Biomaterials Translational ›› 2024, Vol. 5 ›› Issue (1): 46-58.doi: 10.12336/biomatertransl.2024.01.005
• REVIEWS • Previous Articles Next Articles
Xiaoxiang Ren1,5, Ruixue Xu2,*( ), Chenjie Xu3,*(
), Chenjie Xu3,*( ), Jiacan Su1,4,5,*(
), Jiacan Su1,4,5,*( )
)
Received:2024-01-03
Revised:2024-01-22
Accepted:2024-02-06
Online:2024-03-28
Published:2024-03-28
Contact:
Ruixue Xu, 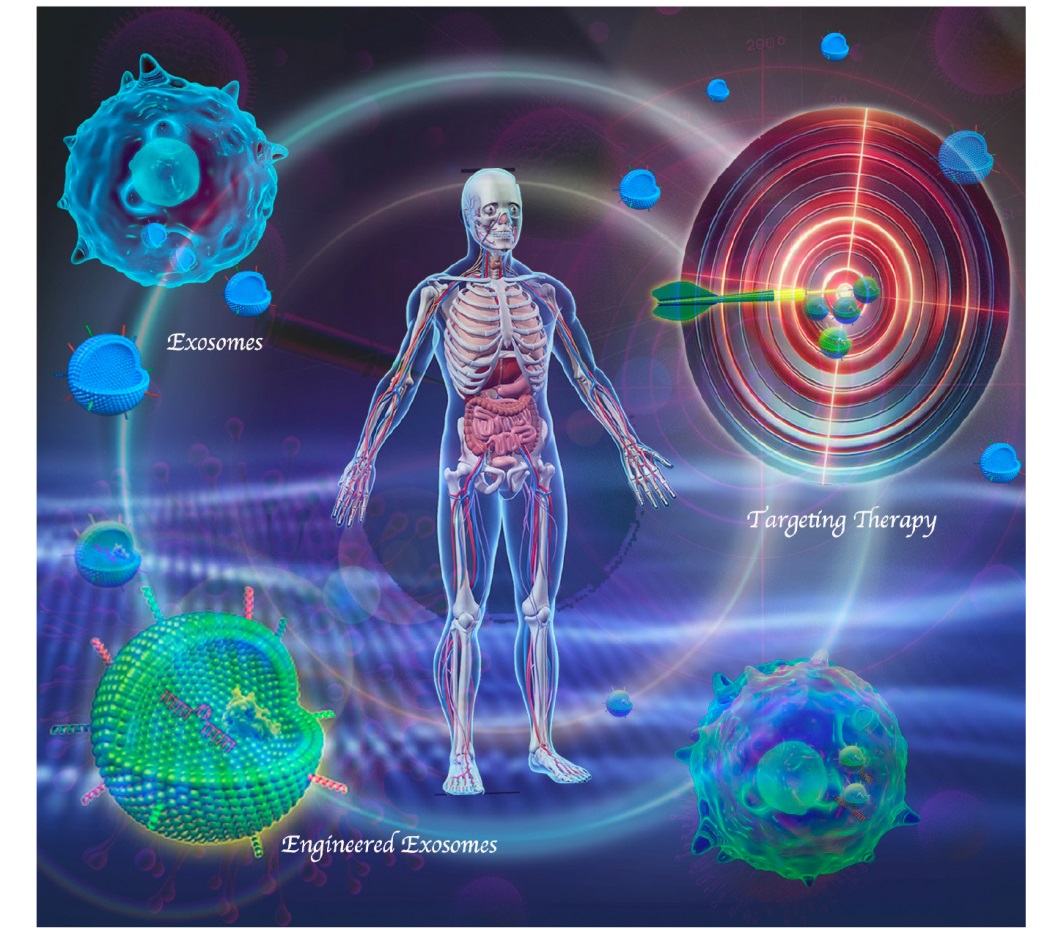
Figure 1. Engineered exosomes offer a targeted therapeutic approach, enhanced by direct or indirect modifications to treat diseases with precision and reduced toxicity. Created with Adobe Photoshop 2024.
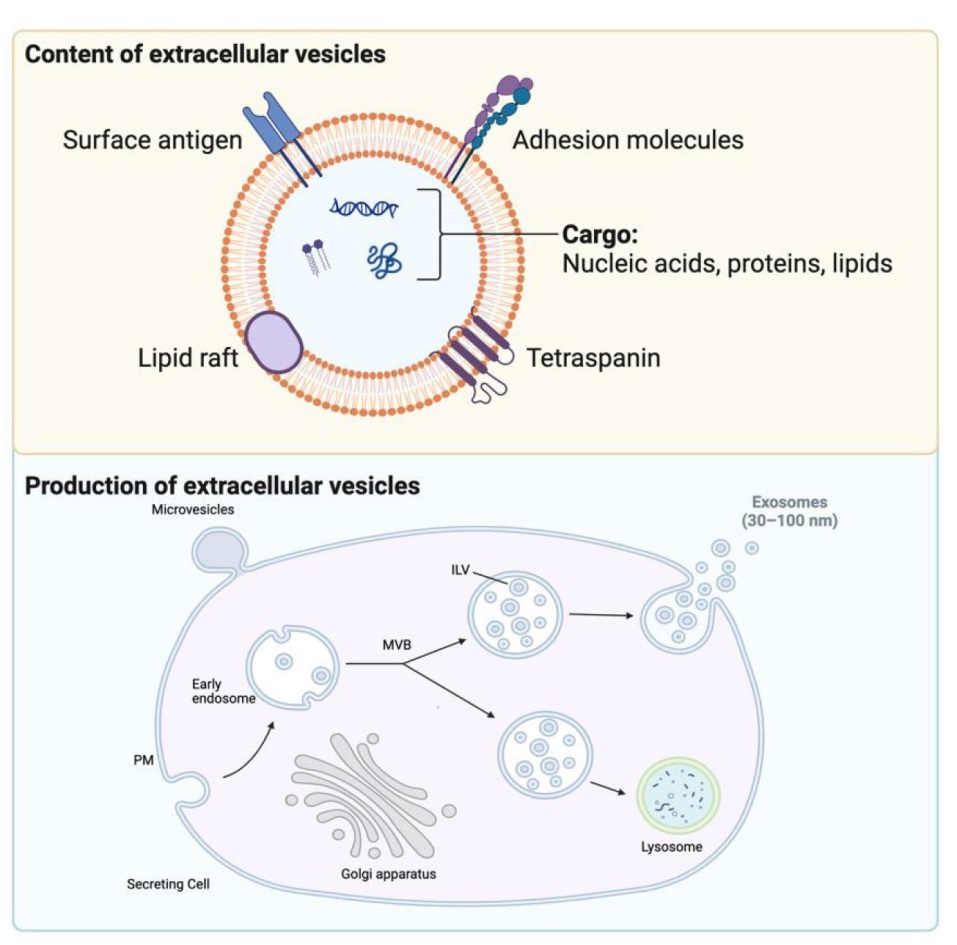
Figure 2. The content and production of exosomes. ILV: intraluminal vesicle; MVB: multivesicular body; PM: plasma membrane. Created with BioRender.com.
