Biomaterials Translational ›› 2024, Vol. 5 ›› Issue (1): 33-45.doi: 10.12336/biomatertransl.2024.01.004
• REVIEWS • Previous Articles Next Articles
Haoyu Guo1,2,3, Mingke Guo4, Zhidao Xia5,*( ), Zengwu Shao6,*(
), Zengwu Shao6,*( )
)
Received:2023-11-30
Revised:2023-12-05
Accepted:2023-12-18
Online:2024-03-28
Published:2024-03-28
Contact:
Zengwu Shao, About author:Zhidao Xia, z.xia@swansea.ac.uk. Zengwu Shao, szwpro@163.com;
Figure 1. Schematic diagram of membrane-coated delivery system for targeted cancer therapy. The preparation, characterisation, and common membrane sources of membrane-coated nanoparticles in targeted cancer therapy. Created with BioRender.com. RBC: red blood cell.
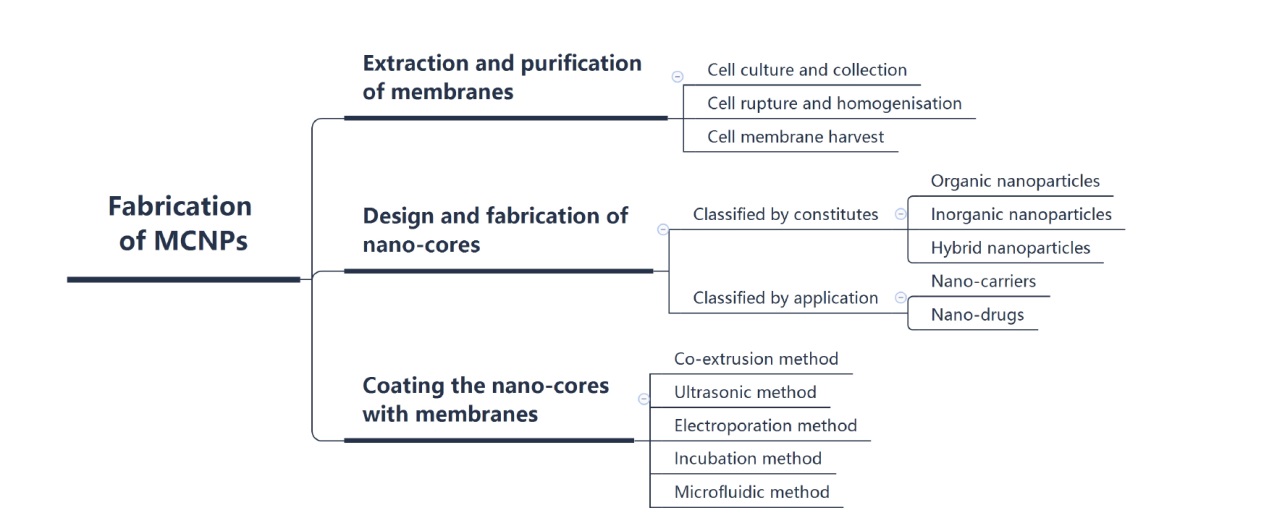
Figure 2. The fabrication of MCNPs. The fabrication of MCNPs contains three major process, extraction and purification of cell membranes, design and fabrication of nanocores, coating the nanocores with membranes. Created with Xmind Software. MCNP: membrane-coated nanoparticle.
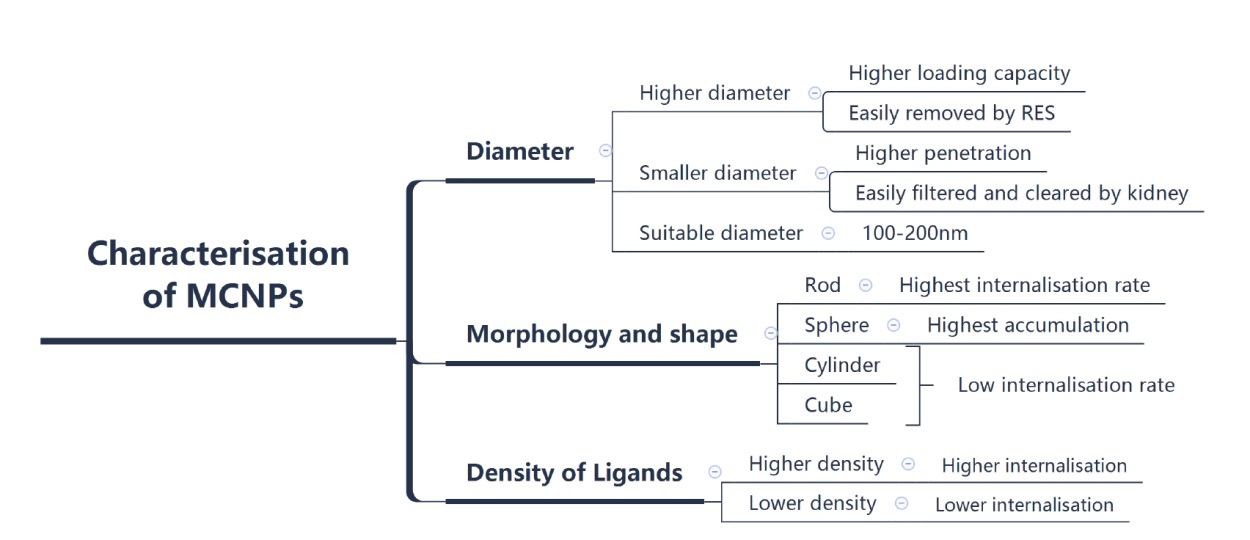
Figure 3. Characterisation of MCNPs. Characteristics such as diameter, morphology and shape of MCNPs, and the density of ligands on the surface of MCNPs have crucial impacts on the behaviour of MCNPs. Created with Xmind software. MCNP: membrane-coated nanoparticle.
