Biomaterials Translational ›› 2020, Vol. 1 ›› Issue (1): 18-32.doi: 10.3877/cma.j.issn.2096-112X.2020.01.003
• REVIEW • Previous Articles Next Articles
Received:2020-08-11
Revised:2020-08-21
Accepted:2020-08-21
Online:2020-12-28
Published:2020-12-28
Contact:
Yuan Lin
E-mail:linyuan@ciac.ac.cn
Guo, J.; Lin, Y. One-dimensional micro/nanomotors for biomedicine: delivery, sensing and surgery. Biomater Transl. 2020, 1(1), 18-32.
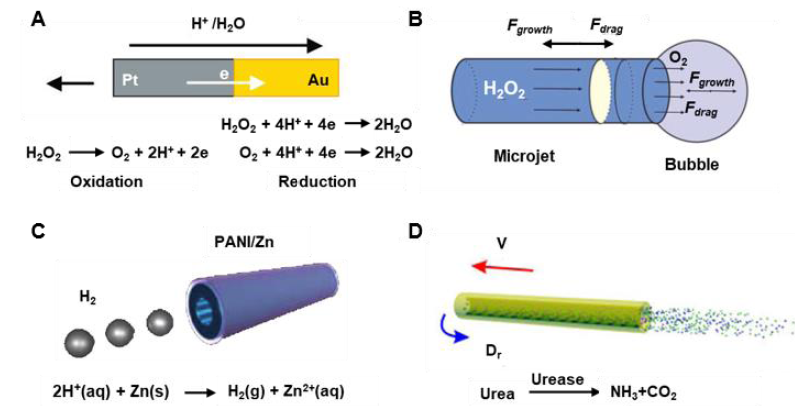
Figure 1. Schematic illustration of chemical propulsion mechanisms used in one-dimensional micro/nanomotors. (A) Scheme of Pt/Au nanowire motors propelled by a self-electrophoretic mechanism in H2O2 solution. Reprinted with permission from Wang et al.38 Copyright (2006) American Chemical Society. (B) Scheme of tubular micromotors propelled by ejecting O2 bubbles in H2O2 solution. Reprinted with permission from Manjare et al.45 Copyright (2013) American Chemical Society. (C) Scheme of PANI/Zn tubular microtube propelled by recoiling H2 bubbles in aqueous acid. Reproduced with permission from Gao et al.33 Copyright (2012) American Chemical Society. (D) SiO2/urease tubular nanomotor propelled by the diffusion flux of reaction products. Reprinted with permission from Ma et al.35 Copyright (2016) American Chemical Society. PANI: polyaniline.
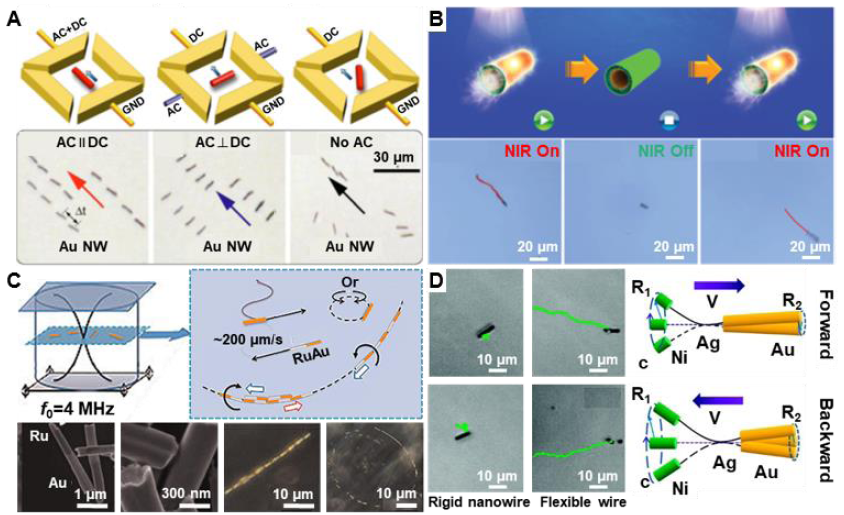
Figure 2. Representative examples of external field propulsion mechanisms of one-dimensional micro/nanomotors. (A) Au nanowire (Au NW) propelled and aligned by AC and DC electric fields in patterned microelectrodes. Reprinted with permission from Fan et al.53 Copyright (2008) American Institute of Physics. (B) Multilayered tubular micromotors decorated with gold nanoparticles propelled by NIR light. Reprinted with permission from Wu et al.59 Copyright (2016) John Wiley and Sons. (C) Au/Ru bimetallic nanowire with concave and convex ends propelled by ultrasound as well as its trajectories of axial directional motion and in-plane rotation. Reproduced with permission from Wang et al.60 Copyright (2012) American Chemical Society. (D) Ni/Ag/Au flexible nanowire propelled by rotational magnetic field. Reproduced with permission from Gao et al.62 Copyright (2010) American Chemical Society. AC: alternating current; DC: direct current; f: frequency; GND: Ground; NIR: near infrared; R1: Ni segment; R2: Au segment; V: magnetic swimming direction.
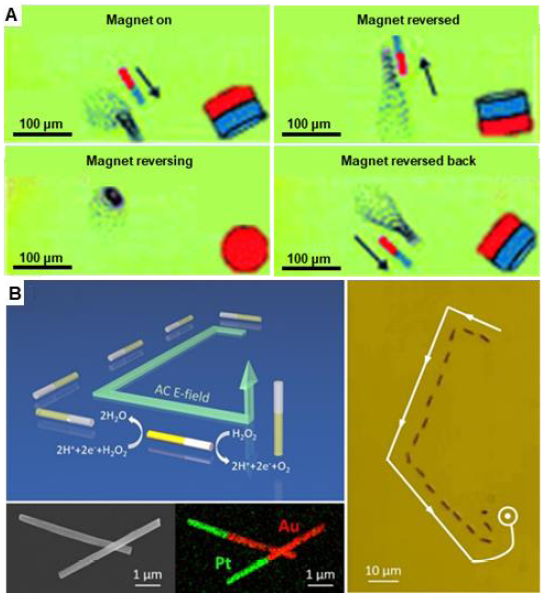
Figure 3. Motion control of one-dimensional micro/nanomotors via external fields. (A) Directional motion and reversing of tubular micromotors remotely guided by a magnetic field. Reproduced with permission from Zhao et al.64 Copyright (2012) American Chemical Society. (B) Two dimensions and three dimensions movement of Pt/Au bimetallic nanowire motors manipulated by three-dimensional orthogonal microelectrodes. Reprinted with permission from Guo et al.66 Copyright (2018) American Chemical Society. AC: alternating current; hv: light radiation.Therefore, the trajectory of a TiO2/Si microwire could be precisely programmed by adjusting the intensity and propagation direction of the light.