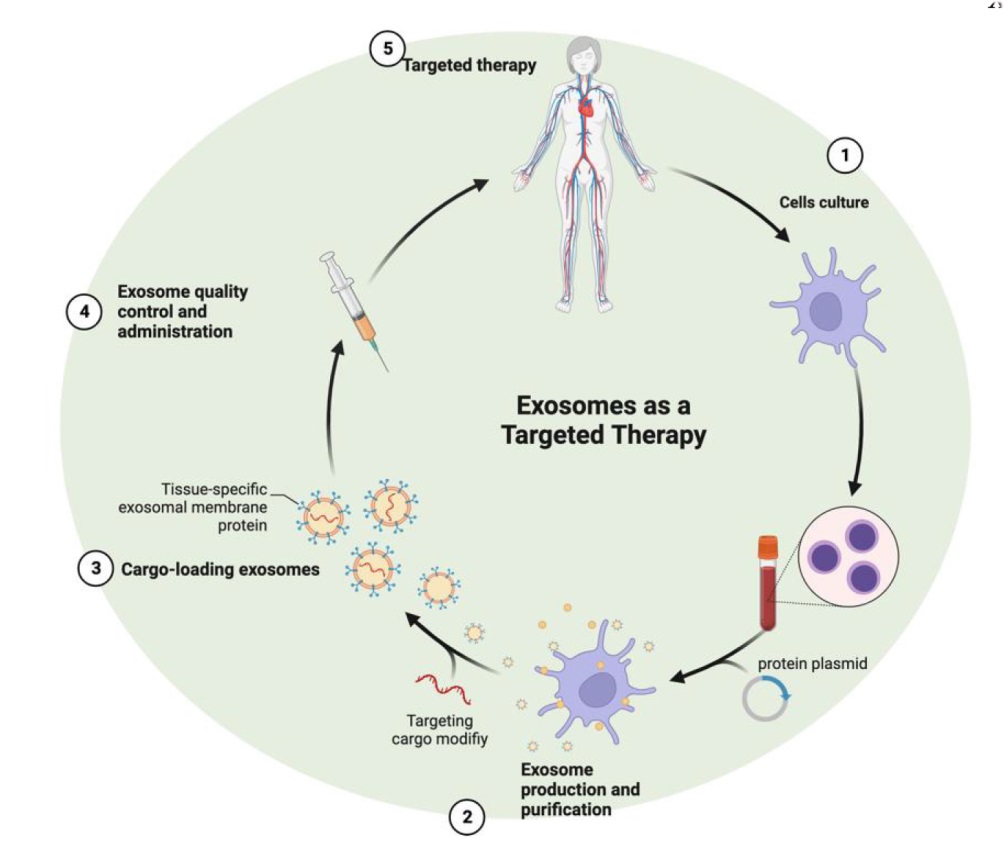
Figure 3. The process of targeted therapy of exosomes. 1. Cells culture: The process begins with the culture of cells, which are the source of exosomes. 2. Exosome production and purification: Following cell culture, exosomes are produced and then isolated and purified from the cell media. 3. Cargo-loading exosomes: The isolated exosomes are then loaded with therapeutic cargo. This step involves incorporating the desired molecules, such as drugs or proteins, into the exosomes. 4. Exosome quality control and administration: After loading the cargo, the exosomes undergo quality control to ensure they meet the necessary standards for therapeutic use. Once approved, they are ready for administration to the patient. 5. Targeted therapy: The final step is the administration of these engineered exosomes to the patient, where they can deliver their therapeutic cargo to the targeted tissues or cells in the body. This sequence represents the full cycle from cell culture to the delivery of targeted therapy using exosomes. Created with BioRender.com.
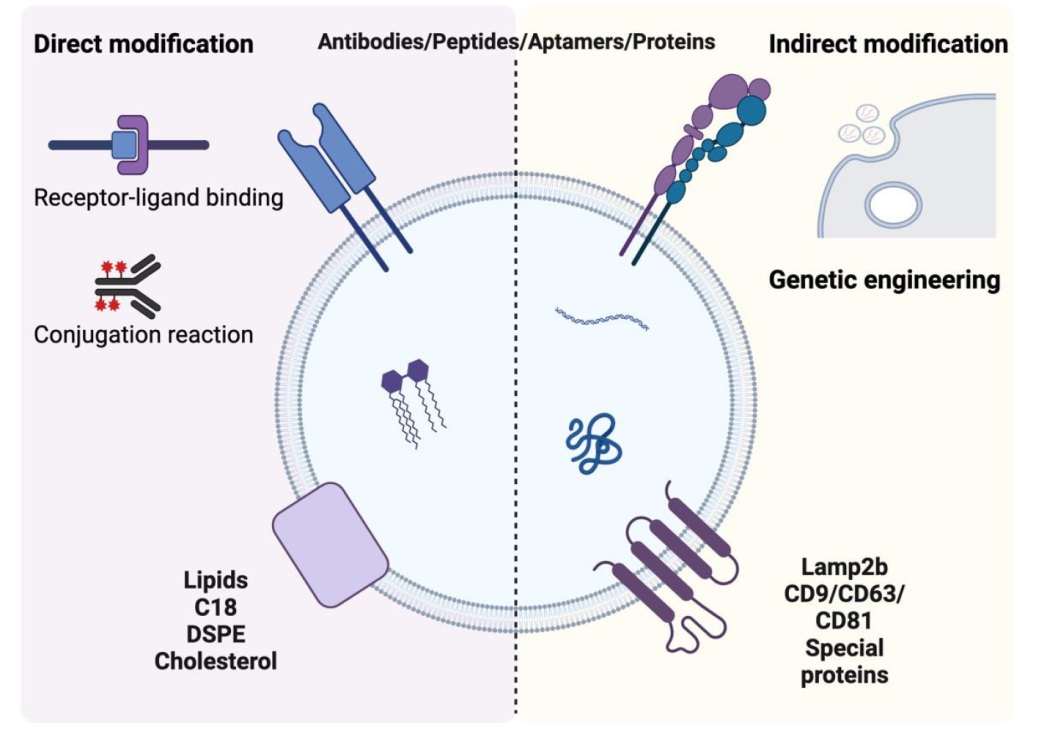
Figure 4. The modification strategy of exosomes. DSPE: 1,2-distearoyl-sn-glycero-3-phosphoethanolamine; Lamp2b: lysosome-associated membrane protein 2b. Created with BioRender.com.