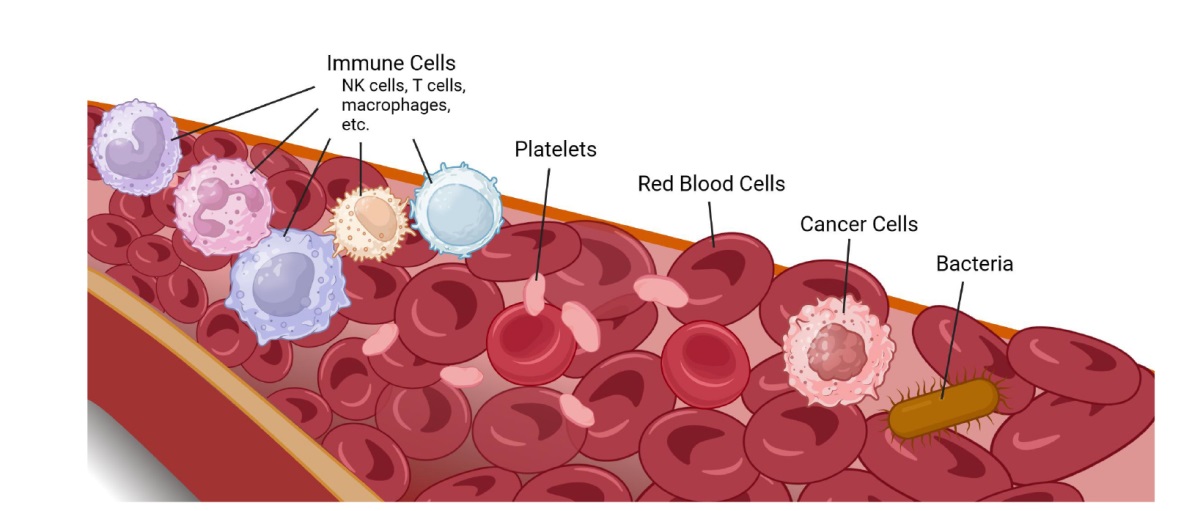
Figure 4. Common sources of membrane materials in MCNPs. The most common natural membrane sources of MCNPs are RBCs, platelets, cancer cells (and stem cells), immune cells and bacteria. Different sources of cell membrane have different physical or chemical properties. Created with BioRender.com. MCNP: membrane-coated nanoparticle; NK: natural killer; RBC: red blood cell.
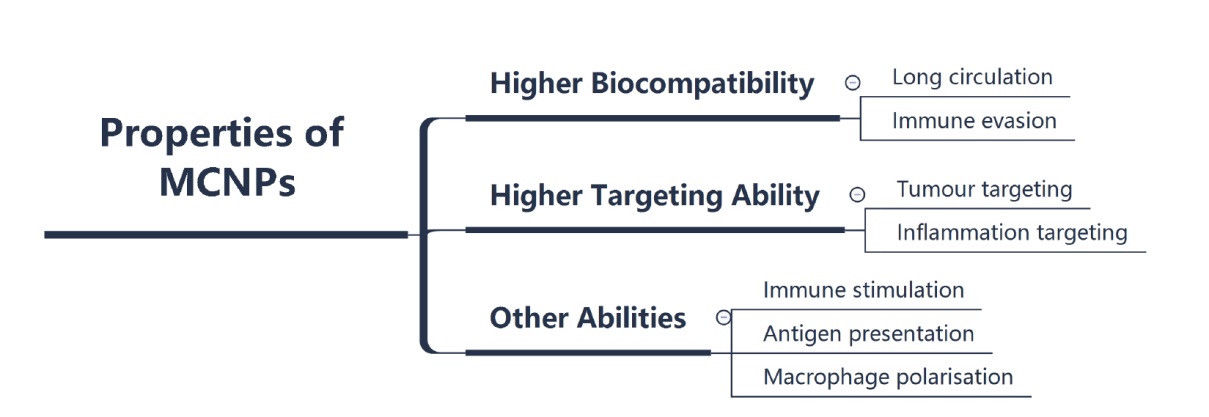
Figure 5. Properties of MCNPs. The membranes can endow the MCNPs with properties such as higher biocompatibility, higher targeting ability and some specific capabilities. Created with Xmind software. MCNP: membrane-coated nanoparticle.
| Membrane source | Immune evasion | Special property |
|---|---|---|
| Red blood cell | √ | Easy fabrication |
| Simple structure | ||
| Platelet | √ | Tumour targeting |
| Inflammation targeting | ||
| Immune cell | √ | Tumour targeting |
| Inflammation targeting | ||
| Immune stimulation | ||
| Antigen presentation | ||
| Macrophage polarisation | ||
| Tumour cell | √ | Homotypic tumour targeting |
| Immune stimulation | ||
| Tumour antigen presentation | ||
| Macrophage polarisation | ||
| Stem cell | √ | Tumour targeting |
| Inflammation targeting | ||
| Bacteria | × | Immune stimulation |
| Antigen presentation | ||
| Macrophage polarisation | ||
| Engineered/hybrid | - | Editable and combinable functions |
Table 1. Common membrane sources and their properties
| Membrane source | Immune evasion | Special property |
|---|---|---|
| Red blood cell | √ | Easy fabrication |
| Simple structure | ||
| Platelet | √ | Tumour targeting |
| Inflammation targeting | ||
| Immune cell | √ | Tumour targeting |
| Inflammation targeting | ||
| Immune stimulation | ||
| Antigen presentation | ||
| Macrophage polarisation | ||
| Tumour cell | √ | Homotypic tumour targeting |
| Immune stimulation | ||
| Tumour antigen presentation | ||
| Macrophage polarisation | ||
| Stem cell | √ | Tumour targeting |
| Inflammation targeting | ||
| Bacteria | × | Immune stimulation |
| Antigen presentation | ||
| Macrophage polarisation | ||
| Engineered/hybrid | - | Editable and combinable functions |
| 1. |
Sugahara, K. N.; Teesalu, T.; Karmali, P. P.; Kotamraju, V. R.; Agemy, L.; Greenwald, D. R.; Ruoslahti, E. Coadministration of a tumor-penetrating peptide enhances the efficacy of cancer drugs. Science. 2010, 328, 1031-1035.
doi: 10.1126/science.1183057 URL |
| 2. |
Wei, G.; Wang, Y.; Yang, G.; Wang, Y.; Ju, R. Recent progress in nanomedicine for enhanced cancer chemotherapy. Theranostics. 2021, 11, 6370-6392.