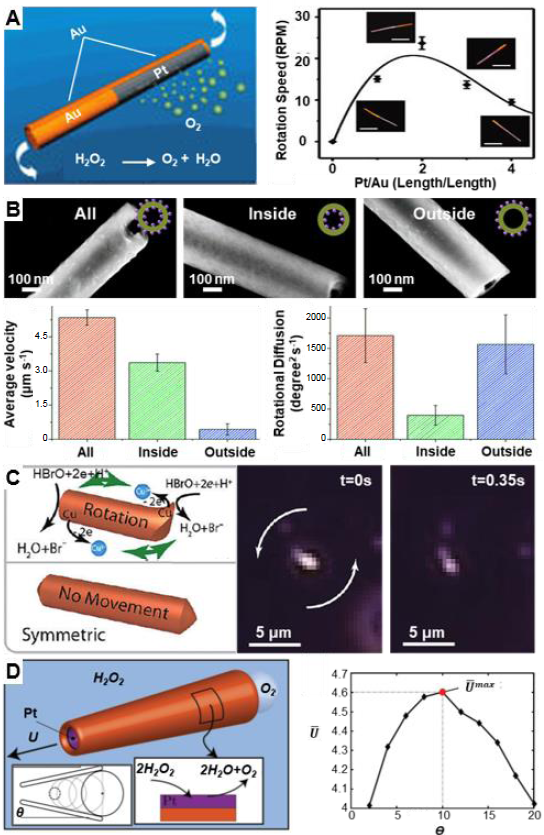
Figure 4. Architectural effects on the motive behaviour of one-dimensional micro/nanomotors. (A) The rotation speed could be adjusted by the distribution ratio of Pt/Au on the Au/Pt/Au nanorods coated with a Au/Cr layer on one face. Reproduced with permission from Qian et al.67 Copyright (2007) American Chemical Society. (B) Enzymes on inner or exterior surface of nanotubes enhanced the longitudinal translational and rotational diffusions, respectively. Reproduced with permission from Ma et al.35 Copyright (2016) American Chemical Society. (C) A copper nanowire with a rachet on one end was able to rotate whereas a symmetric nanowire was not. Reproduced with permission from Liu and Sen.41 Copyright (2011) American Chemical Society. (D) Tuning the velocity of tubular micromotors by adjusting the semi-cone angle. Reproduced with permission from Gallino et al.69 Copyright (2018) John Wiley and Sons.
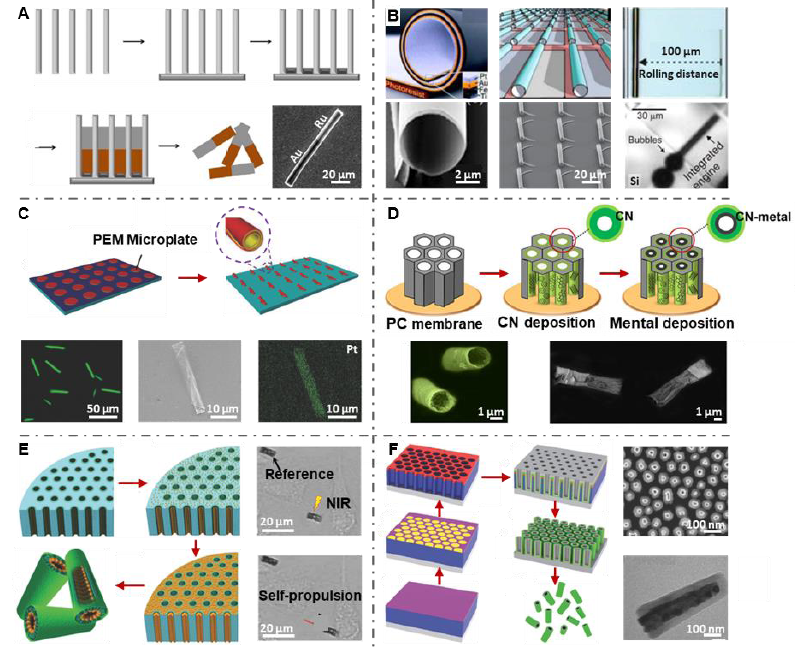
Figure 5. Fabrication of one-dimensional micro/nanomotors. (A) Anodic alumina oxide membrane template-assisted electrochemical deposition for the preparation of metallic nanowires. Reproduced with permission from Wang and Pumera22 and Wang et al.38 Copyright (2015) & (2006) American Chemical Society. (B) Rolled-up technique for the preparation of tubular micromotors. Reproduced with permission from Mei et al.43 and Soloveo et al.44 Copyright (2008) & (2009) & (2010) John Wiley and Sons. (C) Combination of layer-by-layer assembly, microcontact printing and rolled-up technique for the fabrication of tubular micromotors. Reproduced with permission from Hu et al.74 Copyright (2018) John Wiley and Sons. (D) Electroplating with carbon materials for the preparation of tubular micromotors. Reproduced with permission Maria-Hormigos et al.78 Copyright (2016) American Chemical Society. (E) Bottom-up assembly for tubular nanomotors. Reproduced with permission from Peng et al.79 Copyright (2018) Royal Society of Chemistry. (F) Combination of block polymer lithography and atomic layer deposition for the fabrication of tubular nanomotors. Reproduced with permission from Li et al.30 Copyright (2017) John Wiley and Sons. CN: carbon nanotube; NIR: near infrared; PC: polycarbonate; PEM: polyelectrolyte multilayer.
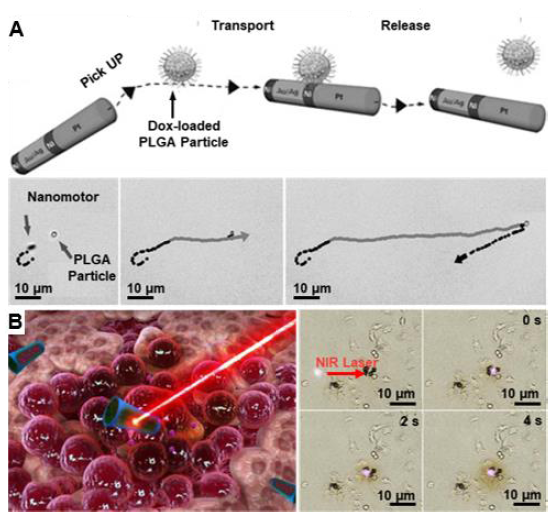
Figure 6. Organic small molecule drug delivery using one-dimensional micro/nanomotors. (A) Ni/(Au50/Ag50 alloy)/Ni/Pt nanowire motors enabled the pick up, transport and release of Dox-loaded PLGA particles at predetermined positions. Reproduced with permission from Kagan et al.84 Copyright (2010) John Wiley and Sons. (B) Tubular micromotors encapsulating Dox could move to targeted cancer cells under magnetic guidance and subsequently be triggered to release Dox by a near infrared laser. Reproduced with permission from Wu et al.47 Copyright (2015) American Chemical Society. PLGA: poly(lactic-co-glycolic acid).
