Biomaterials Translational ›› 2021, Vol. 2 ›› Issue (1): 61-71.doi: 10.3877/cma.j.issn.2096-112X.2021.01.008
• REVIEW • Previous Articles Next Articles
Pingli Wu1, Yangyang Liang1, Guoming Sun2,*( )
)
Received:2020-07-14
Revised:2020-12-10
Accepted:2020-12-29
Online:2021-03-31
Published:2021-03-28
Contact:
Guoming Sun
E-mail:gsun@hbu.edu.cn
Wu, P.; Liang, Y.; Sun, G. Engineering immune-responsive biomaterials for skin regeneration. Biomater Transl. 2021, 2(1), 61-71.
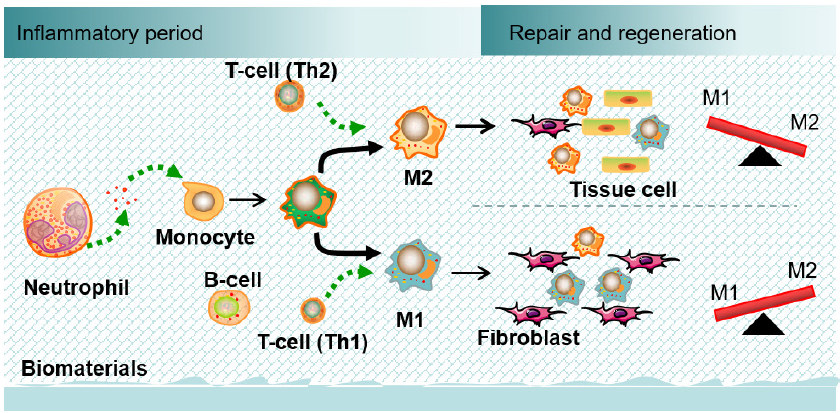
Figure 1. Temporal sequence of immune reactions to biomaterials. The main cells participate in the biomaterial-tissue microenvironment from the initial inflammatory response to tissue repair and regeneration. Biomaterials shape the immune environment by targeting neutrophils, lymphocytes (T-helper cells and B cells) and macrophages.
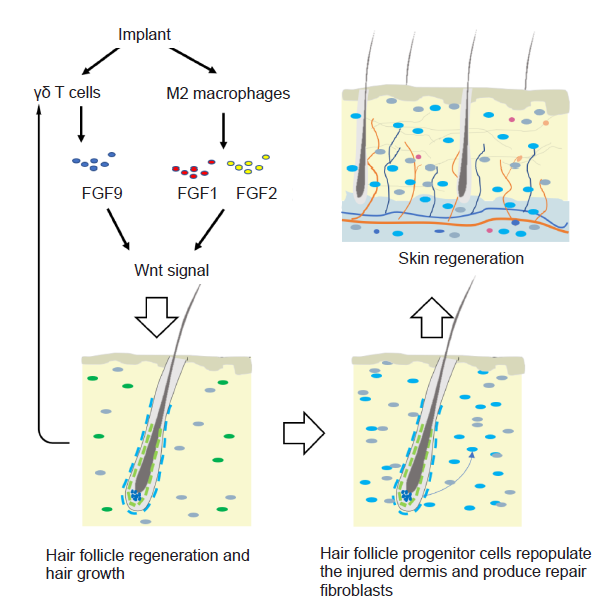
Figure 2. The effect of immune cells on hair growth, hair follicle regeneration, and skin regeneration. M2 macrophages secrete FGF2 and IGF1 that play roles in hair follicle neogenesis. The γδ T cells express FGF9, which activates Wnt signalling and further induces hair follicle regeneration and hair growth. Hair follicle progenitor cells repopulate the injured dermis, and produce repair fibroblasts. FGF: fibroblast growth factor; IGF1: insulin-like growth factor 1.
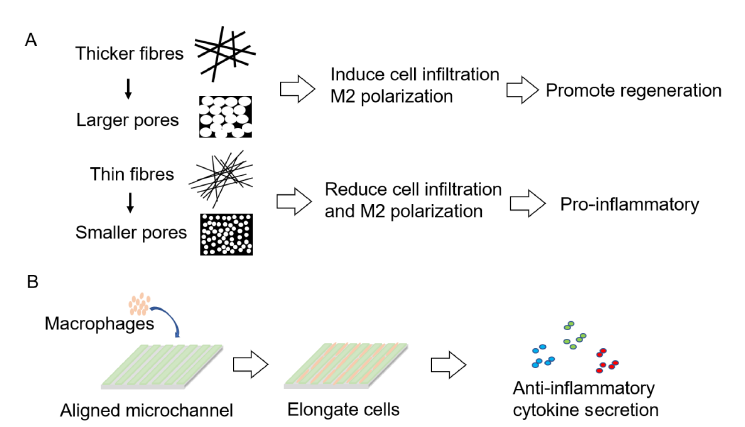
Figure 4. Physical strategies to engineer immunomodulatory biomaterial. (A) Schematic illustration showing that scaffolds with thicker fibres and larger pores promote the transformation of macrophages to the M2 phenotype. (B) Schematic illustration showing that microchannels cause cells to elongate, further facilitating M2 polarization.