doi: 10.7150/thno.57828 URL |
| 3. |
Ward, R. A.; Fawell, S.; Floc’h, N.; Flemington, V.; McKerrecher, D.; Smith, P. D. Challenges and opportunities in cancer drug resistance. Chem Rev. 2021, 121, 3297-3351.
doi: 10.1021/acs.chemrev.0c00383 URL |
| 4. |
Wang, M.; Thanou, M. Targeting nanoparticles to cancer. Pharmacol Res. 2010, 62, 90-99.
doi: 10.1016/j.phrs.2010.03.005 URL |
| 5. |
Seyyednia, E.; Oroojalian, F.; Baradaran, B.; Mojarrad, J. S.; Mokhtarzadeh, A.; Valizadeh, H. Nanoparticles modified with vasculature-homing peptides for targeted cancer therapy and angiogenesis imaging. J Control Release. 2021, 338, 367-393.
doi: 10.1016/j.jconrel.2021.08.044 URL |
| 6. |
Tian, H.; Zhang, T.; Qin, S.; Huang, Z.; Zhou, L.; Shi, J.; Nice, E. C.; Xie, N.; Huang, C.; Shen, Z. Enhancing the therapeutic efficacy of nanoparticles for cancer treatment using versatile targeted strategies. J Hematol Oncol. 2022, 15, 132.
doi: 10.1186/s13045-022-01320-5 |
| 7. | Fang, R. H.; Kroll, A. V.; Gao, W.; Zhang, L. Cell membrane coating nanotechnology. Adv Mater. 2018, 30, e1706759. |
| 8. | Liu, Y.; Luo, J.; Chen, X.; Liu, W.; Chen, T. Cell membrane coating technology: a promising strategy for biomedical applications. Nanomicro Lett. 2019, 11, 100. |
| 9. |
Liu, L.; Pan, D.; Chen, S.; Martikainen, M. V.; Kårlund, A.; Ke, J.; Pulkkinen, H.; Ruhanen, H.; Roponen, M.; Käkelä, R.; Xu, W.; Wang, J.; Lehto, V. P. Systematic design of cell membrane coating to improve tumor targeting of nanoparticles. Nat Commun. 2022, 13, 6181.
doi: 10.1038/s41467-022-33889-3 |
| 10. |
Suk, J. S.; Xu, Q.; Kim, N.; Hanes, J.; Ensign, L. M. PEGylation as a strategy for improving nanoparticle-based drug and gene delivery. Adv Drug Deliv Rev. 2016, 99, 28-51.
doi: 10.1016/j.addr.2015.09.012 URL |
| 11. |
Estapé Senti, M.; de Jongh, C. A.; Dijkxhoorn, K.; Verhoef, J. J. F.; Szebeni, J.; Storm, G.; Hack, C. E.; Schiffelers, R. M.; Fens, M. H.; Boross, P. Anti-PEG antibodies compromise the integrity of PEGylated lipid-based nanoparticles via complement. J Control Release. 2022, 341, 475-486.
doi: 10.1016/j.jconrel.2021.11.042 URL |
| 12. |
Gautam, M.; Jozic, A.; Su, G. L.; Herrera-Barrera, M.; Curtis, A.; Arrizabalaga, S.; Tschetter, W.; Ryals, R. C.; Sahay, G. Lipid nanoparticles with PEG-variant surface modifications mediate genome editing in the mouse retina. Nat Commun. 2023, 14, 6468.
doi: 10.1038/s41467-023-42189-3 |
| 13. |
Hu, C. M.; Zhang, L.; Aryal, S.; Cheung, C.; Fang, R. H.; Zhang, L. Erythrocyte membrane-camouflaged polymeric nanoparticles as a biomimetic delivery platform. Proc Natl Acad Sci U S A. 2011, 108, 10980-10985.
doi: 10.1073/pnas.1106634108 URL |
| 14. |
Xia, Q.; Zhang, Y.; Li, Z.; Hou, X.; Feng, N. Red blood cell membrane-camouflaged nanoparticles: a novel drug delivery system for antitumor application. Acta Pharm Sin B. 2019, 9, 675-689.
doi: 10.1016/j.apsb.2019.01.011 URL |
| 15. |
Hu, C. M.; Fang, R. H.; Zhang, L. Erythrocyte-inspired delivery systems. Adv Healthc Mater. 2012, 1, 537-547.
doi: 10.1002/adhm.v1.5 URL |
| 16. |
Fang, R. H.; Gao, W.; Zhang, L. Targeting drugs to tumours using cell membrane-coated nanoparticles. Nat Rev Clin Oncol. 2023, 20, 33-48.
doi: 10.1038/s41571-022-00699-x |
| 17. |
Fang, R. H.; Jiang, Y.; Fang, J. C.; Zhang, L. Cell membrane-derived nanomaterials for biomedical applications. Biomaterials. 2017, 128, 69-83.
doi: 10.1016/j.biomaterials.2017.02.041 URL |
| 18. |
Wu, M.; Zhang, H.; Tie, C.; Yan, C.; Deng, Z.; Wan, Q.; Liu, X.; Yan, F.; Zheng, H. MR imaging tracking of inflammation-activatable engineered neutrophils for targeted therapy of surgically treated glioma. Nat Commun. 2018, 9, 4777.
doi: 10.1038/s41467-018-07250-6 |
| 19. |
Guo, H.; Wang, L.; Wu, W.; Guo, M.; Yang, L.; Zhang, Z.; Cao, L.; Pu, F.; Huang, X.; Shao, Z. Engineered biomimetic nanoreactor for synergistic photodynamic-chemotherapy against hypoxic tumor. J Control Release. 2022, 351, 151-163.
doi: 10.1016/j.jconrel.2022.09.020 URL |
| 20. |
Guo, H.; Zhang, W.; Wang, L.; Shao, Z.; Huang, X. Biomimetic cell membrane-coated glucose/oxygen-exhausting nanoreactor for remodeling tumor microenvironment in targeted hypoxic tumor therapy. Biomaterials. 2022, 290, 121821.
doi: 10.1016/j.biomaterials.2022.121821 URL |
| 21. |
Parodi, A.; Quattrocchi, N.; van de Ven, A. L.; Chiappini, C.; Evangelopoulos, M.; Martinez, J. O.; Brown, B. S.; Khaled, S. Z.; Yazdi, I. K.; Enzo, M. V.; Isenhart, L.; Ferrari, M.; Tasciotti, E. Synthetic nanoparticles functionalized with biomimetic leukocyte membranes possess cell-like functions. Nat Nanotechnol. 2013, 8, 61-68.