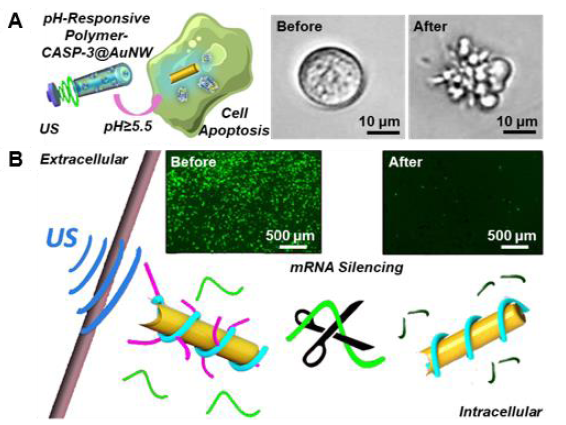
Figure 7. Rapid intracellular delivery of proteins (A) and RNAs (B) with ultrasound-propelled gold nanowire (AuNW) motors. Reproduced with permission from Esteban-Fernández de ávila et al.88 and Díez et al.90 Copyright (2017) & (2016) American Chemical Society. CASP-3: caspase-3; US: ultrasound.
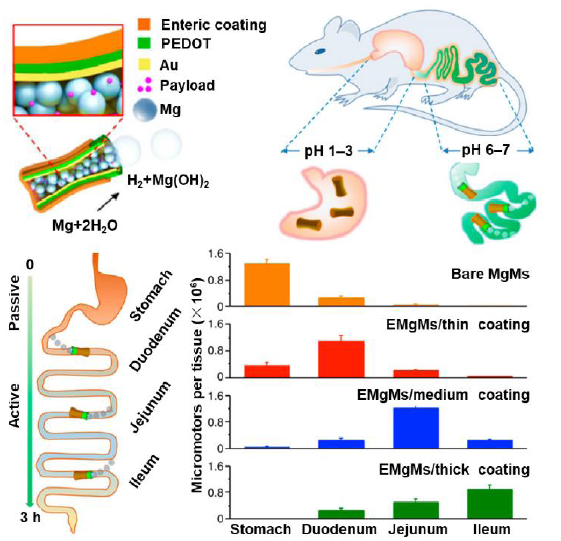
Figure 8. Tuning distribution of drugs in gastrointestinal organs by varying the thickness of a pH-responsive polymer coating on a Mg-based tubular micromotor. Reproduced with permission from Li et al.93 Copyright (2016) American Chemical Society. EMgMs: enteric Mg micromotors; PEDOT: poly(3,4-ethylenedioxythiophene).
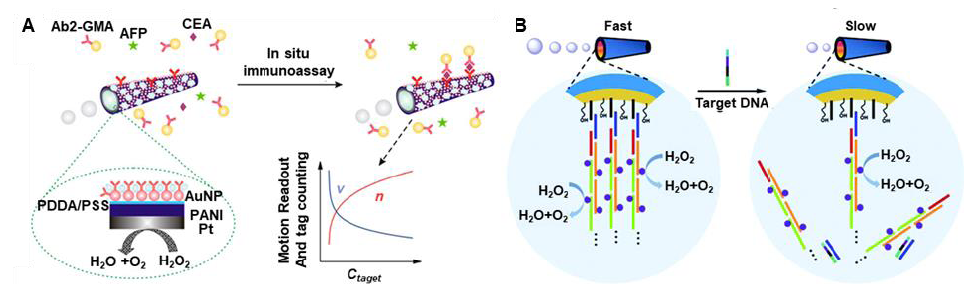
Figure 9. Sensing using one-dimensional micro/nanomotors. (A) Scheme of tubular micromotors tailored with antibody for cancer biomarker detection (e.g. CEA). Reproduced with permission from Yu et al.94 Copyright (2014) American Chemical Society. (B) Scheme of tubular micromotors for targeted DNA detection through the detachment of active materials (catalase). Reproduced with permission from Fu et al.98 Copyright (2017) Royal Society of Chemistry. Ab2-GMA: secondary-antibody-modified glycidyl methacrylate microspheres; AFP: alpha fetoprotein; CEA: carcinoembryonic antigen.
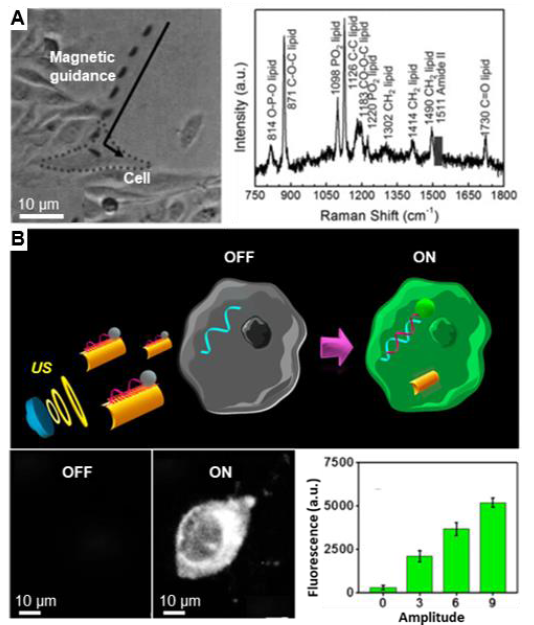
Figure 10. One-dimensional nanomotors for single cell sensing. (A) Ag/Ni/Ag three-segment nanorods coated with Ag nanoparticles used to analyse the chemicals of a single cell. Reproduced with permission from Xu et al.83 Copyright (2013) John Wiley and Sons. (B) Ultrasound-propelled nanorod modified with fluorescent dye-labelled ssDNA for miRNA detection of a single cell. Reproduced with permission from Esteban-Fernández de ávila et al.100 Copyright (2015) Royal Society of Chemistry. US: ultrasound.