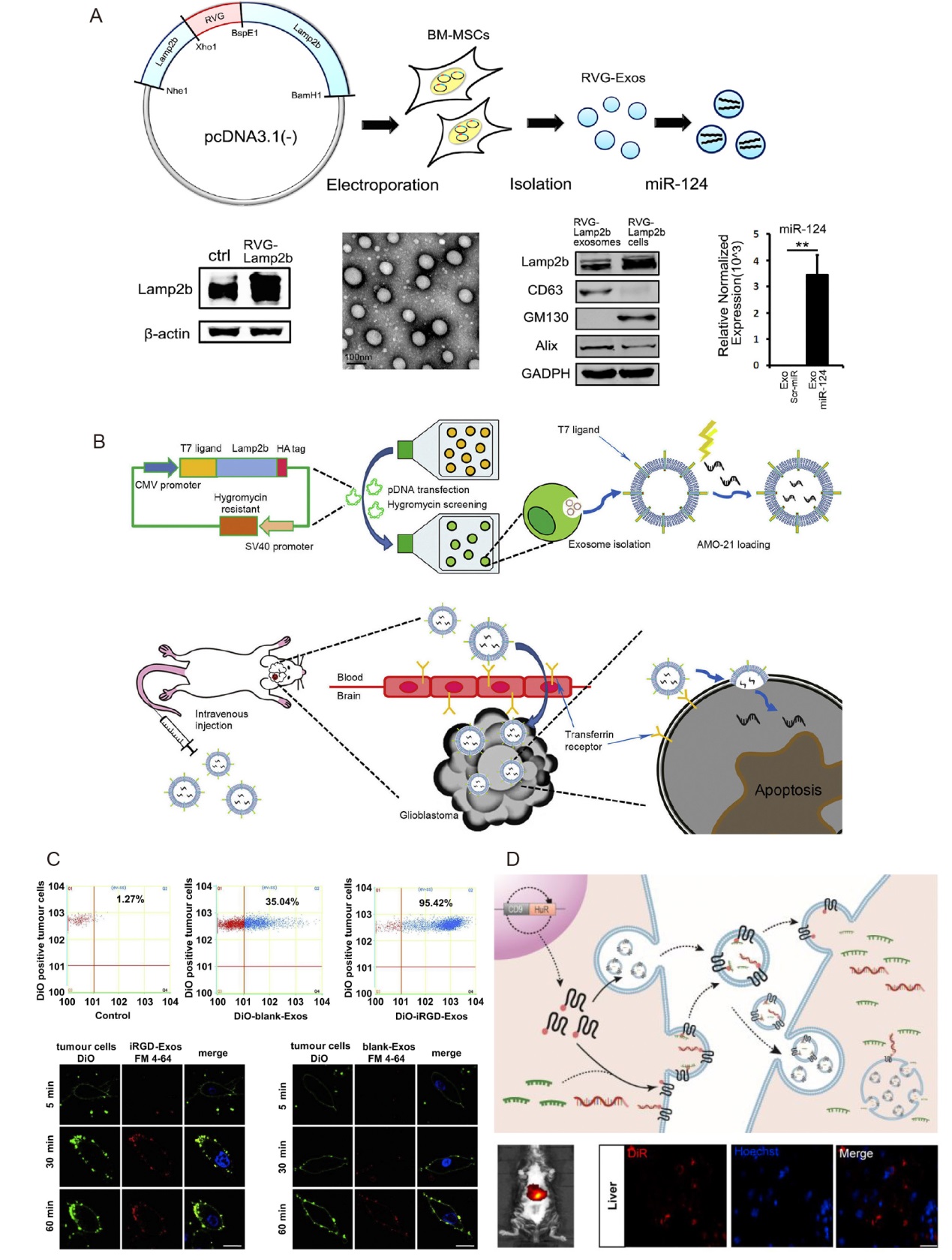
Figure 5. Genetically modified exosomes. (A) Exosomes modified with RVG-Lamp2b promote neurogenesis. Reprinted from Yang et al.39 (B) T7-peptide decorated exosomes deliver microRNA-21 antisense oligonucleotides to the brain. Reprinted from Kim et al.41 Copyright 2019, with permission from Elsevier B.V. (C) Binding of iRGD-Exos to a human breast cancer cell line in vitro. Reprinted from Tian et al.43 Copyright 2013, with permission from Elsevier B.V. (D) CD9-HuR functionalized exosomes encapsulated with miRNA or CRISPR/dCas9. Reprinted with permission from Li et al.45 Copyright 2019, American Chemical Society. Alix: apoptosis linked gene-2-interacting protein X; BM-MSC: bone marrow derived mesenchymal stem cell; CMV: cytomegalovirus; DiO: 3,3′-dioctadecyloxacarbocyanine perchlorate; DiR: 1,1′-dioctadecyl-3,3,3′,3′-tetramethylindotricarbocyanine iodide; Exos: exosomes; GAPDH: glyceraldehyde-3-phosphate dehydrogenase; GM130: Golgi matrix protein 130; HuR: human antigen R; miRNA: microRNA; RVG: rabies virus glycoprotein.