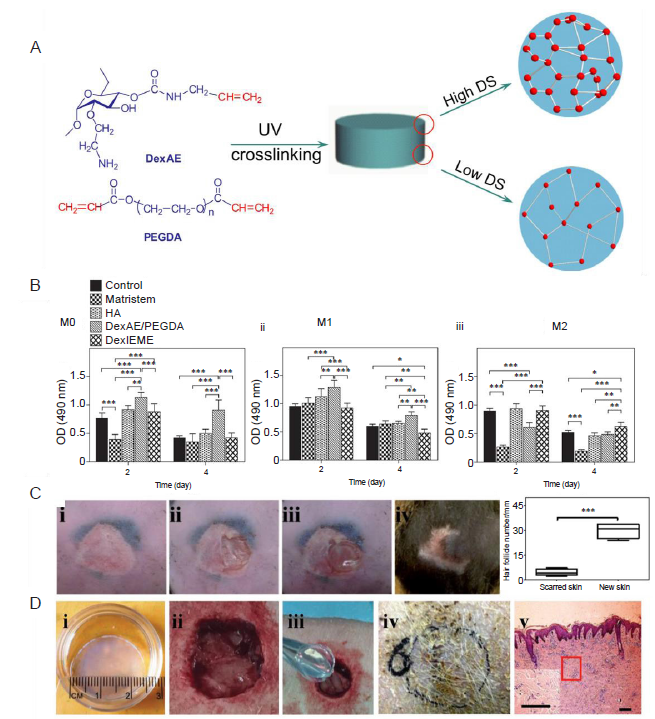
Figure 5. Integrating chemi-physical properties into biomaterials. (A) Increasing the DS of the crosslinkable functional group leads to a less porous structure. Reprinted from Sun et al.81 Copyright 2011, with permission from Elsevier. (B) Macromers affect macrophage differentiation and polarization; DexIEME promotes M2 phenotype transformation. Reprinted from Sun.80 Copyright Wiley-VCH Verlag GmbH & Co. KGaA. Reproduced with permission. (C) A pre-existing skin scar (i) that was partially promoted (ii) and treated with DexIEME hydrogel (iii) exhibited scarless skin healing (iv) with skin appendages (e.g., hair follicles). (D) A full-thickness skin injury (ii) in a preclinical swine model demonstrated that DexIEME (i, iii) regenerated complete skin (iv) structures (v) after 10 weeks. DexAE/PEGDA: dextran-allyl isocyanate-ethylamine and polyethylene glycol-diacrylate hydrogel; DexIEME: dextran-isocyanatoethyl methacrylate-ethylamine; DS: degree of substitution; HA: hyaluronic acid; OD: optical density; PEGDA: poly(ethylene glycol) diacrylate; UV: ultraviolet.
| 1. |
Schulz, J. T3rd; Tompkins, R. G.; Burke, J. F. Artificial skin. Annu Rev Med. 2000, 51, 231-244.
doi: 10.1146/annurev.med.51.1.231 URL pmid: 10774462 |
| 2. | Sun, G.; Mao, J. J. Engineering dextran-based scaffolds for drug delivery and tissue repair. Nanomedicine (Lond). 2012, 7, 1771-1784. |
| 3. |
Dalgard, F. J.; Gieler, U.; Tomas-Aragones, L.; Lien, L.; Poot, F.; Jemec, G. B. E.; Misery, L.; Szabo, C.; Linder, D.; Sampogna, F.; Evers, A. W. M.; Halvorsen, J. A.; Balieva, F.; Szepietowski, J.; Romanov, D.; Marron, S. E.; Altunay, I. K.; Finlay, A. Y.; Salek, S. S.; Kupfer, J. The psychological burden of skin diseases: a cross-sectional multicenter study among dermatological out-patients in 13 European countries. J Invest Dermatol. 2015, 135, 984-991.
doi: 10.1038/jid.2014.530 URL pmid: 25521458 |
| 4. |
Chua, A. W.; Khoo, Y. C.; Tan, B. K.; Tan, K. C.; Foo, C. L.; Chong, S. J. Skin tissue engineering advances in severe burns: review and therapeutic applications. Burns Trauma. 2016, 4, 3.
doi: 10.1186/s41038-016-0027-y URL pmid: 27574673 |
| 5. |
Balieva, F.; Kupfer, J.; Lien, L.; Gieler, U.; Finlay, A. Y.; Tomás-Aragonés, L.; Poot, F.; Misery, L.; Sampogna, F.; van Middendorp, H.; Halvorsen, J. A.; Szepietowski, J. C.; Lvov, A.; Marrón, S. E.; Salek, M. S.; Dalgard, F. J. The burden of common skin diseases assessed with the EQ5DTM: a European multicentre study in 13 countries . Br J Dermatol. 2017, 176, 1170-1178.
doi: 10.1111/bjd.15280 URL pmid: 28032340 |
| 6. |
Sun, B. K.; Siprashvili, Z.; Khavari, P. A. Advances in skin grafting and treatment of cutaneous wounds. Science. 2014, 346, 941-945.
doi: 10.1126/science.1253836 URL pmid: 25414301 |
| 7. |
Hall, A. H.; Mathieu, L.; Maibach, H. I. Acute chemical skin injuries in the United States: a review. Crit Rev Toxicol. 2018, 48, 540-554.
doi: 10.1080/10408444.2018.1493085 URL pmid: 30226392 |
| 8. |
Reinke, J. M.; Sorg, H. Wound repair and regeneration. Eur Surg Res. 2012, 49, 35-43.
URL pmid: 22797712 |
| 9. | Rippa, A. L.; Kalabusheva, E. P.; Vorotelyak, E. A. Regeneration of dermis: Scarring and cells involved. Cells. 2019, 8, 607. |
| 10. |
Weng, T.; Wu, P.; Zhang, W.; Zheng, Y.; Li, Q.; Jin, R.; Chen, H.; You, C.; Guo, S.; Han, C.; Wang, X. Regeneration of skin appendages and nerves: current status and further challenges. J Transl Med. 2020, 18, 53.
doi: 10.1186/s12967-020-02248-5 URL pmid: 32014004 |
| 11. |
Andorko, J. I.; Jewell, C. M. Designing biomaterials with immunomodulatory properties for tissue engineering and regenerative medicine. Bioeng Transl Med. 2017, 2, 139-155.
doi: 10.1002/btm2.10063 URL pmid: 28932817 |
| 12. |
Chung, L.; Maestas, D. R. Jr.; Housseau, F.; Elisseeff, J. H. Key players in the immune response to biomaterial scaffolds for regenerative medicine. Adv Drug Deliv Rev. 2017, 114, 184-192.