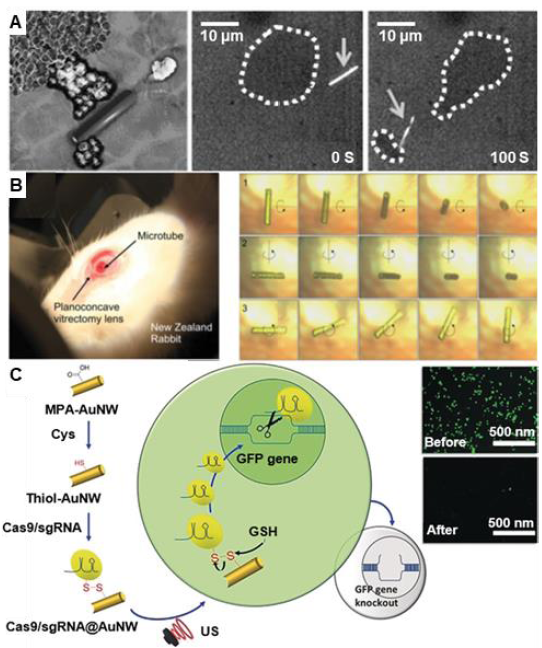
Figure 11. One-dimensional micro/nanomotors for surgery. (A) Tubular micromotors propelled by acoustic droplet vaporization for cellular tissue ablation. Reproduced with permission from Kagan et al.101 Copyright (2012) John Wiley and Sons. (B) Implantable magnetic tubular micromotors for wireless ophthalmologic surgery. Reproduced with permission from Chatzipirpiridis et al.102 Copyright (2014) John Wiley and Sons. (C) Ultrasound-propelled gold nanowires loaded with a Cas9/sgRNA complex for rapid and efficient gene knockout. Reproduced with permission from Hansen-Bruhn et al.104 Copyright (2018) John Wiley and Sons. AuNW: gold nanowire; Cys: cysteine; GFP: green fluorescent protein; GSH: glutathione; MPA: 3-mercaptopropionic acid; sgRNA: single guide RNA; US: ultrasound.
| 1. |
Giulianotti, P. C.; Coratti, A.; Angelini, M.; Sbrana, F.; Cecconi, S.; Balestracci, T.; Caravaglios, G. Robotics in general surgery: personal experience in a large community hospital. Arch Surg. 2003,138, 777-784.
doi: 10.1001/archsurg.138.7.777 URL pmid: 12860761 |
| 2. |
Li, J.; Esteban-Fernández de Ávila, B.; Gao, W.; Zhang, L.; Wang, J. Micro/nanorobots for biomedicine: delivery, surgery, sensing, and detoxification. Sci Robot. 2017,2, eaam6431.
doi: 10.1126/scirobotics.aao1891 URL pmid: 33157885 |
| 3. |
Nelson, B. J.; Kaliakatsos, I. K.; Abbott, J. J. Microrobots for minimally invasive medicine. Annu Rev Biomed Eng. 2010,12, 55-85.
doi: 10.1146/annurev-bioeng-010510-103409 URL pmid: 20415589 |
| 4. |
Abdelmohsen, L.; Peng, F.; Tu, Y.; Wilson, D. A. Micro- and nano-motors for biomedical applications. J Mater Chem B. 2014,2, 2395-2408.
doi: 10.1039/c3tb21451f URL pmid: 32261411 |
| 5. |
Peng, F.; Tu, Y.; Wilson, D. A. Micro/nanomotors towards in vivo application: cell, tissue and biofluid. Chem Soc Rev. 2017,46, 5289-5310.
doi: 10.1039/c6cs00885b URL pmid: 28524919 |
| 6. |
Kim, K.; Guo, J.; Liang, Z.; Fan, D. Artificial micro/nanomachines for bioapplications: biochemical delivery and diagnostic sensing. Adv Funct Mater. 2018,28, 1705867.
doi: 10.1002/adfm.v28.25 URL |
| 7. |
Dey, K. K.; Sen, A. Chemically propelled molecules and machines. J Am Chem Soc. 2017,139, 7666-7676.
doi: 10.1021/jacs.7b02347 URL pmid: 28493690 |
| 8. | Xu, T.; Gao, W.; Xu, L. P.; Zhang, X.; Wang, S. Fuel-free synthetic micro-/nanomachines. Adv Mater. 2017,29, 10. |
| 9. |
Guix, M.; Mayorga-Martinez, C. C.; Merkoçi, A. Nano/micromotors in (bio)chemical science applications. Chem Rev. 2014,114, 6285-6322.
doi: 10.1021/cr400273r URL pmid: 24827167 |
| 10. |
Duan, W.; Wang, W.; Das, S.; Yadav, V.; Mallouk, T. E.; Sen, A. Synthetic nano- and micromachines in analytical chemistry: sensing, migration, capture, delivery, and separation. Annu Rev Anal Chem (Palo Alto Calif). 2015,8, 311-333.
doi: 10.1146/annurev-anchem-071114-040125 URL |
| 11. |
Ceylan, H.; Giltinan, J.; Kozielski, K.; Sitti, M. Mobile microrobots for bioengineering applications. Lab ChiP. 2017,17, 1705-1724.
doi: 10.1039/c7lc00064b URL pmid: 28480466 |
| 12. |
Xu, B.; Zhang, B.; Wang, L.; Huang, G.; Mei, Y. Tubular micro/nanomachines: from the basics to recent advances. Adv Funct Mater. 2018,28, 1705872.
doi: 10.1002/adfm.v28.25 URL |
| 13. |
Li, L.; Liang, L.; Wu, H.; Zhu, X. One-dimensional perovskite manganite oxide nanostructures: recent developments in synthesis, characterization, transport properties, and applications. Nanoscale Res Lett. 2016,11, 121.
doi: 10.1186/s11671-016-1320-1 URL pmid: 26932760 |
| 14. |
Chen, J.; Wiley, B. J.; Xia, Y. One-dimensional nanostructures of metals: large-scale synthesis and some potential applications. Langmuir. 2007,23, 4120-4129.
doi: 10.1021/la063193y URL pmid: 17249708 |
| 15. |
Donald, I. W. Production, properties and applications of microwire and related products. J Mater Sci. 1987,22, 2661-2679.
doi: 10.1007/BF01086455 URL |
| 16. |
Liang, H. W.; Liu, J. W.; Qian, H. S.; Yu, S. H. Multiplex templating process in one-dimensional nanoscale: controllable synthesis, macroscopic assemblies, and applications. Acc Chem Res. 2013,46, 1450-1461.
doi: 10.1021/ar300272m URL pmid: 23441891 |
| 17. |
Zan, X.; Feng, S.; Balizan, E.; Lin, Y.; Wang, Q. Facile method for large scale alignment of one dimensional nanoparticles and control over myoblast orientation and differentiation. ACS Nano. 2013,7, 8385-8396.
doi: 10.1021/nn403908k URL pmid: 24004197 |
| 18. |
Feng, S.; Lu, L.; Zan, X.; Wu, Y.; Lin, Y.; Wang, Q. Genetically engineered plant viral nanoparticles direct neural cells differentiation and orientation. Langmuir. 2015,31, 9402-9409.
doi: 10.1021/acs.langmuir.5b02226 URL pmid: 26247572 |
| 19. |
Chen, L.; Zhao, X.; Lin, Y.; Su, Z.; Wang, Q. Dual stimuli-responsive supramolecular hydrogel of bionanoparticles and hyaluronan. Polym Chem. 2014,5, 6754-6760.
doi: 10.1039/C4PY00819G URL |
| 20. |
Guo, J.; Zhao, X.; Hu, J.; Lin, Y.; Wang, Q. Tobacco mosaic virus with peroxidase-like activity for cancer cell detection through colorimetric assay. Mol Pharm. 2018,15, 2946-2953.