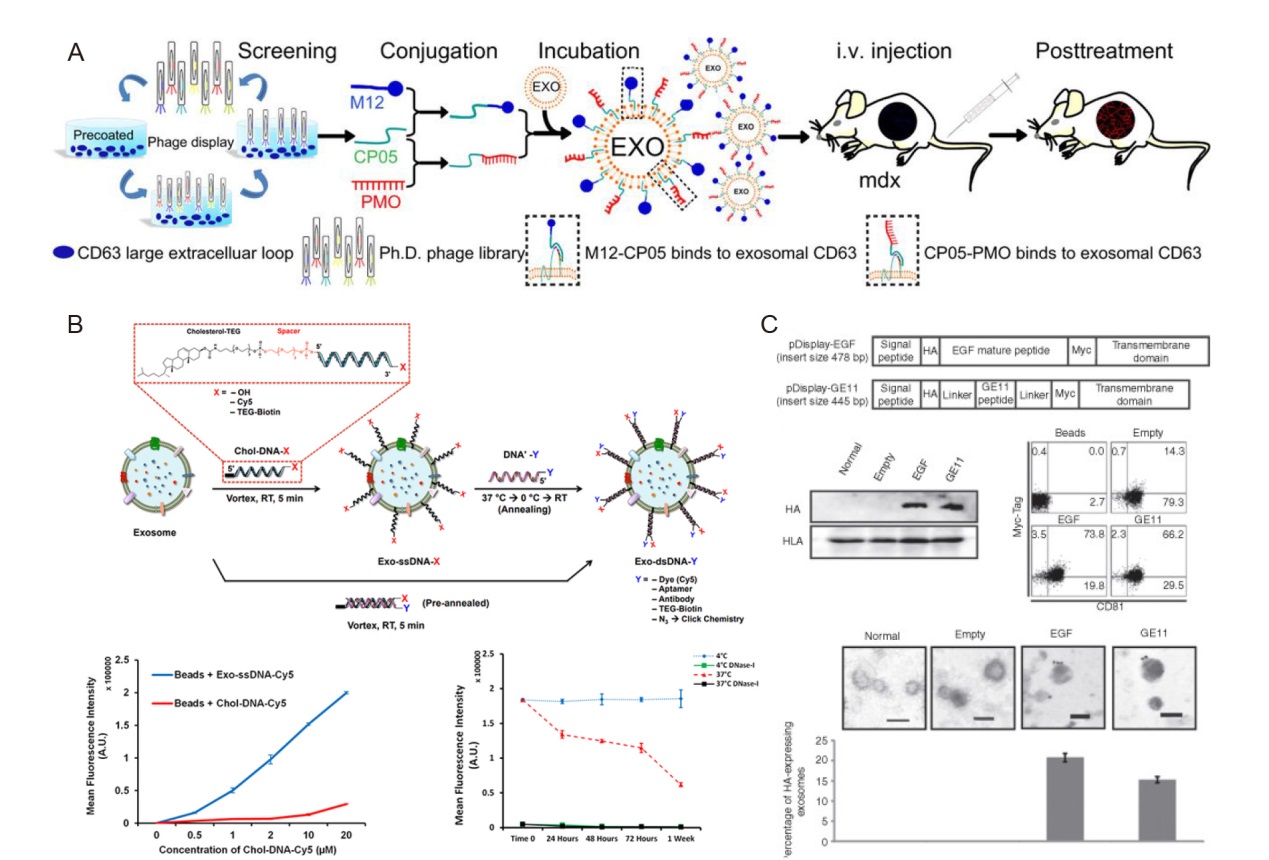
Figure 6. Direct modified exosomes. (A) Screening for exosomal anchor peptides on exosomes. Reprinted from Gao et al.50 Copyright 2018 Gao et al., some rights reserved; exclusive licensee American Association for the Advancement of Science. (B) Cholesterol-oligonucleotide tethering method on exosome membrane. Reprinted with permission from Yerneni et al.51 Copyright 2019, American Chemical Society. (C) Epidermal growth factor receptor (EGFR) ligands on the outer surfaces of the exosomes. Reprinted from Ohno et al.57 Copyright ? 2013 The American Society of Gene & Cell Therapy. Published by Elsevier Inc. A.U.: augmentation unit; dsRNA: double-stranded RNA; EGF: epidermal growth factor; EXO: exosome; i.v.: intravenous; Ph.D. phage library: Ph.D.-12 phage display library (New England BioLabs, Ipswich, MA, USA); PMO: phosphorodiamidate morpholino oligomer; RT: room temperature; ssRNA: single-stranded RNA.
| 1. |
Ren, X.; Chen, X.; Geng, Z.; Su, J. Bone-targeted biomaterials: strategies and applications. Chem Eng J. 2022, 446, 137133.
doi: 10.1016/j.cej.2022.137133 URL |
| 2. |
Liu, J.; Zhang, Y.; Wu, Y.; Li, G.; Ji, N.; Han, R.; Tang, H.; Liu, X.; Liu, H.; Wang, C.; Cui, J.; Song, P.; Jing, Y.; Chen, X.; Su, J. Delivery of m7G methylated Runx2 mRNA by bone-targeted lipid nanoparticle promotes osteoblastic bone formation in senile osteoporosis. Nano Today. 2024, 54, 102074.
doi: 10.1016/j.nantod.2023.102074 URL |
| 3. |
Ren, X.; Sun, Z.; Ma, X.; Wang, Y.; Cui, X.; Yi, Z.; Sun, X.; Guo, B.; Li, X. Alginate-mediated mineralization for ultrafine hydroxyapatite hybrid nanoparticles. Langmuir. 2018, 34, 6797-6805.
doi: 10.1021/acs.langmuir.8b00151 URL |
| 4. | Wang, J.; Li, X.; Wang, S.; Cui, J.; Ren, X.; Su, J. Bone-targeted exosomes: strategies and applications. Adv Healthc Mater. 2023, 12, e2203361. |
| 5. |
Huang, L.; Wu, E.; Liao, J.; Wei, Z.; Wang, J.; Chen, Z. Research advances of engineered exosomes as drug delivery carrier. ACS Omega. 2023, 8, 43374-43387.
doi: 10.1021/acsomega.3c04479 URL |
| 6. |
Moloudizargari, M.; Asghari, M. H.; Goel, A. The therapeutic triad of extracellular vesicles: As drug targets, as drugs, and as drug carriers. Biochem Pharmacol. 2021, 192, 114714.
doi: 10.1016/j.bcp.2021.114714 URL |
| 7. |
Lu, M.; Huang, Y. Bioinspired exosome-like therapeutics and delivery nanoplatforms. Biomaterials. 2020, 242, 119925.
doi: 10.1016/j.biomaterials.2020.119925 URL |
| 8. |
Gao, S.; Yang, X.; Xu, J.; Qiu, N.; Zhai, G. Nanotechnology for boosting cancer immunotherapy and remodeling tumor microenvironment: the horizons in cancer treatment. ACS Nano. 2021, 15, 12567-12603.
doi: 10.1021/acsnano.1c02103 URL |
| 9. |
He, J.; Ren, W.; Wang, W.; Han, W.; Jiang, L.; Zhang, D.; Guo, M. Exosomal targeting and its potential clinical application. Drug Deliv Transl Res. 2022, 12, 2385-2402.
doi: 10.1007/s13346-021-01087-1 |
| 10. |
Jiang, Y.; Li, J.; Xue, X.; Yin, Z.; Xu, K.; Su, J. Engineered extracellular vesicles for bone therapy. Nano Today. 2022, 44, 101487.
doi: 10.1016/j.nantod.2022.101487 URL |
| 11. |
Yáñez-Mó, M.; Siljander, P. R.; Andreu, Z.; Zavec, A. B.; Borràs, F. E.; Buzas, E. I.; Buzas, K.; Casal, E.; Cappello, F.; Carvalho, J.; Colás, E.; Cordeiro-da Silva, A.; Fais, S.; Falcon-Perez, J. M.; Ghobrial, I. M.; Giebel, B.; Gimona, M.; Graner, M.; Gursel, I.; Gursel, M.; Heegaard, N. H.; Hendrix, A.; Kierulf, P.; Kokubun, K.; Kosanovic, M.; Kralj-Iglic, V.; Krämer-Albers, E. M.; Laitinen, S.; Lässer, C.; Lener, T.; Ligeti, E.; Linē, A.; Lipps, G.; Llorente, A.; Lötvall, J.; Manček-Keber, M.; Marcilla, A.; Mittelbrunn, M.; Nazarenko, I.; Nolte-’t Hoen, E. N.; Nyman, T. A.; O’Driscoll, L.; Olivan, M.; Oliveira, C.; Pállinger, É.; Del Portillo, H. A.; Reventós, J.; Rigau, M.; Rohde, E.; Sammar, M.; Sánchez-Madrid, F.; Santarém, N.; Schallmoser, K.; Ostenfeld, M. S.; Stoorvogel, W.; Stukelj, R.; Van der Grein, S. G.; Vasconcelos, M. H.; Wauben, M. H.; De Wever, O. Biological properties of extracellular vesicles and their physiological functions. J Extracell Vesicles. 2015, 4, 27066.