doi: 10.1038/nnano.2012.212 |
| 22. | Yu, J.; Wei, Z.; Li, Q.; Wan, F.; Chao, Z.; Zhang, X.; Lin, L.; Meng, H.; Tian, L. Advanced cancer starvation therapy by simultaneous deprivation of lactate and glucose using a MOF nanoplatform. Adv Sci (Weinh). 2021, 8, e2101467. |
| 23. | Yang, J.; Yang, Y. W. Metal-organic frameworks for biomedical applications. Small. 2020, 16, e1906846. |
| 24. |
Liu, W.; Yan, Q.; Xia, C.; Wang, X.; Kumar, A.; Wang, Y.; Liu, Y.; Pan, Y.; Liu, J. Recent advances in cell membrane coated metal-organic frameworks (MOFs) for tumor therapy. J Mater Chem B. 2021, 9, 4459-4474.
doi: 10.1039/D1TB00453K URL |
| 25. |
Pan, W. L.; Tan, Y.; Meng, W.; Huang, N. H.; Zhao, Y. B.; Yu, Z. Q.; Huang, Z.; Zhang, W. H.; Sun, B.; Chen, J. X. Microenvironment-driven sequential ferroptosis, photodynamic therapy, and chemotherapy for targeted breast cancer therapy by a cancer-cell-membrane-coated nanoscale metal-organic framework. Biomaterials. 2022, 283, 121449.
doi: 10.1016/j.biomaterials.2022.121449 URL |
| 26. | Zhen, X.; Cheng, P.; Pu, K. Recent advances in cell membrane-camouflaged nanoparticles for cancer phototherapy. Small. 2019, 15, e1804105. |
| 27. |
Cui, X.; Ruan, Q.; Zhuo, X.; Xia, X.; Hu, J.; Fu, R.; Li, Y.; Wang, J.; Xu, H. Photothermal nanomaterials: a powerful light-to-heat converter. Chem Rev. 2023, 123, 6891-6952.
doi: 10.1021/acs.chemrev.3c00159 URL |
| 28. |
Wu, F.; Liu, Y.; Cheng, H.; Meng, Y.; Shi, J.; Chen, Y.; Wu, Y. Enhanced cancer starvation therapy based on glucose oxidase/3-methyladenine-loaded dendritic mesoporous organosilicon nanoparticles. Biomolecules. 2021, 11, 1363.
doi: 10.3390/biom11091363 URL |
| 29. |
Ducrot, C.; Loiseau, S.; Wong, C.; Madec, E.; Volatron, J.; Piffoux, M. Hybrid extracellular vesicles for drug delivery. Cancer Lett. 2023, 558, 216107.
doi: 10.1016/j.canlet.2023.216107 URL |
| 30. |
Liu, C.; Zhang, W.; Li, Y.; Chang, J.; Tian, F.; Zhao, F.; Ma, Y.; Sun, J. Microfluidic sonication to assemble exosome membrane-coated nanoparticles for immune evasion-mediated targeting. Nano Lett. 2019, 19, 7836-7844.
doi: 10.1021/acs.nanolett.9b02841 URL |
| 31. | Xia, Y.; Rao, L.; Yao, H.; Wang, Z.; Ning, P.; Chen, X. Engineering macrophages for cancer immunotherapy and drug delivery. Adv Mater. 2020, 32, e2002054. |
| 32. |
Huang, X.; Guo, H.; Wang, L.; Zhang, Z.; Zhang, W. Biomimetic cell membrane-coated nanocarriers for targeted siRNA delivery in cancer therapy. Drug Discov Today. 2023, 28, 103514.
doi: 10.1016/j.drudis.2023.103514 URL |
| 33. |
Jarak, I.; Isabel Santos, A.; Helena Pinto, A.; Domingues, C.; Silva, I.; Melo, R.; Veiga, F.; Figueiras, A. Colorectal cancer cell exosome and cytoplasmic membrane for homotypic delivery of therapeutic molecules. Int J Pharm. 2023, 646, 123456.
doi: 10.1016/j.ijpharm.2023.123456 URL |
| 34. |
Albanese, A.; Tang, P. S.; Chan, W. C. The effect of nanoparticle size, shape, and surface chemistry on biological systems. Annu Rev Biomed Eng. 2012, 14, 1-16.
doi: 10.1146/annurev-bioeng-071811-150124 URL |
| 35. |
Salatin, S.; Maleki Dizaj, S.; Yari Khosroushahi, A. Effect of the surface modification, size, and shape on cellular uptake of nanoparticles. Cell Biol Int. 2015, 39, 881-890.
doi: 10.1002/cbin.v39.8 URL |
| 36. |
Agarwal, R.; Jurney, P.; Raythatha, M.; Singh, V.; Sreenivasan, S. V.; Shi, L.; Roy, K. Effect of shape, size, and aspect ratio on nanoparticle penetration and distribution inside solid tissues using 3D spheroid models. Adv Healthc Mater. 2015, 4, 2269-2280.
doi: 10.1002/adhm.v4.15 URL |
| 37. |
Lagarrigue, P.; Moncalvo, F.; Cellesi, F. Non-spherical polymeric nanocarriers for therapeutics: the effect of shape on biological systems and drug delivery properties. Pharmaceutics. 2022, 15, 32.
doi: 10.3390/pharmaceutics15010032 URL |
| 38. |
Jindal, A. B. The effect of particle shape on cellular interaction and drug delivery applications of micro- and nanoparticles. Int J Pharm. 2017, 532, 450-465.