doi: 10.1016/j.addr.2017.07.006 URL pmid: 28712923 |
| 13. |
Christman, K. L. Biomaterials for tissue repair. Science. 2019, 363, 340-341.
doi: 10.1126/science.aar2955 URL pmid: 30679357 |
| 14. |
Kim, M. H.; Liu, W.; Borjesson, D. L.; Curry, F. R.; Miller, L. S.; Cheung, A. L.; Liu, F. T.; Isseroff, R. R.; Simon, S. I. Dynamics of neutrophil infiltration during cutaneous wound healing and infection using fluorescence imaging. J Invest Dermatol. 2008, 128, 1812-1820.
doi: 10.1038/sj.jid.5701223 URL pmid: 18185533 |
| 15. |
Rodero, M. P.; Khosrotehrani, K. Skin wound healing modulation by macrophages. Int J Clin Exp Pathol. 2010, 3, 643-653.
URL pmid: 20830235 |
| 16. |
Brancato, S. K.; Albina, J. E. Wound macrophages as key regulators of repair: origin, phenotype, and function. Am J Pathol. 2011, 178, 19-25.
doi: 10.1016/j.ajpath.2010.08.003 URL pmid: 21224038 |
| 17. | Sun, G.; Owens, D.; Mao, J. Scarless skin regeneration-are we there yet? JSM Regen Med Bio Eng. 2013, 1, 1007. |
| 18. |
Gurtner, G. C.; Werner, S.; Barrandon, Y.; Longaker, M. T. Wound repair and regeneration. Nature. 2008, 453, 314-321.
doi: 10.1038/nature07039 URL pmid: 18480812 |
| 19. |
Eming, S. A.; Hammerschmidt, M.; Krieg, T.; Roers, A. Interrelation of immunity and tissue repair or regeneration. Semin Cell Dev Biol. 2009, 20, 517-527.
doi: 10.1016/j.semcdb.2009.04.009 URL pmid: 19393325 |
| 20. |
Vagnozzi, R. J.; Maillet, M.; Sargent, M. A.; Khalil, H.; Johansen, A. K. Z.; Schwanekamp, J. A.; York, A. J.; Huang, V.; Nahrendorf, M.; Sadayappan, S.; Molkentin, J. D. An acute immune response underlies the benefit of cardiac stem cell therapy. Nature. 2020, 577, 405-409.
doi: 10.1038/s41586-019-1802-2 URL pmid: 31775156 |
| 21. |
Sadtler, K.; Estrellas, K.; Allen, B. W.; Wolf, M. T.; Fan, H.; Tam, A. J.; Patel, C. H.; Luber, B. S.; Wang, H.; Wagner, K. R.; Powell, J. D.; Housseau, F.; Pardoll, D. M.; Elisseeff, J. H. Developing a pro-regenerative biomaterial scaffold microenvironment requires T helper 2 cells. Science. 2016, 352, 366-370.
doi: 10.1126/science.aad9272 URL pmid: 27081073 |
| 22. |
Matzinger, P.; Kamala, T. Tissue-based class control: the other side of tolerance. Nat Rev Immunol. 2011, 11, 221-230.
doi: 10.1038/nri2940 URL pmid: 21350581 |
| 23. |
Moore, L. B.; Kyriakides, T. R. Molecular characterization of macrophage-biomaterial interactions. Adv Exp Med Biol. 2015, 865, 109-122.
doi: 10.1007/978-3-319-18603-0_7 URL pmid: 26306446 |
| 24. |
Eming, S. A.; Krieg, T.; Davidson, J. M. Inflammation in wound repair: molecular and cellular mechanisms. J Invest Dermatol. 2007, 127, 514-525.
doi: 10.1038/sj.jid.5700701 URL pmid: 17299434 |
| 25. |
Murray, P. J.; Wynn, T. A. Protective and pathogenic functions of macrophage subsets. Nat Rev Immunol. 2011, 11, 723-737.
doi: 10.1038/nri3073 URL pmid: 21997792 |
| 26. |
Willenborg, S.; Eming, S. A. Macrophages - sensors and effectors coordinating skin damage and repair. J Dtsch Dermatol Ges. 2014, 12, 214-221.
doi: 10.1111/ddg.12290 URL pmid: 24580874 |
| 27. |
Garg, K.; Pullen, N. A.; Oskeritzian, C. A.; Ryan, J. J.; Bowlin, G. L. Macrophage functional polarization (M1/M2) in response to varying fiber and pore dimensions of electrospun scaffolds. Biomaterials. 2013, 34, 4439-4451.
doi: 10.1016/j.biomaterials.2013.02.065 URL pmid: 23515178 |
| 28. |
Kreimendahl, F.; Marquardt, Y.; Apel, C.; Bartneck, M.; Zwadlo-Klarwasser, G.; Hepp, J.; Jockenhoevel, S.; Baron, J. M. Macrophages significantly enhance wound healing in a vascularized skin model. J Biomed Mater Res A. 2019, 107, 1340-1350.
doi: 10.1002/jbm.a.36648 URL pmid: 30724457 |
| 29. |
Mahdavian Delavary B.; van der Veer, W. M.; van Egmond, M.; Niessen, F. B.; Beelen, R. H. Macrophages in skin injury and repair. Immunobiology. 2011, 216, 753-762.
doi: 10.1016/j.imbio.2011.01.001 URL pmid: 21281986 |
| 30. |
Vishwakarma, A.; Bhise, N. S.; Evangelista, M. B.; Rouwkema, J.; Dokmeci, M. R.; Ghaemmaghami, A. M.; Vrana, N. E.; Khademhosseini, A. Engineering immunomodulatory biomaterials to tune the inflammatory response. Trends Biotechnol. 2016, 34, 470-482.