doi: 10.1021/acs.molpharmaceut.7b00921 URL pmid: 29300485 |
| 21. |
Tu, Y.; Peng, F.; Wilson, D. A. Motion manipulation of micro- and nanomotors. Adv Mater. 2017,29, 1701970.
doi: 10.1002/adma.v29.39 URL |
| 22. |
Wang, H.; Pumera, M. Fabrication of micro/nanoscale motors. Chem Rev. 2015,115, 8704-8735.
doi: 10.1021/acs.chemrev.5b00047 URL pmid: 26234432 |
| 23. |
Paxton, W. F.; Kistler, K. C.; Olmeda, C. C.; Sen, A.; St Angelo, S. K.; Cao, Y.; Mallouk, T. E.; Lammert, P. E.; Crespi, V. H. Catalytic nanomotors: autonomous movement of striped nanorods. J Am Chem Soc. 2004,126, 13424-13431.
doi: 10.1021/ja047697z URL pmid: 15479099 |
| 24. |
Sundararajan, S.; Lammert, P. E.; Zudans, A. W.; Crespi, V. H.; Sen, A. Catalytic motors for transport of colloidal cargo. Nano Lett. 2008,8, 1271-1276.
doi: 10.1021/nl072275j URL pmid: 18416540 |
| 25. |
Ma, X.; Hortelão, A. C.; Patiño, T.; Sánchez, S. Enzyme catalysis to power micro/nanomachines. ACS Nano. 2016,10, 9111-9122.
doi: 10.1021/acsnano.6b04108 URL pmid: 27666121 |
| 26. |
Kinnear, C.; Moore, T. L.; Rodriguez-Lorenzo, L.; Rothen-Rutishauser, B.; Petri-Fink, A. Form follows function: Nanoparticle shape and its implications for nanomedicine. Chem Rev. 2017,117, 11476-11521.
doi: 10.1021/acs.chemrev.7b00194 URL pmid: 28862437 |
| 27. |
Wen, A. M.; Steinmetz, N. F. Design of virus-based nanomaterials for medicine, biotechnology, and energy. Chem Soc Rev. 2016,45, 4074-4126.
doi: 10.1039/c5cs00287g URL pmid: 27152673 |
| 28. |
Baraban, L.; Harazim, S. M.; Sanchez, S.; Schmidt, O. G. Chemotactic behavior of catalytic motors in microfluidic channels. Angew Chem Int Ed Engl. 2013,52, 5552-5556.
doi: 10.1002/anie.201301460 URL pmid: 23616282 |
| 29. |
Sahari, A.; Headen, D.; Behkam, B. Effect of body shape on the motile behavior of bacteria-powered swimming microrobots (BacteriaBots). Biomed Microdevices. 2012,14, 999-1007.
doi: 10.1007/s10544-012-9712-1 URL pmid: 23053449 |
| 30. |
Li, J.; Liu, W.; Wang, J.; Rozen, I.; He, S.; Chen, C.; Kim, H. G.; Lee, H. J Lee, H. B. R.; Kwon, S. H.; Li, T.; Li, L.; Wang, J.; Mei, Y. Nanoconfined atomic layer deposition of TiO2/Pt nanotubes: toward ultrasmall highly efficient catalytic nanorockets. Adv Funct Mater. 2017,27, 1700598.
doi: 10.1002/adfm.v27.24 URL |
| 31. |
Li, J.; Rozen, I.; Wang, J. Rocket science at the nanoscale. ACS Nano. 2016,10, 5619-5634.
doi: 10.1021/acsnano.6b02518 URL pmid: 27219742 |
| 32. |
Sánchez, S.; Soler, L.; Katuri, J. Chemically powered micro- and nanomotors. Angew Chem Int Ed Engl. 2015,54, 1414-1444.
doi: 10.1002/anie.201406096 URL pmid: 25504117 |
| 33. |
Gao, W.; Uygun, A.; Wang, J. Hydrogen-bubble-propelled zinc-based microrockets in strongly acidic media. J Am Chem Soc. 2012,134, 897-900.
doi: 10.1021/ja210874s URL pmid: 22188367 |
| 34. |
Paxton, W. F.; Sen, A.; Mallouk, T. E. Motility of catalytic nanoparticles through self-generated forces. Chemistry. 2005,11, 6462-6470.
doi: 10.1002/chem.200500167 URL pmid: 16052651 |
| 35. |
Ma, X.; Hortelao, A. C.; Miguel-López, A.; Sánchez, S. Bubble-free propulsion of ultrasmall tubular nanojets powered by biocatalytic reactions. J Am Chem Soc. 2016,138, 13782-13785.
URL pmid: 27718566 |
| 36. |
Dreyfus, R.; Baudry, J.; Roper, M. L.; Fermigier, M.; Stone, H. A.; Bibette, J. Microscopic artificial swimmers. Nature. 2005,437, 862-865.
doi: 10.1038/nature04090 URL pmid: 16208366 |
| 37. |
Mirkovic, T.; Zacharia, N. S.; Scholes, G. D.; Ozin, G. A. Nanolocomotion - catalytic nanomotors and nanorotors. Small. 2010,6, 159-167.
doi: 10.1002/smll.200901340 URL pmid: 19911393 |
| 38. |
Wang, Y.; Hernandez, R. M.; Bartlett, D. J. Jr.; Bingham, J. M.; Kline, T. R.; Sen, A.; Mallouk, T. E. Bipolar electrochemical mechanism for the propulsion of catalytic nanomotors in hydrogen peroxide solutions. Langmuir. 2006,22, 10451-10456.
doi: 10.1021/la0615950 URL pmid: 17129015 |
| 39. | Fournier-Bidoz, S.; Arsenault, A. C.; Manners, I.; Ozin, G. A. Synthetic self-propelled nanorotors. Chem Commun (Camb). 2005, 441-443. |
| 40. |
Demirok, U. K.; Laocharoensuk, R.; Manesh, K. M.; Wang, J. Ultrafast catalytic alloy nanomotors. Angew Chem Int Ed Engl. 2008,47, 9349-9351.
doi: 10.1002/anie.200803841 URL pmid: 18972475 |
| 41. |
Liu, R.; Sen, A. Autonomous nanomotor based on copper-platinum segmented nanobattery. J Am Chem Soc. 2011,133, 20064-20067.
doi: 10.1021/ja2082735 URL pmid: 21961523 |
| 42. |
Pumera, M. Electrochemically powered self-propelled electrophoretic nanosubmarines. Nanoscale. 2010,2, 1643-1649.
doi: 10.1039/c0nr00287a URL pmid: 20680201 |
| 43. |
Mei, Y.; Huang, G.; Solovev, A. A.; Ureña, E. B.; Mönch, I.; Ding, F.; Reindl, T.; Fu, R. K. Y.; Chu, P. K.; Schmidt, O. G. Versatile approach for integrative and functionalized tubes by strain engineering of nanomembranes on polymers. Adv Mater. 2008,20, 4085-4090.