doi: 10.1016/j.ijpharm.2017.09.028 URL |
| 39. | Li, X.; Montague, E. C.; Pollinzi, A.; Lofts, A.; Hoare, T. Design of smart size-, surface-, and shape-switching nanoparticles to improve therapeutic efficacy. Small. 2022, 18, e2104632. |
| 40. | Bilardo, R.; Traldi, F.; Vdovchenko, A.; Resmini, M. Influence of surface chemistry and morphology of nanoparticles on protein corona formation. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2022, 14, e1788. |
| 41. | He, F.; Zhu, L.; Zhou, X.; Zhang, P.; Cheng, J.; Qiao, Y.; Feng, Y.; Yue, S.; Xu, M.; Guan, J.; Li, X.; Ao, Z.; Qin, M.; Hou, Y.; Han, D. Red blood cell membrane-coated ultrasmall NaGdF(4) nanoprobes for high-resolution 3D magnetic resonance angiography. ACS Appl Mater Interfaces. 2022. doi: 10.1021/acsami.2c03530. |
| 42. |
Narain, A.; Asawa, S.; Chhabria, V.; Patil-Sen, Y. Cell membrane coated nanoparticles: next-generation therapeutics. Nanomedicine (Lond). 2017, 12, 2677-2692.
doi: 10.2217/nnm-2017-0225 URL |
| 43. |
Oldenborg, P. A.; Zheleznyak, A.; Fang, Y. F.; Lagenaur, C. F.; Gresham, H. D.; Lindberg, F. P. Role of CD47 as a marker of self on red blood cells. Science. 2000, 288, 2051-2054.
doi: 10.1126/science.288.5473.2051 URL |
| 44. |
Huang, S.; Song, C.; Miao, J.; Zhu, X.; Jia, Y.; Liu, Y.; Fu, D.; Li, B.; Miao, M.; Duan, S.; Zhang, Z.; Hu, Y. Red blood cell membrane-coated functionalized Au nanocage as a biomimetic platform for improved microRNA delivery in hepatocellular carcinoma. Int J Pharm. 2023, 642, 123044.
doi: 10.1016/j.ijpharm.2023.123044 URL |
| 45. |
Telen, M. J.; Rosse, W. F. Phosphatidylinositol-glycan linked proteins of the erythrocyte membrane. Baillieres Clin Haematol. 1991, 4, 849-868.
doi: 10.1016/S0950-3536(06)80033-8 URL |
| 46. |
Tomlinson, S.; Whitlow, M. B.; Nussenzweig, V. A synthetic peptide from complement protein C9 binds to CD59 and enhances lysis of human erythrocytes by C5b-9. J Immunol. 1994, 152, 1927-1934.
doi: 10.4049/jimmunol.152.4.1927 URL |
| 47. |
Zheng, B. D.; Xiao, M. T. Red blood cell membrane nanoparticles for tumor phototherapy. Colloids Surf B Biointerfaces. 2022, 220, 112895.
doi: 10.1016/j.colsurfb.2022.112895 URL |
| 48. |
Zhang, S. Q.; Fu, Q.; Zhang, Y. J.; Pan, J. X.; Zhang, L.; Zhang, Z. R.; Liu, Z. M. Surface loading of nanoparticles on engineered or natural erythrocytes for prolonged circulation time: strategies and applications. Acta Pharmacol Sin. 2021, 42, 1040-1054.
doi: 10.1038/s41401-020-00606-z |
| 49. |
Ren, X.; Zheng, R.; Fang, X.; Wang, X.; Zhang, X.; Yang, W.; Sha, X. Red blood cell membrane camouflaged magnetic nanoclusters for imaging-guided photothermal therapy. Biomaterials. 2016, 92, 13-24.
doi: 10.1016/j.biomaterials.2016.03.026 URL |
| 50. |
Rao, L.; Bu, L. L.; Xu, J. H.; Cai, B.; Yu, G. T.; Yu, X.; He, Z.; Huang, Q.; Li, A.; Guo, S. S.; Zhang, W. F.; Liu, W.; Sun, Z. J.; Wang, H.; Wang, T. H.; Zhao, X. Z. Red blood cell membrane as a biomimetic nanocoating for prolonged circulation time and reduced accelerated blood clearance. Small. 2015, 11, 6225-6236.
doi: 10.1002/smll.v11.46 URL |
| 51. |
Cox, D.; Kerrigan, S. W.; Watson, S. P. Platelets and the innate immune system: mechanisms of bacterial-induced platelet activation. J Thromb Haemost. 2011, 9, 1097-1107.
doi: 10.1111/j.1538-7836.2011.04264.x URL |
| 52. |
Schlesinger, M. Role of platelets and platelet receptors in cancer metastasis. J Hematol Oncol. 2018, 11, 125.
doi: 10.1186/s13045-018-0669-2 |
| 53. | Franco, A. T.; Corken, A.; Ware, J. Platelets at the interface of thrombosis, inflammation, and cancer. Blood. 2015, 126, 582-588. |
| 54. |
Sabrkhany, S.; Kuijpers, M. J. E.; Griffioen, A. W.; Oude Egbrink, M. G. A. Platelets: the holy grail in cancer blood biomarker research? Angiogenesis. 2019, 22, 1-2.
doi: 10.1007/s10456-018-9651-4 |
| 55. |
Nash, G. F.; Turner, L. F.; Scully, M. F.; Kakkar, A. K. Platelets and cancer. Lancet Oncol. 2002, 3, 425-430.
doi: 10.1016/S1470-2045(02)00789-1 URL |
| 56. | Geranpayehvaghei, M.; Dabirmanesh, B.; Khaledi, M.; Atabakhshi-Kashi, M.; Gao, C.; Taleb, M.; Zhang, Y.; Khajeh, K.; Nie, G. Cancer-associated-platelet-inspired nanomedicines for cancer therapy. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2021, 13, e1702. |
| 57. |
Hu, C. M.; Fang, R. H.; Wang, K. C.; Luk, B. T.; Thamphiwatana, S.; Dehaini, D.; Nguyen, P.; Angsantikul, P.; Wen, C. H.; Kroll, A. V.; Carpenter, C.; Ramesh, M.; Qu, V.; Patel, S. H.; Zhu, J.; Shi, W.; Hofman, F. M.; Chen, T. C.; Gao, W.; Zhang, K.; Chien, S.; Zhang, L. Nanoparticle biointerfacing by platelet membrane cloaking. Nature. 2015, 526, 118-121.