doi: 10.1016/j.tibtech.2016.03.009 URL pmid: 27138899 |
| 31. |
Lucas, T.; Waisman, A.; Ranjan, R.; Roes, J.; Krieg, T.; Müller, W.; Roers, A.; Eming, S. A. Differential roles of macrophages in diverse phases of skin repair. J Immunol. 2010, 184, 3964-3977.
doi: 10.4049/jimmunol.0903356 URL pmid: 20176743 |
| 32. |
Bouaziz, J. D.; Yanaba, K.; Tedder, T. F. Regulatory B cells as inhibitors of immune responses and inflammation. Immunol Rev. 2008, 224, 201-214.
doi: 10.1111/j.1600-065X.2008.00661.x URL pmid: 18759928 |
| 33. |
Groth, T.; Altankov, G.; Klosz, K. Adhesion of human peripheral blood lymphocytes is dependent on surface wettability and protein preadsorption. Biomaterials. 1994, 15, 423-428.
doi: 10.1016/0142-9612(94)90220-8 URL pmid: 8080932 |
| 34. |
Rodriguez, A.; Anderson, J. M. Evaluation of clinical biomaterial surface effects on T lymphocyte activation. J Biomed Mater Res A. 2010, 92, 214-220.
doi: 10.1002/jbm.a.32362 URL pmid: 19172618 |
| 35. |
Jameson, J.; Ugarte, K.; Chen, N.; Yachi, P.; Fuchs, E.; Boismenu, R.; Havran, W. L. A role for skin gammadelta T cells in wound repair. Science. 2002, 296, 747-749.
URL pmid: 11976459 |
| 36. |
Steinman, R. M. Dendritic cells and the control of immunity: enhancing the efficiency of antigen presentation. Mt Sinai J Med. 2001, 68, 160-166.
URL pmid: 11373688 |
| 37. |
Rani, M.; Schwacha, M. G. The composition of T-cell subsets are altered in the burn wound early after injury. PLoS One. 2017, 12, e0179015.
doi: 10.1371/journal.pone.0179015 URL pmid: 28575063 |
| 38. |
Qi, C.; Xu, L.; Deng, Y.; Wang, G.; Wang, Z.; Wang, L. Sericin hydrogels promote skin wound healing with effective regeneration of hair follicles and sebaceous glands after complete loss of epidermis and dermis. Biomater Sci. 2018, 6, 2859-2870.
doi: 10.1039/c8bm00934a URL pmid: 30259043 |
| 39. |
Rahmani, W.; Liu, Y.; Rosin, N. L.; Kline, A.; Raharjo, E.; Yoon, J.; Stratton, J. A.; Sinha, S.; Biernaskie, J. Macrophages promote wound-induced hair follicle regeneration in a CX(3)CR1- and TGF-β1-dependent manner. J Invest Dermatol. 2018, 138, 2111-2122.
doi: 10.1016/j.jid.2018.04.010 URL pmid: 29705291 |
| 40. |
Kasuya, A.; Ito, T.; Tokura, Y. M2 macrophages promote wound-induced hair neogenesis. J Dermatol Sci. 2018, 91, 250-255.
doi: 10.1016/j.jdermsci.2018.05.004 URL pmid: 29776717 |
| 41. |
Gay, D.; Kwon, O.; Zhang, Z.; Spata, M.; Plikus, M. V.; Holler, P. D.; Ito, M.; Yang, Z.; Treffeisen, E.; Kim, C. D.; Nace, A.; Zhang, X.; Baratono, S.; Wang, F.; Ornitz, D. M.; Millar, S. E.; Cotsarelis, G. Fgf9 from dermal γδ T cells induces hair follicle neogenesis after wounding. Nat Med. 2013, 19, 916-923.
doi: 10.1038/nm.3181 URL pmid: 23727932 |
| 42. |
Havran, W. L.; Jameson, J. M. Epidermal T cells and wound healing. J Immunol. 2010, 184, 5423-5428.
doi: 10.4049/jimmunol.0902733 URL pmid: 20483798 |
| 43. |
Veltri, A.; Lang, C.; Lien, W. H. Concise review: Wnt signaling pathways in skin development and epidermal stem cells. Stem Cells. 2018, 36, 22-35.
doi: 10.1002/stem.2723 URL pmid: 29047191 |
| 44. |
Newick, K.; Moon, E.; Albelda, S. M. Chimeric antigen receptor T-cell therapy for solid tumors. Mol Ther Oncolytics. 2016, 3, 16006.
doi: 10.1038/mto.2016.6 URL pmid: 27162934 |
| 45. |
Shin, J. U.; Abaci, H. E.; Herron, L.; Guo, Z.; Sallee, B.; Pappalardo, A.; Jackow, J.; Wang, E. H. C.; Doucet, Y.; Christiano, A. M. Recapitulating T cell infiltration in 3D psoriatic skin models for patient-specific drug testing. Sci Rep. 2020, 10, 4123.
doi: 10.1038/s41598-020-60275-0 URL pmid: 32139717 |
| 46. |
Jahoda, C. A.; Reynolds, A. J. Hair follicle dermal sheath cells: unsung participants in wound healing. Lancet. 2001, 358, 1445-1448.
doi: 10.1016/S0140-6736(01)06532-1 URL pmid: 11705511 |
| 47. | Abbasi, S.; Sinha, S.; Labit, E.; Rosin, N. L.; Yoon, G.; Rahmani, W.; Jaffer, A.; Sharma, N.; Hagner, A.; Shah, P.; Arora, R.; Yoon, J.; Islam, A.; Uchida, A.; Chang, C. K.; Stratton, J. A.; Scott, R. W.; Rossi, F. M. V.; Underhill, T. M.; Biernaskie, J.. Distinct regulatory programs control the latent regenerative potential of dermal fibroblasts during wound healing. Cell Stem Cell. 2020, 27, 396-412.e6. |
| 48. |
Graney, P. L.; Ben-Shaul, S.; Landau, S.; Bajpai, A.; Singh, B.; Eager, J.; Cohen, A.; Levenberg, S.; Spiller, K. L. Macrophages of diverse phenotypes drive vascularization of engineered tissues. Sci Adv. 2020, 6, eaay6391.
doi: 10.1126/sciadv.aay6391 URL pmid: 32494664 |
| 49. |
Blais, M.;.; Parenteau-Bareil, R.; Cadau, S.; Berthod, F. Concise review: tissue-engineered skin and nerve regeneration in burn treatment. Stem Cells Transl Med. 2013, 2, 545-551.