doi: 10.1002/adma.v20:21 URL |
| 44. |
Solovev, A. A.; Mei, Y.; Bermúdez Ureña, E.; Huang, G.; Schmidt, O. G. Catalytic microtubular jet engines self-propelled by accumulated gas bubbles. Small. 2009,5, 1688-1692.
doi: 10.1002/smll.200900021 URL pmid: 19373828 |
| 45. |
Manjare, M.; Yang, B.; Zhao, Y. P. Bubble-propelled microjets: model and experiment. J Phys Chem C. 2013,117, 4657-4665.
doi: 10.1021/jp311977d URL |
| 46. |
Luo, M.; Feng, Y.; Wang, T.; Guan, J. Micro-/nanorobots at work in active drug delivery. Adv Funct Mater. 2018,28, 1706100.
doi: 10.1002/adfm.v28.25 URL |
| 47. |
Wu, Z.; Lin, X.; Zou, X.; Sun, J.; He, Q. Biodegradable protein-based rockets for drug transportation and light-triggered release. ACS Appl Mater Interfaces. 2015,7, 250-255.
doi: 10.1021/am507680u URL pmid: 25496011 |
| 48. |
Simmchen, J.; Magdanz, V.; Sanchez, S.; Chokmaviroj, S.; Ruiz-Molina, D.; Baeza, A.; Schmidt, O. G. Effect of surfactants on the performance of tubular and spherical micromotors - a comparative study. RSC Adv. 2014,4, 20334-20340.
doi: 10.1039/C4RA02202E URL pmid: 25364501 |
| 49. |
Kagan, D.; Calvo-Marzal, P.; Balasubramanian, S.; Sattayasamitsathit, S.; Manesh, K. M.; Flechsig, G. U.; Wang, J. Chemical sensing based on catalytic nanomotors: motion-based detection of trace silver. J Am Chem Soc. 2009,131, 12082-12083.
doi: 10.1021/ja905142q URL pmid: 19670862 |
| 50. |
Li, J.; Liu, W.; Wu, X.; Gao, X. Mechanism of pH-switchable peroxidase and catalase-like activities of gold, silver, platinum and palladium. Biomaterials. 2015,48, 37-44.
doi: 10.1016/j.biomaterials.2015.01.012 URL pmid: 25701030 |
| 51. |
Wu, Z.; Wu, Y.; He, W.; Lin, X.; Sun, J.; He, Q. Self-propelled polymer-based multilayer nanorockets for transportation and drug release. Angew Chem Int Ed Engl. 2013,52, 7000-7003.
doi: 10.1002/anie.201301643 URL pmid: 23703837 |
| 52. |
Hong, Y.; Blackman, N. M.; Kopp, N. D.; Sen, A.; Velegol, D. Chemotaxis of nonbiological colloidal rods. Phys Rev Lett. 2007,99, 178103.
doi: 10.1103/PhysRevLett.99.178103 URL pmid: 17995374 |
| 53. |
Fan, D. L.; Cammarata, R. C.; Chien, C. L. Precision transport and assembling of nanowires in suspension by electric fields. Appl Phys Lett. 2008,92, 093115.
doi: 10.1063/1.2891091 URL |
| 54. |
Kim, K.; Xu, X.; Guo, J.; Fan, D. L. Ultrahigh-speed rotating nanoelectromechanical system devices assembled from nanoscale building blocks. Nat Commun. 2014,5, 3632.
doi: 10.1038/ncomms4632 URL pmid: 24709694 |
| 55. |
Xu, L.; Mou, F.; Gong, H.; Luo, M.; Guan, J. Light-driven micro/nanomotors: from fundamentals to applications. Chem Soc Rev. 2017,46, 6905-6926.
doi: 10.1039/c7cs00516d URL pmid: 28949354 |
| 56. |
Gorostiza, P.; Isacoff, E. Y. Optical switches for remote and noninvasive control of cell signaling. Science. 2008,322, 395-399.
doi: 10.1126/science.1166022 URL pmid: 18927384 |
| 57. |
Giudicatti, S.; Marz, S. M.; Soler, L.; Madani, A.; Jorgensen, M. R.; Sanchez, S.; Schmidt, O. G. Photoactive rolled-up TiO(2) microtubes: fabrication, characterization and applications. J Mater Chem C Mater. 2014,2, 5892-5901.
doi: 10.1039/c4tc00796d URL pmid: 25580249 |
| 58. |
Mou, F.; Li, Y.; Chen, C.; Li, W.; Yin, Y.; Ma, H.; Guan, J. Single-component TiO2 tubular microengines with motion controlled by light-induced bubbles. Small. 2015,11, 2564-2570.
doi: 10.1002/smll.201403372 URL pmid: 25627213 |
| 59. |
Wu, Z.; Si, T.; Gao, W.; Lin, X.; Wang, J.; He, Q. Superfast near-infrared light-driven polymer multilayer rockets. Small. 2016,12, 577-582.
doi: 10.1002/smll.201502605 URL pmid: 26690728 |
| 60. |
Wang, W.; Castro, L. A.; Hoyos, M.; Mallouk, T. E. Autonomous motion of metallic microrods propelled by ultrasound. ACS Nano. 2012,6, 6122-6132.
doi: 10.1021/nn301312z URL |
| 61. |
Pak, O. S.; Gao, W.; Wang, J.; Lauga, E. High-speed propulsion of flexible nanowire motors: Theory and experiments. Soft Matter. 2011,7, 8169-8181.
doi: 10.1039/c1sm05503h URL |
| 62. |
Gao, W.; Sattayasamitsathit, S.; Manesh, K. M.; Weihs, D.; Wang, J. Magnetically powered flexible metal nanowire motors. J Am Chem Soc. 2010,132, 14403-14405.
doi: 10.1021/ja1072349 URL pmid: 20879711 |
| 63. |
Wu, Z.; Lin, X.; Wu, Y.; Si, T.; Sun, J.; He, Q. Near-infrared light-triggered “on/off” motion of polymer multilayer rockets. ACS Nano. 2014,8, 6097-6105.
doi: 10.1021/nn501407r URL pmid: 24806430 |
| 64. |
Zhao, G.; Sanchez, S.; Schmidt, O. G.; Pumera, M. Micromotors with built-in compasses. Chem Commun (Camb). 2012,48, 10090-10092.
doi: 10.1039/c2cc35671f URL |
| 65. |
Dai, B.; Wang, J.; Xiong, Z.; Zhan, X.; Dai, W.; Li, C. C.; Feng, S. P.; Tang, J. Programmable artificial phototactic microswimmer. Nat Nanotechnol. 2016,11, 1087-1092.
doi: 10.1038/nnano.2016.187 URL pmid: 27749832 |
| 66. |
Guo, J.; Gallegos, J. J.; Tom, A. R.; Fan, D. Electric-field-guided precision manipulation of catalytic nanomotors for cargo delivery and powering nanoelectromechanical devices. ACS Nano. 2018,12, 1179-1187.