doi: 10.1038/nature15373 |
| 58. | Zhuang, J.; Gong, H.; Zhou, J.; Zhang, Q.; Gao, W.; Fang, R. H.; Zhang, L. Targeted gene silencing in vivo by platelet membrane-coated metal-organic framework nanoparticles. Sci Adv. 2020, 6, eaaz6108. |
| 59. |
Bahmani, B.; Gong, H.; Luk, B. T.; Haushalter, K. J.; DeTeresa, E.; Previti, M.; Zhou, J.; Gao, W.; Bui, J. D.; Zhang, L.; Fang, R. H.; Zhang, J. Intratumoral immunotherapy using platelet-cloaked nanoparticles enhances antitumor immunity in solid tumors. Nat Commun. 2021, 12, 1999.
doi: 10.1038/s41467-021-22311-z |
| 60. | Wang, D.; Wang, S.; Zhou, Z.; Bai, D.; Zhang, Q.; Ai, X.; Gao, W.; Zhang, L. White blood cell membrane-coated nanoparticles: recent development and medical applications. Adv Healthc Mater. 2022, 11, e2101349. |
| 61. | Rao, L.; Zhao, S. K.; Wen, C.; Tian, R.; Lin, L.; Cai, B.; Sun, Y.; Kang, F.; Yang, Z.; He, L.; Mu, J.; Meng, Q. F.; Yao, G.; Xie, N.; Chen, X. Activating macrophage-mediated cancer immunotherapy by genetically edited nanoparticles. Adv Mater. 2020, 32, e2004853. |
| 62. |
Franklin, R. A.; Liao, W.; Sarkar, A.; Kim, M. V.; Bivona, M. R.; Liu, K.; Pamer, E. G.; Li, M. O. The cellular and molecular origin of tumor-associated macrophages. Science. 2014, 344, 921-925.
doi: 10.1126/science.1252510 URL |
| 63. | Oroojalian, F.; Beygi, M.; Baradaran, B.; Mokhtarzadeh, A.; Shahbazi, M. A. Immune cell membrane-coated biomimetic nanoparticles for targeted cancer therapy. Small. 2021, 17, e2006484. |
| 64. |
Jain, N.; Shahrukh, S.; Famta, P.; Shah, S.; Vambhurkar, G.; Khatri, D. K.; Singh, S. B.; Srivastava, S. Immune cell-camouflaged surface-engineered nanotherapeutics for cancer management. Acta Biomater. 2023, 155, 57-79.
doi: 10.1016/j.actbio.2022.11.001 URL |
| 65. | Lopes, J.; Lopes, D.; Pereira-Silva, M.; Peixoto, D.; Veiga, F.; Hamblin, M. R.; Conde, J.; Corbo, C.; Zare, E. N.; Ashrafizadeh, M.; Tay, F. R.; Chen, C.; Donnelly, R. F.; Wang, X.; Makvandi, P.; Paiva-Santos, A. C. Macrophage cell membrane-cloaked nanoplatforms for biomedical applications. Small Methods. 2022, 6, e2200289. |
| 66. |
Zhang, Q.; Dehaini, D.; Zhang, Y.; Zhou, J.; Chen, X.; Zhang, L.; Fang, R. H.; Gao, W.; Zhang, L. Neutrophil membrane-coated nanoparticles inhibit synovial inflammation and alleviate joint damage in inflammatory arthritis. Nat Nanotechnol. 2018, 13, 1182-1190.
doi: 10.1038/s41565-018-0254-4 |
| 67. |
Bhattacharyya, S.; Ghosh, S. S. Transmembrane TNFα-expressed macrophage membrane-coated chitosan nanoparticles as cancer therapeutics. ACS Omega. 2020, 5, 1572-1580.
doi: 10.1021/acsomega.9b03531 URL |
| 68. |
Meng, Q. F.; Rao, L.; Zan, M.; Chen, M.; Yu, G. T.; Wei, X.; Wu, Z.; Sun, Y.; Guo, S. S.; Zhao, X. Z.; Wang, F. B.; Liu, W. Macrophage membrane-coated iron oxide nanoparticles for enhanced photothermal tumor therapy. Nanotechnology. 2018, 29, 134004.
doi: 10.1088/1361-6528/aaa7c7 URL |
| 69. |
Johnson, D. T.; Zhou, J.; Kroll, A. V.; Fang, R. H.; Yan, M.; Xiao, C.; Chen, X.; Kline, J.; Zhang, L.; Zhang, D. E. Acute myeloid leukemia cell membrane-coated nanoparticles for cancer vaccination immunotherapy. Leukemia. 2022, 36, 994-1005.
doi: 10.1038/s41375-021-01432-w |
| 70. |
Kang, T.; Zhu, Q.; Wei, D.; Feng, J.; Yao, J.; Jiang, T.; Song, Q.; Wei, X.; Chen, H.; Gao, X.; Chen, J. Nanoparticles coated with neutrophil membranes can effectively treat cancer metastasis. ACS Nano. 2017, 11, 1397-1411.
doi: 10.1021/acsnano.6b06477 URL |
| 71. |
Cao, X.; Hu, Y.; Luo, S.; Wang, Y.; Gong, T.; Sun, X.; Fu, Y.; Zhang, Z. Neutrophil-mimicking therapeutic nanoparticles for targeted chemotherapy of pancreatic carcinoma. Acta Pharm Sin B. 2019, 9, 575-589.
doi: 10.1016/j.apsb.2018.12.009 URL |
| 72. |
Deng, G.; Sun, Z.; Li, S.; Peng, X.; Li, W.; Zhou, L.; Ma, Y.; Gong, P.; Cai, L. Cell-membrane immunotherapy based on natural killer cell membrane coated nanoparticles for the effective inhibition of primary and abscopal tumor growth. ACS Nano. 2018, 12, 12096-12108.
doi: 10.1021/acsnano.8b05292 URL |
| 73. |
Wu, L.; Zhang, F.; Wei, Z.; Li, X.; Zhao, H.; Lv, H.; Ge, R.; Ma, H.; Zhang, H.; Yang, B.; Li, J.; Jiang, J. Magnetic delivery of Fe(3)O(4)@polydopamine nanoparticle-loaded natural killer cells suggest a promising anticancer treatment. Biomater Sci. 2018, 6, 2714-2725.
doi: 10.1039/C8BM00588E URL |
| 74. |
Gardner, A.; Ruffell, B. Dendritic cells and cancer immunity. Trends Immunol. 2016, 37, 855-865.