URL pmid: 23734060 |
| 50. |
Gemici, B.; Elsheikh, W.; Feitosa, K. B.; Costa, S. K.; Muscara, M. N.; Wallace, J. L. H2S-releasing drugs: anti-inflammatory, cytoprotective and chemopreventative potential. Nitric Oxide. 2015, 46, 25-31.
doi: 10.1016/j.niox.2014.11.010 URL pmid: 25461269 |
| 51. |
Wallace, J. L.; Blackler, R. W.; Chan, M. V.; Da Silva, G. J.; Elsheikh, W.; Flannigan, K. L.; Gamaniek, I.; Manko, A.; Wang, L.; Motta, J. P.; Buret, A. G. Anti-inflammatory and cytoprotective actions of hydrogen sulfide: translation to therapeutics. Antioxid Redox Signal. 2015, 22, 398-410.
doi: 10.1089/ars.2014.5901 URL pmid: 24635322 |
| 52. |
Wu, J.; Chen, A.; Zhou, Y.; Zheng, S.; Yang, Y.; An, Y.; Xu, K.; He, H.; Kang, J.; Luckanagul, J. A.; Xian, M.; Xiao, J.; Wang, Q. Novel H(2)S-Releasing hydrogel for wound repair via in situ polarization of M2 macrophages. Biomaterials. 2019, 222, 119398.
doi: 10.1016/j.biomaterials.2019.119398 URL pmid: 31487582 |
| 53. |
Crossley, G. H.; Brinker, J. A.; Reynolds, D.; Spencer, W.; Johnson, W. B.; Hurd, H.; Tonder, L.; Zmijewski, M. Steroid elution improves the stimulation threshold in an active-fixation atrial permanent pacing lead. A randomized, controlled study. Model 4068 Investigators. Circulation. 1995, 92, 2935-2939.
doi: 10.1161/01.cir.92.10.2935 URL pmid: 7586262 |
| 54. |
Udipi, K.; Ornberg, R. L.; Thurmond, K. B., 2nd; Settle, S. L.; Forster, D.; Riley, D. Modification of inflammatory response to implanted biomedical materials in vivo by surface bound superoxide dismutase mimics. J Biomed Mater Res. 2000, 51, 549-560.
doi: 10.1002/1097-4636(20000915)51:4<549::aid-jbm2>3.0.co;2-z URL pmid: 10880102 |
| 55. |
Zhong, Y.; Bellamkonda, R. V. Dexamethasone-coated neural probes elicit attenuated inflammatory response and neuronal loss compared to uncoated neural probes. Brain Res. 2007, 1148, 15-27.
doi: 10.1016/j.brainres.2007.02.024 URL pmid: 17376408 |
| 56. | Mercanzini, A.; Reddy, S. T.; Velluto, D.; Colin, P.; Maillard, A.; Bensadoun, J. C.; Hubbell, J. A.; Renaud, P. Controlled release nanoparticle-embedded coatings reduce the tissue reaction to neuroprostheses. J Control Release. 2010, 145, 196-202. |
| 57. |
Kim, D. H.; Martin, D. C. Sustained release of dexamethasone from hydrophilic matrices using PLGA nanoparticles for neural drug delivery. Biomaterials. 2006, 27, 3031-3037.
doi: 10.1016/j.biomaterials.2005.12.021 URL pmid: 16443270 |
| 58. |
Zlotnik, A.; Yoshie, O. The chemokine superfamily revisited. Immunity. 2012, 36, 705-716.
doi: 10.1016/j.immuni.2012.05.008 URL pmid: 22633458 |
| 59. | Boehler, R. M.; Graham, J. G.; Shea, L. D. Tissue engineering tools for modulation of the immune response. BioTechniques. 2011, 51, 239-240. |
| 60. | Hume, P. S.; He, J.; Haskins, K.; Anseth, K. S. Strategies to reduce dendritic cell activation through functional biomaterial design. Biomaterials. 2012, 33, 3615-3625. |
| 61. |
Johnston, C. J.; Smyth, D. J.; Dresser, D. W.; Maizels, R. M. TGF-β in tolerance, development and regulation of immunity. Cell Immunol. 2016, 299, 14-22.
doi: 10.1016/j.cellimm.2015.10.006 URL pmid: 26617281 |
| 62. |
Morris, A. H.; Chang, J.; Kyriakides, T. R. Inadequate processing of decellularized dermal matrix reduces cell viability in vitro and increases apoptosis and acute inflammation in vivo. Biores Open Access. 2016, 5, 177-187.
doi: 10.1089/biores.2016.0021 URL pmid: 27500014 |
| 63. |
Keane, T. J.; Londono, R.; Turner, N. J.; Badylak, S. F. Consequences of ineffective decellularization of biologic scaffolds on the host response. Biomaterials. 2012, 33, 1771-1781.
doi: 10.1016/j.biomaterials.2011.10.054 URL pmid: 22137126 |
| 64. |
Keane, T. J.; Swinehart, I. T.; Badylak, S. F. Methods of tissue decellularization used for preparation of biologic scaffolds and in vivo relevance. Methods. 2015, 84, 25-34.
doi: 10.1016/j.ymeth.2015.03.005 URL pmid: 25791470 |
| 65. |
Badylak, S. F.; Valentin, J. E.; Ravindra, A. K.; McCabe, G. P.; Stewart-Akers, A. M. Macrophage phenotype as a determinant of biologic scaffold remodeling. Tissue Eng Part A. 2008, 14, 1835-1842.