doi: 10.3402/jev.v4.27066 URL |
| 12. |
Saad, M. H.; Badierah, R.; Redwan, E. M.; El-Fakharany, E. M. A comprehensive insight into the role of exosomes in viral infection: dual faces bearing different functions. Pharmaceutics. 2021, 13, 1405.
doi: 10.3390/pharmaceutics13091405 URL |
| 13. |
Gurung, S.; Perocheau, D.; Touramanidou, L.; Baruteau, J. The exosome journey: from biogenesis to uptake and intracellular signalling. Cell Commun Signal. 2021, 19, 47.
doi: 10.1186/s12964-021-00730-1 URL |
| 14. |
Isaac, R.; Reis, F. C. G.; Ying, W.; Olefsky, J. M. Exosomes as mediators of intercellular crosstalk in metabolism. Cell Metab. 2021, 33, 1744-1762.
doi: 10.1016/j.cmet.2021.08.006 URL |
| 15. |
Sun, Z.; Wu, Y.; Gao, F.; Li, H.; Wang, C.; Du, L.; Dong, L.; Jiang, Y. In situ detection of exosomal RNAs for cancer diagnosis. Acta Biomater. 2023, 155, 80-98.
doi: 10.1016/j.actbio.2022.10.061 URL |
| 16. |
Liu, S. L.; Sun, P.; Li, Y.; Liu, S. S.; Lu, Y. Exosomes as critical mediators of cell-to-cell communication in cancer pathogenesis and their potential clinical application. Transl Cancer Res. 2019, 8, 298-311.
doi: 10.21037/tcr URL |
| 17. |
Spanos, M.; Gokulnath, P.; Chatterjee, E.; Li, G.; Varrias, D.; Das, S. Expanding the horizon of EV-RNAs: lncRNAs in EVs as biomarkers for disease pathways. Extracell Vesicle. 2023, 2, 100025.
doi: 10.1016/j.vesic.2023.100025 URL |
| 18. |
Han, R.; Wu, Y.; Han, Y.; Liu, X.; Liu, H.; Su, J. Engineered plant extracellular vesicles for autoimmune diseases therapy. Nano Res. 2024, 17, 2857-2873.
doi: 10.1007/s12274-023-6112-1 |
| 19. |
Abels, E. R.; Breakefield, X. O. Introduction to extracellular vesicles: biogenesis, RNA cargo selection, content, release, and uptake. Cell Mol Neurobiol. 2016, 36, 301-312.
doi: 10.1007/s10571-016-0366-z URL |
| 20. | Janas, A. M.; Sapoń, K.; Janas, T.; Stowell, M. H.; Janas, T. Exosomes and other extracellular vesicles in neural cells and neurodegenerative diseases. Biochim Biophys Acta. 2016, 1858, 1139-1151. |
| 21. | Xu, M.; Ji, J.; Jin, D.; Wu, Y.; Wu, T.; Lin, R.; Zhu, S.; Jiang, F.; Ji, Y.; Bao, B.; Li, M.; Xu, W.; Xiao, M. The biogenesis and secretion of exosomes and multivesicular bodies (MVBs): intercellular shuttles and implications in human diseases. Genes Dis. 2023, 10, 1894-1907. |
| 22. | Liu, C.; Liu, D.; Wang, S.; Gan, L.; Yang, X.; Ma, C. Identification of the SNARE complex that mediates the fusion of multivesicular bodies with the plasma membrane in exosome secretion. J Extracell Vesicles. 2023, 12, e12356. |
| 23. | Kalluri, R.; LeBleu, V. S. The biology, function, and biomedical applications of exosomes. Science. 2020, 367, eaau6977. |
| 24. |
Wang, Y.; Guo, M.; Lin, D.; Liang, D.; Zhao, L.; Zhao, R.; Wang, Y. Docetaxel-loaded exosomes for targeting non-small cell lung cancer: preparation and evaluation in vitro and in vivo. Drug Deliv. 2021, 28, 1510-1523.
doi: 10.1080/10717544.2021.1951894 URL |
| 25. |
Montecalvo, A.; Larregina, A. T.; Shufesky, W. J.; Stolz, D. B.; Sullivan, M. L.; Karlsson, J. M.; Baty, C. J.; Gibson, G. A.; Erdos, G.; Wang, Z.; Milosevic, J.; Tkacheva, O. A.; Divito, S. J.; Jordan, R.; Lyons-Weiler, J.; Watkins, S. C.; Morelli, A. E. Mechanism of transfer of functional microRNAs between mouse dendritic cells via exosomes. Blood. 2012, 119, 756-766.
doi: 10.1182/blood-2011-02-338004 URL |
| 26. |
Zhu, L.; Sun, H. T.; Wang, S.; Huang, S. L.; Zheng, Y.; Wang, C. Q.; Hu, B. Y.; Qin, W.; Zou, T. T.; Fu, Y.; Shen, X. T.; Zhu, W. W.; Geng, Y.; Lu, L.; Jia, H. L.; Qin, L. X.; Dong, Q. Z. Isolation and characterization of exosomes for cancer research. J Hematol Oncol. 2020, 13, 152.
doi: 10.1186/s13045-020-00987-y |
| 27. |
Nolte-’t Hoen, E. N.; Buschow, S. I.; Anderton, S. M.; Stoorvogel, W.; Wauben, M. H. Activated T cells recruit exosomes secreted by dendritic cells via LFA-1. Blood. 2009, 113, 1977-1981.
doi: 10.1182/blood-2008-08-174094 URL |
| 28. |
Xie, Y.; Zhang, H.; Li, W.; Deng, Y.; Munegowda, M. A.; Chibbar, R.; Qureshi, M.; Xiang, J. Dendritic cells recruit T cell exosomes via exosomal LFA-1 leading to inhibition of CD8+ CTL responses through downregulation of peptide/MHC class I and Fas ligand-mediated cytotoxicity. J Immunol. 2010, 185, 5268-5278.
doi: 10.4049/jimmunol.1000386 URL |
| 29. |
Saunderson, S. C.; Dunn, A. C.; Crocker, P. R.; McLellan, A. D. CD169 mediates the capture of exosomes in spleen and lymph node. Blood. 2014, 123, 208-216.