doi: 10.1016/j.it.2016.09.006 URL |
| 75. |
Wculek, S. K.; Cueto, F. J.; Mujal, A. M.; Melero, I.; Krummel, M. F.; Sancho, D. Dendritic cells in cancer immunology and immunotherapy. Nat Rev Immunol. 2020, 20, 7-24.
doi: 10.1038/s41577-019-0210-z |
| 76. | Cheng, S.; Xu, C.; Jin, Y.; Li, Y.; Zhong, C.; Ma, J.; Yang, J.; Zhang, N.; Li, Y.; Wang, C.; Yang, Z.; Wang, Y. Artificial mini dendritic cells boost T cell-based immunotherapy for ovarian cancer. Adv Sci (Weinh). 2020, 7, 1903301. |
| 77. |
Ferreira-Faria, I.; Yousefiasl, S.; Macário-Soares, A.; Pereira-Silva, M.; Peixoto, D.; Zafar, H.; Raza, F.; Faneca, H.; Veiga, F.; Hamblin, M. R.; Tay, F. R.; Gao, J.; Sharifi, E.; Makvandi, P.; Paiva-Santos, A. C. Stem cell membrane-coated abiotic nanomaterials for biomedical applications. J Control Release. 2022, 351, 174-197.
doi: 10.1016/j.jconrel.2022.09.012 URL |
| 78. |
Chen, Z.; Zhao, P.; Luo, Z.; Zheng, M.; Tian, H.; Gong, P.; Gao, G.; Pan, H.; Liu, L.; Ma, A.; Cui, H.; Ma, Y.; Cai, L. Cancer cell membrane-biomimetic nanoparticles for homologous-targeting dual-modal imaging and photothermal therapy. ACS Nano. 2016, 10, 10049-10057.
doi: 10.1021/acsnano.6b04695 URL |
| 79. | Su, N.; Villicana, C.; Barati, D.; Freeman, P.; Luo, Y.; Yang, F. Stem cell membrane-coated microribbon scaffolds induce regenerative innate and adaptive immune responses in a critical-size cranial bone defect model. Adv Mater. 2023, 35, e2208781. |
| 80. |
Fang, R. H.; Hu, C. M.; Luk, B. T.; Gao, W.; Copp, J. A.; Tai, Y.; O’Connor, D. E.; Zhang, L. Cancer cell membrane-coated nanoparticles for anticancer vaccination and drug delivery. Nano Lett. 2014, 14, 2181-2188.
doi: 10.1021/nl500618u URL |
| 81. |
Chen, Q.; Zhang, L.; Li, L.; Tan, M.; Liu, W.; Liu, S.; Xie, Z.; Zhang, W.; Wang, Z.; Cao, Y.; Shang, T.; Ran, H. Cancer cell membrane-coated nanoparticles for bimodal imaging-guided photothermal therapy and docetaxel-enhanced immunotherapy against cancer. J Nanobiotechnology. 2021, 19, 449.
doi: 10.1186/s12951-021-01202-x |
| 82. | Jiang, Y.; Krishnan, N.; Zhou, J.; Chekuri, S.; Wei, X.; Kroll, A. V.; Yu, C. L.; Duan, Y.; Gao, W.; Fang, R. H.; Zhang, L. Engineered cell-membrane-coated nanoparticles directly present tumor antigens to promote anticancer immunity. Adv Mater. 2020, 32, e2001808. |
| 83. |
He, Z.; Zhang, Y.; Feng, N. Cell membrane-coated nanosized active targeted drug delivery systems homing to tumor cells: a review. Mater Sci Eng C Mater Biol Appl. 2020, 106, 110298.
doi: 10.1016/j.msec.2019.110298 URL |
| 84. |
Zeng, Y.; Li, S.; Zhang, S.; Wang, L.; Yuan, H.; Hu, F. Cell membrane coated-nanoparticles for cancer immunotherapy. Acta Pharm Sin B. 2022, 12, 3233-3254.
doi: 10.1016/j.apsb.2022.02.023 URL |
| 85. |
Rao, L.; Bu, L. L.; Cai, B.; Xu, J. H.; Li, A.; Zhang, W. F.; Sun, Z. J.; Guo, S. S.; Liu, W.; Wang, T. H.; Zhao, X. Z. Cancer cell membrane-coated upconversion nanoprobes for highly specific tumor imaging. Adv Mater. 2016, 28, 3460-3466.
doi: 10.1002/adma.v28.18 URL |
| 86. |
Yang, R.; Xu, J.; Xu, L.; Sun, X.; Chen, Q.; Zhao, Y.; Peng, R.; Liu, Z. Cancer cell membrane-coated adjuvant nanoparticles with mannose modification for effective anticancer vaccination. ACS Nano. 2018, 12, 5121-5129.
doi: 10.1021/acsnano.7b09041 URL |
| 87. |
Wang, D.; Liu, C.; You, S.; Zhang, K.; Li, M.; Cao, Y.; Wang, C.; Dong, H.; Zhang, X. Bacterial vesicle-cancer cell hybrid membrane-coated nanoparticles for tumor specific immune activation and photothermal therapy. ACS Appl Mater Interfaces. 2020, 12, 41138-41147.
doi: 10.1021/acsami.0c13169 URL |
| 88. |
Fang, R. H.; Hu, C. M.; Chen, K. N.; Luk, B. T.; Carpenter, C. W.; Gao, W.; Li, S.; Zhang, D. E.; Lu, W.; Zhang, L. Lipid-insertion enables targeting functionalization of erythrocyte membrane-cloaked nanoparticles. Nanoscale. 2013, 5, 8884-8888.
doi: 10.1039/c3nr03064d URL |
| 89. |
Ruoslahti, E. RGD and other recognition sequences for integrins. Annu Rev Cell Dev Biol. 1996, 12, 697-715.
doi: 10.1146/cellbio.1996.12.issue-1 URL |
| 90. |
Cossu, J.; Thoreau, F.; Boturyn, D. Multimeric RGD-based strategies for selective drug delivery to tumor tissues. Pharmaceutics. 2023, 15, 525.
doi: 10.3390/pharmaceutics15020525 URL |
| 91. |
Sun, J.; Jiang, L.; Lin, Y.; Gerhard, E. M.; Jiang, X.; Li, L.; Yang, J.; Gu, Z. Enhanced anticancer efficacy of paclitaxel through multistage tumor-targeting liposomes modified with RGD and KLA peptides. Int J Nanomedicine. 2017, 12, 1517-1537.