URL pmid: 18950271 |
| 66. |
van der Smissen, A.; Hintze, V.; Scharnweber, D.; Moeller, S.; Schnabelrauch, M.; Majok, A.; Simon, J. C.; Anderegg, U. Growth promoting substrates for human dermal fibroblasts provided by artificial extracellular matrices composed of collagen I and sulfated glycosaminoglycans. Biomaterials. 2011, 32, 8938-8946.
doi: 10.1016/j.biomaterials.2011.08.025 URL pmid: 21875749 |
| 67. |
Kajahn, J.; Franz, S.; Rueckert, E.; Forstreuter, I.; Hintze, V.; Moeller, S.; Simon, J. C. Artificial extracellular matrices composed of collagen I and high sulfated hyaluronan modulate monocyte to macrophage differentiation under conditions of sterile inflammation. Biomatter. 2012, 2, 226-236.
doi: 10.4161/biom.22855 URL pmid: 23507888 |
| 68. |
Wolf, M. T.; Dearth, C. L.; Ranallo, C. A.; LoPresti, S. T.; Carey, L. E.; Daly, K. A.; Brown, B. N.; Badylak, S. F. Macrophage polarization in response to ECM coated polypropylene mesh. Biomaterials. 2014, 35, 6838-6849.
URL pmid: 24856104 |
| 69. |
Huleihel, L.; Hussey, G. S.; Naranjo, J. D.; Zhang, L.; Dziki, J. L.; Turner, N. J.; Stolz, D. B.; Badylak, S. F. Matrix-bound nanovesicles within ECM bioscaffolds. Sci Adv. 2016, 2, e1600502.
doi: 10.1126/sciadv.1600502 URL pmid: 27386584 |
| 70. |
Sridharan, R.; Cavanagh, B.; Cameron, A. R.; Kelly, D. J.; O’Brien, F. J. Material stiffness influences the polarization state, function and migration mode of macrophages. Acta Biomater. 2019, 89, 47-59.
URL pmid: 30826478 |
| 71. |
Okamoto, T.; Takagi, Y.; Kawamoto, E.; Park, E. J.; Usuda, H.; Wada, K.; Shimaoka, M. Reduced substrate stiffness promotes M2-like macrophage activation and enhances peroxisome proliferator-activated receptor γ expression. Exp Cell Res. 2018, 367, 264-273.
doi: 10.1016/j.yexcr.2018.04.005 URL pmid: 29627321 |
| 72. |
Jiang, S.; Lyu, C.; Zhao, P.; Li, W.; Kong, W.; Huang, C.; Genin, G. M.; Du, Y. Cryoprotectant enables structural control of porous scaffolds for exploration of cellular mechano-responsiveness in 3D. Nat Commun. 2019, 10, 3491.
doi: 10.1038/s41467-019-11397-1 URL pmid: 31375674 |
| 73. |
Wang, Z.; Cui, Y.; Wang, J.; Yang, X.; Wu, Y.; Wang, K.; Gao, X.; Li, D.; Li, Y.; Zheng, X. L.; Zhu, Y.; Kong, D.; Zhao, Q. The effect of thick fibers and large pores of electrospun poly(ε-caprolactone) vascular grafts on macrophage polarization and arterial regeneration. Biomaterials. 2014, 35, 5700-5710.
doi: 10.1016/j.biomaterials.2014.03.078 URL pmid: 24746961 |
| 74. | Sridharan, R.; Cameron, A. R.; Kelly, D. J.; Kearney, C. J.; O’Brien, F. J. Biomaterial based modulation of macrophage polarization: a review and suggested design principles. Mater Today. 2015, 18, 313-325. |
| 75. |
McWhorter, F. Y.; Wang, T.; Nguyen, P.; Chung, T.; Liu, W. F. Modulation of macrophage phenotype by cell shape. Proc Natl Acad Sci U S A. 2013, 110, 17253-17258.
doi: 10.1073/pnas.1308887110 URL pmid: 24101477 |
| 76. |
Li, W.; Wu, P.; Zhang, Y.; Midgley, A. C.; Yuan, X.; Wu, Y.; Wang, L.; Wang, Z.; Zhu, M.; Kong, D. Bilayered polymeric micro- and nanofiber vascular grafts as abdominal aorta replacements: Long-term in vivo studies in a rat model. ACS Appl Bio Mater. 2019, 2, 4493-4502.
doi: 10.1021/acsabm.9b00641 URL |
| 77. |
Wu, P.; Wang, L.; Li, W.; Zhang, Y.; Wu, Y.; Zhi, D.; Wang, H.; Wang, L.; Kong, D.; Zhu, M. Construction of vascular graft with circumferentially oriented microchannels for improving artery regeneration. Biomaterials. 2020, 242, 119922.
doi: 10.1016/j.biomaterials.2020.119922 URL pmid: 32155476 |
| 78. |
Zhu, M.; Wu, Y.; Li, W.; Dong, X.; Chang, H.; Wang, K.; Wu, P.; Zhang, J.; Fan, G.; Wang, L.; Liu, J.; Wang, H.; Kong, D. Biodegradable and elastomeric vascular grafts enable vascular remodeling. Biomaterials. 2018, 183, 306-318.
doi: 10.1016/j.biomaterials.2018.08.063 URL pmid: 30189358 |
| 79. |
Blankenbaker, D. G.; Ullrick, S. R.; Davis, K. W.; De Smet, A. A.; Haaland, B.; Fine, J. P. Correlation of MRI findings with clinical findings of trochanteric pain syndrome. Skeletal Radiol. 2008, 37, 903-909.