doi: 10.1182/blood-2013-03-489732 URL |
| 30. |
Enomoto, Y.; Li, P.; Jenkins, L. M.; Anastasakis, D.; Lyons, G. C.; Hafner, M.; Leonard, W. J. Cytokine-enhanced cytolytic activity of exosomes from NK cells. Cancer Gene Ther. 2022, 29, 734-749.
doi: 10.1038/s41417-021-00352-2 |
| 31. |
Ma, Y. S.; Wu, T. M.; Ling, C. C.; Yu, F.; Zhang, J.; Cao, P. S.; Gu, L. P.; Wang, H. M.; Xu, H.; Li, L.; Wu, Z. J.; Wang, G. R.; Li, W.; Lin, Q. L.; Liu, J. B.; Fu, D. M2 macrophage-derived exosomal microRNA-155-5p promotes the immune escape of colon cancer by downregulating ZC3H12B. Mol Ther Oncolytics. 2021, 20, 484-498.
doi: 10.1016/j.omto.2021.02.005 URL |
| 32. |
Chen, H.; Wang, L.; Zeng, X.; Schwarz, H.; Nanda, H. S.; Peng, X.; Zhou, Y. Exosomes, a new star for targeted delivery. Front Cell Dev Biol. 2021, 9, 751079.
doi: 10.3389/fcell.2021.751079 URL |
| 33. |
Liang, Y.; Duan, L.; Lu, J.; Xia, J. Engineering exosomes for targeted drug delivery. Theranostics. 2021, 11, 3183-3195.
doi: 10.7150/thno.52570 URL |
| 34. | Andreu, Z.; Yáñez-Mó, M. Tetraspanins in extracellular vesicle formation and function. Front Immunol. 2014, 5, 442. |
| 35. | Simhadri, V. R.; Reiners, K. S.; Hansen, H. P.; Topolar, D.; Simhadri, V. L.; Nohroudi, K.; Kufer, T. A.; Engert, A.; Pogge von Strandmann, E. Dendritic cells release HLA-B-associated transcript-3 positive exosomes to regulate natural killer function. PLoS One. 2008, 3, e3377. |
| 36. | Chistiakov, D. A.; Killingsworth, M. C.; Myasoedova, V. A.; Orekhov, A. N.; Bobryshev, Y. V. CD68/macrosialin: not just a histochemical marker. Lab Invest. 2017, 97, 4-13. |
| 37. |
Wilke, S.; Krausze, J.; Büssow, K. Crystal structure of the conserved domain of the DC lysosomal associated membrane protein: implications for the lysosomal glycocalyx. BMC Biol. 2012, 10, 62.
doi: 10.1186/1741-7007-10-62 |
| 38. |
El-Andaloussi, S.; Lee, Y.; Lakhal-Littleton, S.; Li, J.; Seow, Y.; Gardiner, C.; Alvarez-Erviti, L.; Sargent, I. L.; Wood, M. J. Exosome-mediated delivery of siRNA in vitro and in vivo. Nat Protoc. 2012, 7, 2112-2126.
doi: 10.1038/nprot.2012.131 |
| 39. |
Yang, J.; Zhang, X.; Chen, X.; Wang, L.; Yang, G. Exosome mediated delivery of miR-124 promotes neurogenesis after ischemia. Mol Ther Nucleic Acids. 2017, 7, 278-287.
doi: 10.1016/j.omtn.2017.04.010 URL |
| 40. |
Liu, Y.; Li, D.; Liu, Z.; Zhou, Y.; Chu, D.; Li, X.; Jiang, X.; Hou, D.; Chen, X.; Chen, Y.; Yang, Z.; Jin, L.; Jiang, W.; Tian, C.; Zhou, G.; Zen, K.; Zhang, J.; Zhang, Y.; Li, J.; Zhang, C. Y. Targeted exosome-mediated delivery of opioid receptor Mu siRNA for the treatment of morphine relapse. Sci Rep. 2015, 5, 17543.
doi: 10.1038/srep17543 |
| 41. |
Kim, G.; Kim, M.; Lee, Y.; Byun, J. W.; Hwang, D. W.; Lee, M. Systemic delivery of microRNA-21 antisense oligonucleotides to the brain using T7-peptide decorated exosomes. J Control Release. 2020, 317, 273-281.
doi: 10.1016/j.jconrel.2019.11.009 URL |
| 42. |
Kim, G.; Lee, Y.; Ha, J.; Han, S.; Lee, M. Engineering exosomes for pulmonary delivery of peptides and drugs to inflammatory lung cells by inhalation. J Control Release. 2021, 330, 684-695.
doi: 10.1016/j.jconrel.2020.12.053 URL |
| 43. |
Tian, Y.; Li, S.; Song, J.; Ji, T.; Zhu, M.; Anderson, G. J.; Wei, J.; Nie, G. A doxorubicin delivery platform using engineered natural membrane vesicle exosomes for targeted tumor therapy. Biomaterials. 2014, 35, 2383-2390.
doi: 10.1016/j.biomaterials.2013.11.083 URL |
| 44. |
Bellavia, D.; Raimondo, S.; Calabrese, G.; Forte, S.; Cristaldi, M.; Patinella, A.; Memeo, L.; Manno, M.; Raccosta, S.; Diana, P.; Cirrincione, G.; Giavaresi, G.; Monteleone, F.; Fontana, S.; De Leo, G.; Alessandro, R. Interleukin 3- receptor targeted exosomes inhibit in vitro and in vivo chronic myelogenous leukemia cell growth. Theranostics. 2017, 7, 1333-1345.
doi: 10.7150/thno.17092 URL |
| 45. |
Li, Z.; Zhou, X.; Wei, M.; Gao, X.; Zhao, L.; Shi, R.; Sun, W.; Duan, Y.; Yang, G.; Yuan, L. In vitro and in vivo RNA inhibition by CD9-HuR functionalized exosomes encapsulated with miRNA or CRISPR/dCas9. Nano Lett. 2019, 19, 19-28.
doi: 10.1021/acs.nanolett.8b02689 URL |
| 46. |
Shi, X.; Cheng, Q.; Hou, T.; Han, M.; Smbatyan, G.; Lang, J. E.; Epstein, A. L.; Lenz, H. J.; Zhang, Y. Genetically engineered cell-derived nanoparticles for targeted breast cancer immunotherapy. Mol Ther. 2020, 28, 536-547.
doi: 10.1016/j.ymthe.2019.11.020 URL |
| 47. |
Molavipordanjani, S.; Khodashenas, S.; Abedi, S. M.; Moghadam, M. F.; Mardanshahi, A.; Hosseinimehr, S. J. (99m)Tc-radiolabeled HER2 targeted exosome for tumor imaging. Eur J Pharm Sci. 2020, 148, 105312.
