 781-787. |
| [223] | Li, Y.; Jahr, H.; Zhang, X. Y.; Leeflang, M. A.; Li, W.; Pouran, B.; Tichelaar, F. D.; Weinans, H.; Zhou, J.; Zadpoor, A. A. Biodegradation-affected fatigue behavior of additively manufactured porous magnesium. Addit Manuf. 2019, 28, 299-311. |
| [224] |
Wei, K.; Gao, M.; Wang, Z.; Zeng, X. Effect of energy input on formability, microstructure and mechanical properties of selective laser melted AZ91D magnesium alloy. Mater Sci Eng A. 2014, 611, 212-222.
doi: 10.1016/j.msea.2014.05.092 URL |
| [225] |
Zhang, B.; Liao, H.; Coddet, C. Effects of processing parameters on properties of selective laser melting Mg-9%Al powder mixture. Mater Des. 2012, 34, 753-758.
doi: 10.1016/j.matdes.2011.06.061 URL |
| [226] |
Shuai, C.; Yang, Y.; Wu, P.; Lin, X.; Liu, Y.; Zhou, Y.; Feng, P.; Liu, X.; Peng, S. Laser rapid solidification improves corrosion behavior of Mg-Zn-Zr alloy. J Alloys Compd. 2017, 691, 961-969.
doi: 10.1016/j.jallcom.2016.09.019 URL |
| [227] |
Wei, K.; Wang, Z.; Zeng, X. Influence of element vaporization on formability, composition, microstructure, and mechanical performance of the selective laser melted Mg-Zn-Zr components. Mater Lett. 2015, 156, 187-190.
doi: 10.1016/j.matlet.2015.05.074 URL |
| [228] |
Salehi, M.; Maleksaeedi, S.; Sapari, M. A. B.; Nai, M. L. S.; Meenashisundaram, G. K.; Gupta, M. Additive manufacturing of magnesium-zinc-zirconium (ZK) alloys via capillary-mediated binderless three-dimensional printing. Mater Des. 2019, 169, 107683.
doi: 10.1016/j.matdes.2019.107683 URL |
| [229] |
He, C.; Bin, S.; Wu, P.; Gao, C.; Feng, P.; Yang, Y.; Liu, L.; Zhou, Y.; Zhao, M.; Yang, S.; Shuai, C. Microstructure evolution and biodegradation behavior of laser rapid solidified Mg-Al-Zn alloy. Metals (Basel). 2017, 7, 105.
doi: 10.3390/met7030105 URL |
| [230] |
Shuai, C.; He, C.; Feng, P.; Guo, W.; Gao, C.; Wu, P.; Yang, Y.; Bin, S. Biodegradation mechanisms of selective laser-melted Mg-xAl-Zn alloy: grain size and intermetallic phase. Virtual Phys Prototyp. 2018, 13, 59-69.
doi: 10.1080/17452759.2017.1408918 URL |
| [1] | Seyed Ataollah Naghavi, Changning Sun, Mahbubeh Hejazi, Maryam Tamaddon, Jibao Zheng, Leilei Wang, Chenrui Zhang, Swastina Nath Varma, Dichen Li, Mehran Moazen, Ling Wang, Chaozong Liu. On the mechanical aspect of additive manufactured polyether-ether-ketone scaffold for repair of large bone defects [J]. Biomaterials Translational, 2022, 3(2): 142-151. |
| [2] | Hongtao Yang, Wenjiao Lin, Yufeng Zheng. Advances and perspective on the translational medicine of biodegradable metals [J]. Biomaterials Translational, 2021, 2(3): 177-187. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||