doi: 10.1007/s00256-008-0514-8 URL pmid: 18566811 |
| 80. |
Sun, G. Pro-regenerative hydrogel restores scarless skin during cutaneous wound healing. Adv Healthc Mater. 2017, 6.
doi: 10.1002/adhm.201700954 URL pmid: 28975768 |
| 81. |
Sun, G.; Shen, Y. I.; Kusuma, S.; Fox-Talbot, K.; Steenbergen, C. J.; Gerecht, S. Functional neovascularization of biodegradable dextran hydrogels with multiple angiogenic growth factors. Biomaterials. 2011, 32, 95-106.
doi: 10.1016/j.biomaterials.2010.08.091 URL pmid: 20870284 |
| 82. |
Sun, G.; Zhang, X.; Shen, Y. I.; Sebastian, R.; Dickinson, L. E.; Fox-Talbot, K.; Reinblatt, M.; Steenbergen, C.; Harmon, J. W.; Gerecht, S. Dextran hydrogel scaffolds enhance angiogenic responses and promote complete skin regeneration during burn wound healing. Proc Natl Acad Sci U S A. 2011, 108, 20976-20981.
URL pmid: 22171002 |
| 83. |
Jackson, W. M.; Nesti, L. J.; Tuan, R. S. Mesenchymal stem cell therapy for attenuation of scar formation during wound healing. Stem Cell Res Ther. 2012, 3, 20.
doi: 10.1186/scrt111 URL pmid: 22668751 |
| 84. |
Rahimnejad, M.; Derakhshanfar, S.; Zhong, W. Biomaterials and tissue engineering for scar management in wound care. Burns Trauma. 2017, 5, 4.
doi: 10.1186/s41038-017-0069-9 URL pmid: 28127573 |
| 85. | Bertozzi, N.; Simonacci, F.; Grieco, M. P.; Grignaffini, E.; Raposio, E. The biological and clinical basis for the use of adipose-derived stem cells in the field of wound healing. Ann Med Surg (Lond). 2017, 20, 41-48. |
| 86. |
Maggini, J.; Mirkin, G.; Bognanni, I.; Holmberg, J.; Piazzón, I. M.; Nepomnaschy, I.; Costa, H.; Cañones, C.; Raiden, S.; Vermeulen, M.; Geffner, J. R. Mouse bone marrow-derived mesenchymal stromal cells turn activated macrophages into a regulatory-like profile. PLoS One. 2010, 5, e9252.
doi: 10.1371/journal.pone.0009252 URL pmid: 20169081 |
| 87. |
Scharp, D. W.; Marchetti, P. Encapsulated islets for diabetes therapy: history, current progress, and critical issues requiring solution. Adv Drug Deliv Rev. 2014, 67-68, 35-73.
doi: 10.1016/j.addr.2013.07.018 URL pmid: 23916992 |
| 88. | Kanki-Horimoto, S.; Horimoto, H.; Mieno, S.; Kishida, K.; Watanabe, F.; Furuya, E.; Katsumata, T. Synthetic vascular prosthesis impregnated with mesenchymal stem cells overexpressing endothelial nitric oxide synthase. Circulation. 2006, 114, I327-330. |
| 89. |
Merino-González, C.; Zuñiga, F. A.; Escudero, C.; Ormazabal, V.; Reyes, C.; Nova-Lamperti, E.; Salomón, C.; Aguayo, C. Mesenchymal stem cell-derived extracellular vesicles promote angiogenesis: Potencial clinical application. Front Physiol. 2016, 7, 24.
doi: 10.3389/fphys.2016.00024 URL pmid: 26903875 |
| 90. |
Noishiki, Y.; Tomizawa, Y.; Yamane, Y.; Matsumoto, A. Autocrine angiogenic vascular prosthesis with bone marrow transplantation. Nat Med. 1996, 2, 90-93.
doi: 10.1038/nm0196-90 URL pmid: 8564850 |
| 91. |
Liang, X.; Ding, Y.; Zhang, Y.; Tse, H. F.; Lian, Q. Paracrine mechanisms of mesenchymal stem cell-based therapy: current status and perspectives. Cell Transplant. 2014, 23, 1045-1059.
doi: 10.3727/096368913X667709 URL pmid: 23676629 |
| 92. |
Swartzlander, M. D.; Blakney, A. K.; Amer, L. D.; Hankenson, K. D.; Kyriakides, T. R.; Bryant, S. J. Immunomodulation by mesenchymal stem cells combats the foreign body response to cell-laden synthetic hydrogels. Biomaterials. 2015, 41, 79-88.
doi: 10.1016/j.biomaterials.2014.11.020 URL pmid: 25522967 |
| 93. |
Kim, J.; Hematti, P. Mesenchymal stem cell-educated macrophages: a novel type of alternatively activated macrophages. Exp Hematol. 2009, 37, 1445-1453.
doi: 10.1016/j.exphem.2009.09.004 URL pmid: 19772890 |
| 94. |
Dayan, V.; Yannarelli, G.; Billia, F.; Filomeno, P.; Wang, X. H.; Davies, J. E.; Keating, A. Mesenchymal stromal cells mediate a switch to alternatively activated monocytes/macrophages after acute myocardial infarction. Basic Res Cardiol. 2011, 106, 1299-1310.