doi: 10.2147/IJN URL |
| 92. | Wu, W.; Guo, H.; Jing, D.; Zhang, Z.; Zhang, Z.; Pu, F.; Yang, W.; Jin, X.; Huang, X.; Shao, Z. Targeted delivery of PD-L1-derived phosphorylation-mimicking peptides by engineered biomimetic nanovesicles to enhance osteosarcoma treatment. Adv Healthc Mater. 2022, 11, e2200955. |
| 93. | Han, Y.; Pan, H.; Li, W.; Chen, Z.; Ma, A.; Yin, T.; Liang, R.; Chen, F.; Ma, Y.; Jin, Y.; Zheng, M.; Li, B.; Cai, L. T cell membrane mimicking nanoparticles with bioorthogonal targeting and immune recognition for enhanced photothermal therapy. Adv Sci (Weinh). 2019, 6, 1900251. |
| 94. | Park, J. H.; Jiang, Y.; Zhou, J.; Gong, H.; Mohapatra, A.; Heo, J.; Gao, W.; Fang, R. H.; Zhang, L. Genetically engineered cell membrane-coated nanoparticles for targeted delivery of dexamethasone to inflamed lungs. Sci Adv. 2021, 7, eabf7820. |
| 95. | Ma, J.; Jiang, L.; Liu, G. Cell membrane-coated nanoparticles for the treatment of bacterial infection. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2022, 14, e1825. |
| 96. |
Zhu, C.; Ma, J.; Ji, Z.; Shen, J.; Wang, Q. Recent advances of cell membrane coated nanoparticles in treating cardiovascular disorders. Molecules. 2021, 26, 3428.
doi: 10.3390/molecules26113428 URL |
| 97. |
Xiong, J.; Wu, M.; Chen, J.; Liu, Y.; Chen, Y.; Fan, G.; Liu, Y.; Cheng, J.; Wang, Z.; Wang, S.; Liu, Y.; Zhang, W. Cancer-erythrocyte hybrid membrane-camouflaged magnetic nanoparticles with enhanced photothermal-immunotherapy for ovarian cancer. ACS Nano. 2021, 15, 19756-19770.
doi: 10.1021/acsnano.1c07180 URL |
| 98. |
Chen, H. Y.; Deng, J.; Wang, Y.; Wu, C. Q.; Li, X.; Dai, H. W. Hybrid cell membrane-coated nanoparticles: A multifunctional biomimetic platform for cancer diagnosis and therapy. Acta Biomater. 2020, 112, 1-13.
doi: 10.1016/j.actbio.2020.05.028 URL |
| 99. |
Dehaini, D.; Wei, X.; Fang, R. H.; Masson, S.; Angsantikul, P.; Luk, B. T.; Zhang, Y.; Ying, M.; Jiang, Y.; Kroll, A. V.; Gao, W.; Zhang, L. Erythrocyte-platelet hybrid membrane coating for enhanced nanoparticle functionalization. Adv Mater. 2017, 29, 1606209.
doi: 10.1002/adma.v29.16 URL |
| 100. |
Jiang, Q.; Liu, Y.; Guo, R.; Yao, X.; Sung, S.; Pang, Z.; Yang, W. Erythrocyte-cancer hybrid membrane-camouflaged melanin nanoparticles for enhancing photothermal therapy efficacy in tumors. Biomaterials. 2019, 192, 292-308.
doi: 10.1016/j.biomaterials.2018.11.021 URL |
| 101. |
Zhang, Y.; Cai, K.; Li, C.; Guo, Q.; Chen, Q.; He, X.; Liu, L.; Zhang, Y.; Lu, Y.; Chen, X.; Sun, T.; Huang, Y.; Cheng, J.; Jiang, C. Macrophage-membrane-coated nanoparticles for tumor-targeted chemotherapy. Nano Lett. 2018, 18, 1908-1915.
doi: 10.1021/acs.nanolett.7b05263 URL |
| 102. |
Sun, M.; Duan, Y.; Ma, Y.; Zhang, Q. Cancer cell-erythrocyte hybrid membrane coated gold nanocages for near infrared light-activated photothermal/radio/chemotherapy of breast cancer. Int J Nanomedicine. 2020, 15, 6749-6760.
doi: 10.2147/IJN.S266405 URL |
| 103. |
Li, M.; Zhou, H.; Jiang, W.; Yang, C.; Miao, H.; Wang, Y. Nanovaccines integrating endogenous antigens and pathogenic adjuvants elicit potent antitumor immunity. Nano Today. 2020, 35, 101007.
doi: 10.1016/j.nantod.2020.101007 URL |
| 104. |
Ma, J.; Liu, F.; Sheu, W. C.; Meng, Z.; Xie, Y.; Xu, H.; Li, M.; Chen, A. T.; Liu, J.; Bao, Y.; Zhang, X.; Zhang, S.; Zhang, L.; Zou, Z.; Wu, H.; Wang, H.; Zhu, Y.; Zhou, J. Copresentation of tumor antigens and costimulatory molecules via biomimetic nanoparticles for effective cancer immunotherapy. Nano Lett. 2020, 20, 4084-4094.
doi: 10.1021/acs.nanolett.9b05171 URL |
| 105. |
Liu, W. L.; Zou, M. Z.; Liu, T.; Zeng, J. Y.; Li, X.; Yu, W. Y.; Li, C. X.; Ye, J. J.; Song, W.; Feng, J.; Zhang, X. Z. Cytomembrane nanovaccines show therapeutic effects by mimicking tumor cells and antigen presenting cells. Nat Commun. 2019, 10, 3199.
doi: 10.1038/s41467-019-11157-1 |
| 106. | Chen, R.; Yang, J.; Wu, M.; Zhao, D.; Yuan, Z.; Zeng, L.; Hu, J.; Zhang, X.; Wang, T.; Xu, J.; Zhang, J. M2 macrophage hybrid membrane-camouflaged targeted biomimetic nanosomes to reprogram inflammatory microenvironment for enhanced enzyme-thermo-immunotherapy. Adv Mater. 2023, 35, e2304123. |
| No related articles found! |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||