doi: 10.1016/j.ejps.2020.105312 URL |
| 48. |
Kooijmans, S. A.; Aleza, C. G.; Roffler, S. R.; van Solinge, W. W.; Vader, P.; Schiffelers, R. M. Display of GPI-anchored anti-EGFR nanobodies on extracellular vesicles promotes tumour cell targeting. J Extracell Vesicles. 2016, 5, 31053.
doi: 10.3402/jev.v5.31053 URL |
| 49. | Sun, S.; Liu, H.; Hu, Y.; Wang, Y.; Zhao, M.; Yuan, Y.; Han, Y.; Jing, Y.; Cui, J.; Ren, X.; Chen, X.; Su, J. Selection and identification of a novel ssDNA aptamer targeting human skeletal muscle. Bioact Mater. 2023, 20, 166-178. |
| 50. | Gao, X.; Ran, N.; Dong, X.; Zuo, B.; Yang, R.; Zhou, Q.; Moulton, H. M.; Seow, Y.; Yin, H. Anchor peptide captures, targets, and loads exosomes of diverse origins for diagnostics and therapy. Sci Transl Med. 2018, 10, eaat0195. |
| 51. |
Yerneni, S. S.; Lathwal, S.; Shrestha, P.; Shirwan, H.; Matyjaszewski, K.; Weiss, L.; Yolcu, E. S.; Campbell, P. G.; Das, S. R. Rapid on-demand extracellular vesicle augmentation with versatile oligonucleotide tethers. ACS Nano. 2019, 13, 10555-10565.
doi: 10.1021/acsnano.9b04651 URL |
| 52. |
Luo, Z. W.; Li, F. X.; Liu, Y. W.; Rao, S. S.; Yin, H.; Huang, J.; Chen, C. Y.; Hu, Y.; Zhang, Y.; Tan, Y. J.; Yuan, L. Q.; Chen, T. H.; Liu, H. M.; Cao, J.; Liu, Z. Z.; Wang, Z. X.; Xie, H. Aptamer-functionalized exosomes from bone marrow stromal cells target bone to promote bone regeneration. Nanoscale. 2019, 11, 20884-20892.
doi: 10.1039/C9NR02791B URL |
| 53. |
Wang, J.; Li, W.; Lu, Z.; Zhang, L.; Hu, Y.; Li, Q.; Du, W.; Feng, X.; Jia, H.; Liu, B. F. The use of RGD-engineered exosomes for enhanced targeting ability and synergistic therapy toward angiogenesis. Nanoscale. 2017, 9, 15598-15605.
doi: 10.1039/C7NR04425A URL |
| 54. |
Liu, S. Radiolabeled cyclic RGD peptides as integrin alpha(v)beta(3)-targeted radiotracers: maximizing binding affinity via bivalency. Bioconjug Chem. 2009, 20, 2199-2213.
doi: 10.1021/bc900167c URL |
| 55. |
Avgoulas, D. I.; Tasioulis, K. S.; Papi, R. M.; Pantazaki, A. A. Therapeutic and diagnostic potential of exosomes as drug delivery systems in brain cancer. Pharmaceutics. 2023, 15, 1439.
doi: 10.3390/pharmaceutics15051439 URL |
| 56. |
Li, S.; Wu, Y.; Ding, F.; Yang, J.; Li, J.; Gao, X.; Zhang, C.; Feng, J. Engineering macrophage-derived exosomes for targeted chemotherapy of triple-negative breast cancer. Nanoscale. 2020, 12, 10854-10862.
doi: 10.1039/D0NR00523A URL |
| 57. |
Ohno, S.; Takanashi, M.; Sudo, K.; Ueda, S.; Ishikawa, A.; Matsuyama, N.; Fujita, K.; Mizutani, T.; Ohgi, T.; Ochiya, T.; Gotoh, N.; Kuroda, M. Systemically injected exosomes targeted to EGFR deliver antitumor microRNA to breast cancer cells. Mol Ther. 2013, 21, 185-191.
doi: 10.1038/mt.2012.180 URL |
| 58. |
Ji, N.; Wang, F.; Wang, M.; Zhang, W.; Liu, H.; Su, J. Engineered bacterial extracellular vesicles for central nervous system diseases. J Control Release. 2023, 364, 46-60.
doi: 10.1016/j.jconrel.2023.10.027 URL |
| 59. |
Mentkowski, K. I.; Lang, J. K. Exosomes engineered to express a cardiomyocyte binding peptide demonstrate improved cardiac retention in vivo. Sci Rep. 2019, 9, 10041.
doi: 10.1038/s41598-019-46407-1 |
| 60. |
Sun, J.; Yin, Z.; Wang, X.; Su, J. Exosome-laden hydrogels: a novel cell-free strategy for in-situ bone tissue regeneration. Front Bioeng Biotechnol. 2022, 10, 866208.
doi: 10.3389/fbioe.2022.866208 URL |
| 61. |
Ferreira, D.; Moreira, J. N.; Rodrigues, L. R. New advances in exosome-based targeted drug delivery systems. Crit Rev Oncol Hematol. 2022, 172, 103628.
doi: 10.1016/j.critrevonc.2022.103628 URL |
| 62. |
Kim, H.; Jang, H.; Cho, H.; Choi, J.; Hwang, K. Y.; Choi, Y.; Kim, S. H.; Yang, Y. Recent advances in exosome-based drug delivery for cancer therapy. Cancers (Basel). 2021, 13, 4435.
doi: 10.3390/cancers13174435 URL |
| 63. |
Li, X.; Corbett, A. L.; Taatizadeh, E.; Tasnim, N.; Little, J. P.; Garnis, C.; Daugaard, M.; Guns, E.; Hoorfar, M.; Li, I. T. S. Challenges and opportunities in exosome research-perspectives from biology, engineering, and cancer therapy. APL Bioeng. 2019, 3, 011503.
doi: 10.1063/1.5087122 URL |
| 64. |
Ren, X.; Yi, Z.; Sun, Z.; Ma, X.; Chen, G.; Chen, Z.; Li, X. Natural polysaccharide-incorporated hydroxyapatite as size-changeable, nuclear-targeted nanocarrier for efficient cancer therapy. Biomater Sci. 2020, 8, 5390-5401.
doi: 10.1039/D0BM01320J URL |
| 65. |
Jiang, L.; Gu, Y.; Du, Y.; Liu, J. Exosomes: diagnostic biomarkers and therapeutic delivery vehicles for cancer. Mol Pharm. 2019, 16, 3333-3349.
doi: 10.1021/acs.molpharmaceut.9b00409 URL |
| 66. |
Qi, H.; Liu, C.; Long, L.; Ren, Y.; Zhang, S.; Chang, X.; Qian, X.; Jia, H.; Zhao, J.; Sun, J.; Hou, X.; Yuan, X.; Kang, C. Blood exosomes endowed with magnetic and targeting properties for cancer therapy. ACS Nano. 2016, 10, 3323-3333.