URL pmid: 29303550 |
| 67. |
Qin, L.; Banholzer, M. J.; Xu, X.; Huang, L.; Mirkin, C. A. Rational design and synjournal of catalytically driven nanorotors. J Am Chem Soc. 2007,129, 14870-14871.
doi: 10.1021/ja0772391 URL pmid: 17988136 |
| 68. |
Dhar, P.; Fischer, T. M.; Wang, Y.; Mallouk, T. E.; Paxton, W. F.; Sen, A. Autonomously moving nanorods at a viscous interface. Nano Lett. 2006,6, 66-72.
doi: 10.1021/nl052027s URL pmid: 16402789 |
| 69. |
Gallino, G.; Gallaire, F.; Lauga, E.; Michelin, S. Physics of bubble-propelled microrockets. Adv Funct Mater. 2018,28, 1800686.
doi: 10.1002/adfm.v28.25 URL |
| 70. |
Klingner, A.; Khalil, I. S. M.; Magdanz, V.; Fomin, V. M.; Schmidt, O. G.; Misra, S. Modeling of unidirectional-overloaded transition in catalytic tubular microjets. J Phys Chem C. 2017,121, 14854-14863.
doi: 10.1021/acs.jpcc.7b02447 URL |
| 71. |
Kline, T. R.; Paxton, W. F.; Mallouk, T. E.; Sen, A. Catalytic nanomotors: remote-controlled autonomous movement of striped metallic nanorods. Angew Chem Int Ed Engl. 2005,44, 744-746.
doi: 10.1002/anie.200461890 URL pmid: 15612073 |
| 72. |
Yao, K.; Manjare, M.; Barrett, C. A.; Yang, B.; Salguero, T. T.; Zhao, Y. Nanostructured scrolls from graphene oxide for microjet engines. J Phys Chem Lett. 2012,3, 2204-2208.
URL pmid: 26295771 |
| 73. |
Zhao, G.; Ambrosi, A.; Pumera, M. Clean room-free rapid fabrication of roll-up self-powered catalytic microengines. J Mater Chem A. 2014,2, 1219-1223.
doi: 10.1039/C3TA14318J URL |
| 74. |
Hu, N.; Sun, M.; Lin, X.; Gao, C.; Zhang, B.; Zheng, C.; Xie, H.; He, Q. Self-propelled rolled-up polyelectrolyte multilayer microrockets. Adv Funct Mater. 2018,28, 1705684.
doi: 10.1002/adfm.v28.25 URL |
| 75. |
Manesh, K. M.; Cardona, M.; Yuan, R.; Clark, M.; Kagan, D.; Balasubramanian, S.; Wang, J. Template-assisted fabrication of salt-independent catalytic tubular microengines. ACS Nano. 2010,4, 1799-1804.
doi: 10.1021/nn1000468 URL pmid: 20230041 |
| 76. |
Gao, W.; Sattayasamitsathit, S.; Orozco, J.; Wang, J. Highly efficient catalytic microengines: template electrosynjournal of polyaniline/platinum microtubes. J Am Chem Soc. 2011,133, 11862-11864.
doi: 10.1021/ja203773g URL pmid: 21749138 |
| 77. |
Gao, W.; Sattayasamitsathit, S.; Uygun, A.; Pei, A.; Ponedal, A.; Wang, J. Polymer-based tubular microbots: role of composition and preparation. Nanoscale. 2012,4, 2447-2453.
doi: 10.1039/c2nr30138e URL pmid: 22374514 |
| 78. |
Maria-Hormigos, R.; Jurado-Sanchez, B.; Vazquez, L.; Escarpa, A. Carbon allotrope nanomaterials based catalytic micromotors. Chem Mater. 2016,28, 8962-8970.
doi: 10.1021/acs.chemmater.6b03689 URL |
| 79. |
Peng, F.; Men, Y.; Tu, Y.; Chen, Y.; Wilson, D. A. Nanomotor-based strategy for enhanced penetration across vasculature model. Adv Funct Mater. 2018,28, 1706117.
doi: 10.1002/adfm.v28.25 URL |
| 80. |
Dong, B.; Zhou, T.; Zhang, H.; Li, C. Y. Directed self-assembly of nanoparticles for nanomotors. ACS Nano. 2013,7, 5192-5198.
doi: 10.1021/nn400925q URL pmid: 23647410 |
| 81. |
Peng, F.; Tu, Y.; Adhikari, A.; Hintzen, J. C.; Löwik, D. W.; Wilson, D. A. A peptide functionalized nanomotor as an efficient cell penetrating tool. Chem Commun (Camb). 2017,53, 1088-1091.
doi: 10.1039/C6CC09169E URL |
| 82. |
Toebes, B. J.; Abdelmohsen, L. K. E. A.; Wilson, D. A. Enzyme-driven biodegradable nanomotor based on tubular-shaped polymeric vesicles. Polym Chem. 2018,9, 3190-3194.
doi: 10.1039/C8PY00559A URL |
| 83. |
Xu, X.; Li, H.; Hasan, D.; Ruoff, R. S.; Wang, A. X.; Fan, D. L. Near-field enhanced plasmonic-magnetic bifunctional nanotubes for single cell bioanalysis. Adv Funct Mater. 2013,23, 4332-4338.
doi: 10.1002/adfm.v23.35 URL |
| 84. |
Kagan, D.; Laocharoensuk, R.; Zimmerman, M.; Clawson, C.; Balasubramanian, S.; Kang, D.; Bishop, D.; Sattayasamitsathit, S.; Zhang, L.; Wang, J. Rapid delivery of drug carriers propelled and navigated by catalytic nanoshuttles. Small. 2010,6, 2741-2747.
doi: 10.1002/smll.201001257 URL pmid: 20979242 |
| 85. |
Leader, B.; Baca, Q. J.; Golan, D. E. Protein therapeutics: a summary and pharmacological classification. Nat Rev Drug Discov. 2008,7, 21-39.
doi: 10.1038/nrd2399 URL pmid: 18097458 |
| 86. |
Tang, R.; Kim, C. S.; Solfiell, D. J.; Rana, S.; Mout, R.; Velázquez-Delgado, E. M.; Chompoosor, A.; Jeong, Y.; Yan, B.; Zhu, Z. J.; Kim, C.; Hardy, J. A.; Rotello, V. M. Direct delivery of functional proteins and enzymes to the cytosol using nanoparticle-stabilized nanocapsules. ACS Nano. 2013,7, 6667-6673.