URL pmid: 21901289 |
| 95. |
Adutler-Lieber, S.; Ben-Mordechai, T.; Naftali-Shani, N.; Asher, E.; Loberman, D.; Raanani, E.; Leor, J. Human macrophage regulation via interaction with cardiac adipose tissue-derived mesenchymal stromal cells. J Cardiovasc Pharmacol Ther. 2013, 18, 78-86.
doi: 10.1177/1074248412453875 URL pmid: 22894882 |
| 96. |
Nambu, M.; Kishimoto, S.; Nakamura, S.; Mizuno, H.; Yanagibayashi, S.; Yamamoto, N.; Azuma, R.; Nakamura, S.; Kiyosawa, T.; Ishihara, M.; Kanatani, Y. Accelerated wound healing in healing-impaired db/db mice by autologous adipose tissue-derived stromal cells combined with atelocollagen matrix. Ann Plast Surg. 2009, 62, 317-321.
doi: 10.1097/SAP.0b013e31817f01b6 URL pmid: 19240532 |
| 97. |
Park, B. S.; Jang, K. A.; Sung, J. H.; Park, J. S.; Kwon, Y. H.; Kim, K. J.; Kim, W. S. Adipose-derived stem cells and their secretory factors as a promising therapy for skin aging. Dermatol Surg. 2008, 34, 1323-1326.
doi: 10.1111/j.1524-4725.2008.34283.x URL pmid: 18616537 |
| 98. | Hassan, W. U.; Greiser, U.; Wang, W. Role of adipose-derived stem cells in wound healing. Wound Repair Regen. 2014, 22, 313-325. |
| 99. | Fraser, J. K.; Wulur, I.; Alfonso, Z.; Hedrick, M. H. Fat tissue: an underappreciated source of stem cells for biotechnology. Trends Biotechnol. 2006, 24, 150-154. |
| 100. |
Hass, R.; Kasper, C.; Böhm, S.; Jacobs, R. Different populations and sources of human mesenchymal stem cells (MSC): A comparison of adult and neonatal tissue-derived MSC. Cell Commun Signal. 2011, 9, 12.
doi: 10.1186/1478-811X-9-12 URL pmid: 21569606 |
| 101. |
Yoshimura, K.; Suga, H.; Eto, H. Adipose-derived stem/progenitor cells: roles in adipose tissue remodeling and potential use for soft tissue augmentation. Regen Med. 2009, 4, 265-273.
URL pmid: 19317645 |
| 102. | Phinney, D. G.; Pittenger, M. F. Concise review: MSC-derived exosomes for cell-free therapy. Stem Cells. 2017, 35, 851-858. |
| 103. | Kourembanas, S. Exosomes: vehicles of intercellular signaling, biomarkers, and vectors of cell therapy. Annu Rev Physiol. 2015, 77, 13-27. |
| 104. | Li, T.; Yan, Y.; Wang, B.; Qian, H.; Zhang, X.; Shen, L.; Wang, M.; Zhou, Y.; Zhu, W.; Li, W.; Xu, W. Exosomes derived from human umbilical cord mesenchymal stem cells alleviate liver fibrosis. Stem Cells Dev. 2013, 22, 845-854. |
| 105. | Hyun, J.; Wang, S.; Kim, J.; Kim, G. J.; Jung, Y. MicroRNA125b-mediated Hedgehog signaling influences liver regeneration by chorionic plate-derived mesenchymal stem cells. Sci Rep. 2015, 5, 14135. |
| 106. |
Wei, Y.; Wu, Y.; Zhao, R.; Zhang, K.; Midgley, A. C.; Kong, D.; Li, Z.; Zhao, Q. MSC-derived sEVs enhance patency and inhibit calcification of synthetic vascular grafts by immunomodulation in a rat model of hyperlipidemia. Biomaterials. 2019, 204, 13-24.
URL pmid: 30875515 |
| 107. | Chen, Z.; Wu, C.; Gu, W.; Klein, T.; Crawford, R.; Xiao, Y. Osteogenic differentiation of bone marrow MSCs by β-tricalcium phosphate stimulating macrophages via BMP2 signalling pathway. Biomaterials. 2014, 35, 1507-1518. |
| 108. |
Abaci, H. E.; Coffman, A.; Doucet, Y.; Chen, J.; Jacków, J.; Wang, E.; Guo, Z.; Shin, J. U.; Jahoda, C. A.; Christiano, A. M. Tissue engineering of human hair follicles using a biomimetic developmental approach. Nat Commun. 2018, 9, 5301.
doi: 10.1038/s41467-018-07579-y URL pmid: 30546011 |
| 109. | Plikus, M. V.; Guerrero-Juarez, C. F.; Ito, M.; Li, Y. R.; Dedhia, P. H.; Zheng, Y.; Shao, M.; Gay, D. L.; Ramos, R.; Hsi, T. C.; Oh, J. W.; Wang, X.; Ramirez, A.; Konopelski, S. E.; Elzein, A.; Wang, A.; Supapannachart, R. J.; Lee, H. L.; Lim, C. H.; Nace, A.; Guo, A.; Treffeisen, E.; Andl, T.; Ramirez, R. N.; Murad, R.; Offermanns, S.; Metzger, D.; Chambon, P.; Widgerow, A. D.; Tuan, T. L.; Mortazavi, A.; Gupta, R. K.; Hamilton, B. A.; Millar, S. E.; Seale, P.; Pear, W. S.; Lazar, M. A.; Cotsarelis, G. Regeneration of fat cells from myofibroblasts during wound healing. Science. 2017, 355, 748-752. |
| [1] | Kamolrat Metavarayuth, Esteban Villarreal, Hui Wang, Qian Wang. Surface topography and free energy regulate osteogenesis of stem cells: effects of shape-controlled gold nanoparticles [J]. Biomaterials Translational, 2021, 2(2): 165-173. |
| [2] | Yizhong Peng, Xiangcheng Qing, Hongyang Shu, Shuo Tian, Wenbo Yang, Songfeng Chen, Hui Lin, Xiao Lv, Lei Zhao, Xi Chen, Feifei Pu, Donghua Huang, Xu Cao, Zengwu Shao. Proper animal experimental designs for preclinical research of biomaterials for intervertebral disc regeneration [J]. Biomaterials Translational, 2021, 2(2): 91-142. |
| [3] | Isak Jatoi, Jingyu Fan. A biomaterials viewpoint for the 2020 SARS-CoV-2 vaccine development [J]. Biomaterials Translational, 2021, 2(1): 30-42. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||