doi: 10.1021/acsnano.5b06939 URL |
| 67. |
Jiang, L.; Gu, Y.; Du, Y.; Tang, X.; Wu, X.; Liu, J. Engineering exosomes endowed with targeted delivery of triptolide for malignant melanoma therapy. ACS Appl Mater Interfaces. 2021, 13, 42411-42428.
doi: 10.1021/acsami.1c10325 URL |
| 68. |
Zou, J.; Shi, M.; Liu, X.; Jin, C.; Xing, X.; Qiu, L.; Tan, W. Aptamer-functionalized exosomes: elucidating the cellular uptake mechanism and the potential for cancer-targeted chemotherapy. Anal Chem. 2019, 91, 2425-2430.
doi: 10.1021/acs.analchem.8b05204 URL |
| 69. | Zhang, M.; Hu, S.; Liu, L.; Dang, P.; Liu, Y.; Sun, Z.; Qiao, B.; Wang, C. Engineered exosomes from different sources for cancer-targeted therapy. Signal Transduct Target Ther. 2023, 8, 124. |
| 70. |
Kim, H.; Yun, N.; Mun, D.; Kang, J. Y.; Lee, S. H.; Park, H.; Park, H.; Joung, B. Cardiac-specific delivery by cardiac tissue-targeting peptide-expressing exosomes. Biochem Biophys Res Commun. 2018, 499, 803-808.
doi: 10.1016/j.bbrc.2018.03.227 URL |
| 71. |
Wang, J.; Liu, Y.; Liu, Y.; Huang, H.; Roy, S.; Song, Z.; Guo, B. Recent advances in nanomedicines for imaging and therapy of myocardial ischemia-reperfusion injury. J Control Release. 2023, 353, 563-590.
doi: 10.1016/j.jconrel.2022.11.057 URL |
| 72. | Wang, X.; Chen, Y.; Zhao, Z.; Meng, Q.; Yu, Y.; Sun, J.; Yang, Z.; Chen, Y.; Li, J.; Ma, T.; Liu, H.; Li, Z.; Yang, J.; Shen, Z. Engineered exosomes with ischemic myocardium-targeting peptide for targeted therapy in myocardial infarction. J Am Heart Assoc. 2018, 7, e008737. |
| 73. |
Sen, S.; Xavier, J.; Kumar, N.; Ahmad, M. Z.; Ranjan, O. P. Exosomes as natural nanocarrier-based drug delivery system: recent insights and future perspectives. 3 Biotech. 2023, 13, 101.
doi: 10.1007/s13205-023-03521-2 |
| 74. |
Alvarez-Erviti, L.; Seow, Y.; Yin, H.; Betts, C.; Lakhal, S.; Wood, M. J. Delivery of siRNA to the mouse brain by systemic injection of targeted exosomes. Nat Biotechnol. 2011, 29, 341-345.
doi: 10.1038/nbt.1807 |
| 75. |
Liu, H.; Zhang, H.; Wang, S.; Cui, J.; Weng, W.; Liu, X.; Tang, H.; Hu, Y.; Li, X.; Zhang, K.; Zhou, F.; Jing, Y.; Su, J. Bone-targeted bioengineered bacterial extracellular vesicles delivering siRNA to ameliorate osteoporosis. Compos B Eng. 2023, 255, 110610.
doi: 10.1016/j.compositesb.2023.110610 URL |
| 76. | Liu, H.; Zhang, Q.; Wang, S.; Weng, W.; Jing, Y.; Su, J. Bacterial extracellular vesicles as bioactive nanocarriers for drug delivery: Advances and perspectives. Bioact Mater. 2022, 14, 169-181. |
| 77. |
Meng, F.; Yin, Z.; Ren, X.; Geng, Z.; Su, J. Construction of local drug delivery system on titanium-based implants to improve osseointegration. Pharmaceutics. 2022, 14, 1069.
doi: 10.3390/pharmaceutics14051069 URL |
| 78. | Zhang, Y.; Li, G.; Wang, J.; Zhou, F.; Ren, X.; Su, J. Small joint organoids 3D bioprinting: construction strategy and application. Small. 2023, e2302506. |
| 79. |
Song, H.; Li, X.; Zhao, Z.; Qian, J.; Wang, Y.; Cui, J.; Weng, W.; Cao, L.; Chen, X.; Hu, Y.; Su, J. Reversal of osteoporotic activity by endothelial cell-secreted bone targeting and biocompatible exosomes. Nano Lett. 2019, 19, 3040-3048.
doi: 10.1021/acs.nanolett.9b00287 URL |
| 80. | Hu, Y.; Li, X.; Zhang, Q.; Gu, Z.; Luo, Y.; Guo, J.; Wang, X.; Jing, Y.; Chen, X.; Su, J. Exosome-guided bone targeted delivery of Antagomir-188 as an anabolic therapy for bone loss. Bioact Mater. 2021, 6, 2905-2913. |
| 81. | Cui, Y.; Guo, Y.; Kong, L.; Shi, J.; Liu, P.; Li, R.; Geng, Y.; Gao, W.; Zhang, Z.; Fu, D. A bone-targeted engineered exosome platform delivering siRNA to treat osteoporosis. Bioact Mater. 2022, 10, 207-221. |
| 82. |
Wang, Y.; Yao, J.; Cai, L.; Liu, T.; Wang, X.; Zhang, Y.; Zhou, Z.; Li, T.; Liu, M.; Lai, R.; Liu, X. Bone-targeted extracellular vesicles from mesenchymal stem cells for osteoporosis therapy. Int J Nanomedicine. 2020, 15, 7967-7977.
doi: 10.2147/IJN.S263756 URL |
| [1] | Han Liu, Jiacan Su. Organoid extracellular vesicle-based therapeutic strategies for bone therapy [J]. Biomaterials Translational, 2023, 4(4): 199-212. |
| [2] | Monchupa Kingsak, Thongpon Meethong, Jinnawat Jongkhumkrong, Li Cai, Qian Wang. Therapeutic potential of oncolytic viruses in the era of precision oncology [J]. Biomaterials Translational, 2023, 4(2): 67-84. |
| [3] | Zhao–Lin Zeng, Hui Xie. Mesenchymal stem cell–derived extracellular vesicles: a possible therapeutic strategy for orthopaedic diseases: a narrative review [J]. Biomaterials Translational, 2022, 3(3): 175-187. |
| [4] | Ronghua Tan, Ying Wan, Xiangliang Yang. Hydroxyethyl starch and its derivatives as nanocarriers for delivery of diagnostic and therapeutic agents towards cancers [J]. Biomaterials Translational, 2020, 1(1): 46-57. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||