doi: 10.1021/nn402753y URL pmid: 23815280 |
| 87. |
Scaletti, F.; Hardie, J.; Lee, Y. W.; Luther, D. C.; Ray, M.; Rotello, V. M. Protein delivery into cells using inorganic nanoparticle-protein supramolecular assemblies. Chem Soc Rev. 2018,47, 3421-3432.
doi: 10.1039/c8cs00008e URL pmid: 29537040 |
| 88. |
Esteban-Fernández de Ávila, B.; Ramírez-Herrera, D. E.; Campuzano, S.; Angsantikul, P.; Zhang, L.; Wang, J. Nanomotor-enabled pH-responsive intracellular delivery of caspase-3: toward rapid cell apoptosis. ACS Nano. 2017,11, 5367-5374.
doi: 10.1021/acsnano.7b01926 URL pmid: 28467853 |
| 89. |
Uygun, M.; Jurado-Sánchez, B.; Uygun, D. A.; Singh, V. V.; Zhang, L.; Wang, J. Ultrasound-propelled nanowire motors enhance asparaginase enzymatic activity against cancer cells. Nanoscale. 2017,9, 18423-18429.
doi: 10.1039/c7nr07396h URL pmid: 29148558 |
| 90. |
Díez, P.; Esteban-Fernández de Ávila, B.; Ramírez-Herrera, D. E.; Villalonga, R.; Wang, J. Biomedical nanomotors: efficient glucose-mediated insulin release. Nanoscale. 2017,9, 14307-14311.
doi: 10.1039/c7nr05535h URL pmid: 28930338 |
| 91. |
Esteban-Fernández de Ávila, B.; Angell, C.; Soto, F.; Lopez-Ramirez, M. A.; Báez, D. F.; Xie, S.; Wang, J.; Chen, Y. Acoustically propelled nanomotors for intracellular siRNA delivery. ACS Nano. 2016,10, 4997-5005.
doi: 10.1021/acsnano.6b01415 URL pmid: 27022755 |
| 92. |
Gao, W.; Dong, R.; Thamphiwatana, S.; Li, J.; Gao, W.; Zhang, L.; Wang, J. Artificial micromotors in the mouse’s stomach: a step toward in vivo use of synthetic motors. ACS Nano. 2015,9, 117-123.
doi: 10.1021/nn507097k URL pmid: 25549040 |
| 93. |
Li, J.; Thamphiwatana, S.; Liu, W.; Esteban-Fernández de Ávila, B.; Angsantikul, P.; Sandraz, E.; Wang, J.; Xu, T.; Soto, F.; Ramez, V.; Wang, X.; Gao, W.; Zhang, L.; Wang, J. Enteric micromotor can selectively position and spontaneously propel in the gastrointestinal tract. ACS Nano. 2016,10, 9536-9542.
doi: 10.1021/acsnano.6b04795 URL pmid: 27648483 |
| 94. |
Yu, X.; Li, Y.; Wu, J.; Ju, H. Motor-based autonomous microsensor for motion and counting immunoassay of cancer biomarker. Anal Chem. 2014,86, 4501-4507.
doi: 10.1021/ac500912c URL pmid: 24731140 |
| 95. |
Qin, W.; Peng, T.; Gao, Y.; Wang, F.; Hu, X.; Wang, K.; Shi, J.; Li, D.; Ren, J.; Fan, C. Catalysis-driven self-thermophoresis of janus plasmonic nanomotors. Angew Chem Int Ed Engl. 2017,56, 515-518.
doi: 10.1002/anie.201609121 URL pmid: 27921355 |
| 96. |
Bunea, A. I.; Pavel, I. A.; David, S.; Gáspár, S. Sensing based on the motion of enzyme-modified nanorods. Biosens Bioelectron. 2015,67, 42-48.
doi: 10.1016/j.bios.2014.05.062 URL pmid: 24953025 |
| 97. |
Van Nguyen, K.; Minteer, S. D. DNA-functionalized Pt nanoparticles as catalysts for chemically powered micromotors: toward signal-on motion-based DNA biosensor. Chem Commun (Camb). 2015,51, 4782-4784.
doi: 10.1039/C4CC10250A URL |
| 98. |
Fu, S.; Zhang, X.; Xie, Y.; Wu, J.; Ju, H. An efficient enzyme-powered micromotor device fabricated by cyclic alternate hybridization assembly for DNA detection. Nanoscale. 2017,9, 9026-9033.
doi: 10.1039/c7nr01168g URL pmid: 28639653 |
| 99. |
Xie, Y.; Fu, S.; Wu, J.; Lei, J.; Ju, H. Motor-based microprobe powered by bio-assembled catalase for motion detection of DNA. Biosens Bioelectron. 2017,87, 31-37.
doi: 10.1016/j.bios.2016.07.104 URL pmid: 27517735 |
| 100. |
Esteban-Fernández de Ávila, B.; Martín, A.; Soto, F.; Lopez-Ramirez, M. A.; Campuzano, S.; Vásquez-Machado, G. M.; Gao, W.; Zhang, L.; Wang, J. Single cell real-time miRNAs sensing based on nanomotors. ACS Nano. 2015,9, 6756-6764.
doi: 10.1021/acsnano.5b02807 URL pmid: 26035455 |
| 101. |
Kagan, D.; Benchimol, M. J.; Claussen, J. C.; Chuluun-Erdene, E.; Esener, S.; Wang, J. Acoustic droplet vaporization and propulsion of perfluorocarbon-loaded microbullets for targeted tissue penetration and deformation. Angew Chem Int Ed Engl. 2012,51, 7519-7522.
doi: 10.1002/anie.201201902 URL pmid: 22692791 |
| 102. |
Chatzipirpiridis, G.; Ergeneman, O.; Pokki, J.; Ullrich, F.; Fusco, S.; Ortega, J. A.; Sivaraman, K. M.; Nelson, B. J.; Pané, S. Electroforming of implantable tubular magnetic microrobots for wireless ophthalmologic applications. Adv Healthc Mater. 2015,4, 209-214.
doi: 10.1002/adhm.201400256 URL pmid: 24986087 |
| 103. |
Cai, D.; Mataraza, J. M.; Qin, Z. H.; Huang, Z.; Huang, J.; Chiles, T. C.; Carnahan, D.; Kempa, K.; Ren, Z. Highly efficient molecular delivery into mammalian cells using carbon nanotube spearing. Nat Methods. 2005,2, 449-454.
doi: 10.1038/nmeth761 URL pmid: 15908924 |
| 104. |
Hansen-Bruhn, M.; de Ávila, B. E.; Beltrán-Gastélum, M.; Zhao, J.; Ramírez-Herrera, D. E.; Angsantikul, P.; Vesterager Gothelf, K.; Zhang, L.; Wang, J. Active intracellular delivery of a Cas9/sgRNA complex using ultrasound-propelled nanomotors. Angew Chem Int Ed Engl. 2018,57, 2657-2661.
doi: 10.1002/anie.201713082 URL pmid: 29325201 |
| No related articles found! |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
