Biomaterials Translational ›› 2021, Vol. 2 ›› Issue (2): 91-142.doi: 10.12336/biomatertransl.2021.02.003
• REVIEW • Previous Articles Next Articles
Yizhong Peng1, Xiangcheng Qing1, Hongyang Shu2,3, Shuo Tian1, Wenbo Yang1, Songfeng Chen4, Hui Lin1, Xiao Lv1, Lei Zhao1, Xi Chen1, Feifei Pu1, Donghua Huang4, Xu Cao5,*( ), Zengwu Shao1,*(
), Zengwu Shao1,*( )
)
Received:2021-04-08
Revised:2021-06-04
Accepted:2021-06-09
Online:2021-06-28
Published:2021-06-28
Contact:
Xu Cao,Zengwu Shao
E-mail:xcao11@jhmi.edu, szwpro@163.com
Peng, Y.; Qing, X.; Shu, H.; Tian, S.; Yang, W.; Chen, S.; Lin, H.; Lv, X.; Zhao, L.; Chen, X.; Pu, F.; Huang, D.; Cao, X.; Shao, Z. Proper animal experimental designs for preclinical research of biomaterials for intervertebral disc regeneration. Biomater Transl. 2021, 2(2), 91-142.
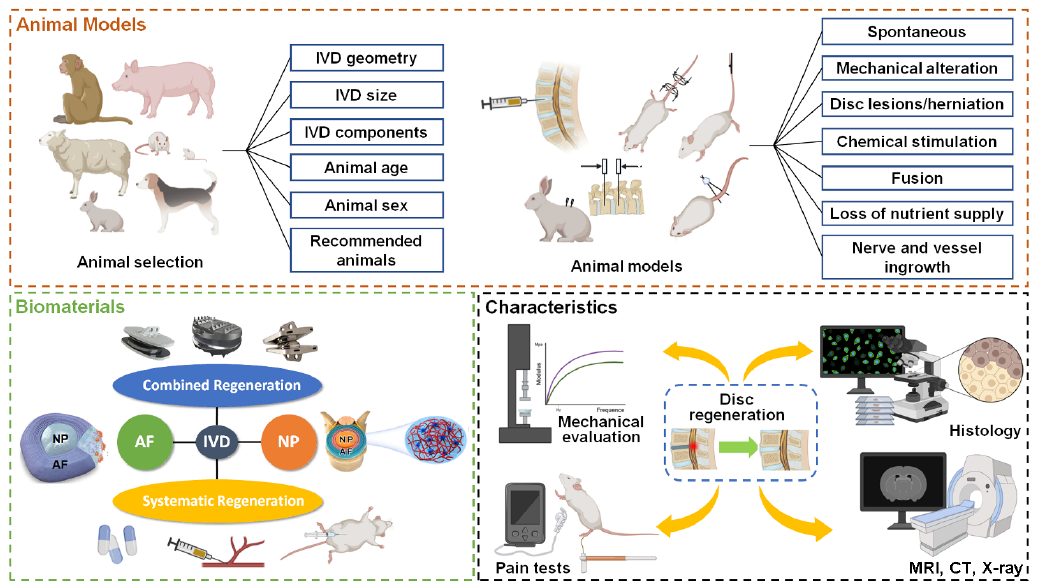
Figure 2. The structural diagram of this review. AF: annulus fibrosus; CT: computed tomography; IVD: intervertebral disc; MRI: magnetic resonance imaging; NP: nucleus pulposus.
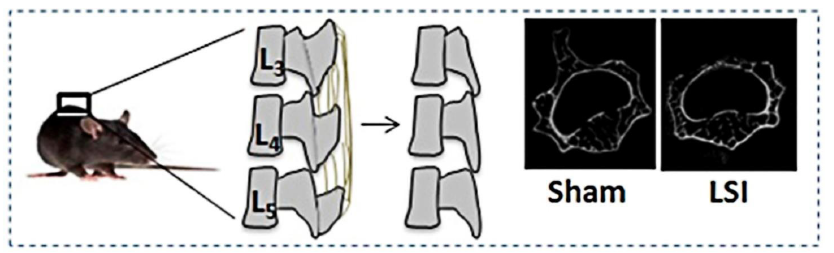
Figure 3. Lumbar spine instability mouse model (LSI). Mouse L3-5 spinous processes were resected along with the supraspinous and interspinous ligaments to induce instability of lumbar spine. Reprinted by permission from Macmillan Publishers Ltd.: Bian et al.83 Copyright 2016.
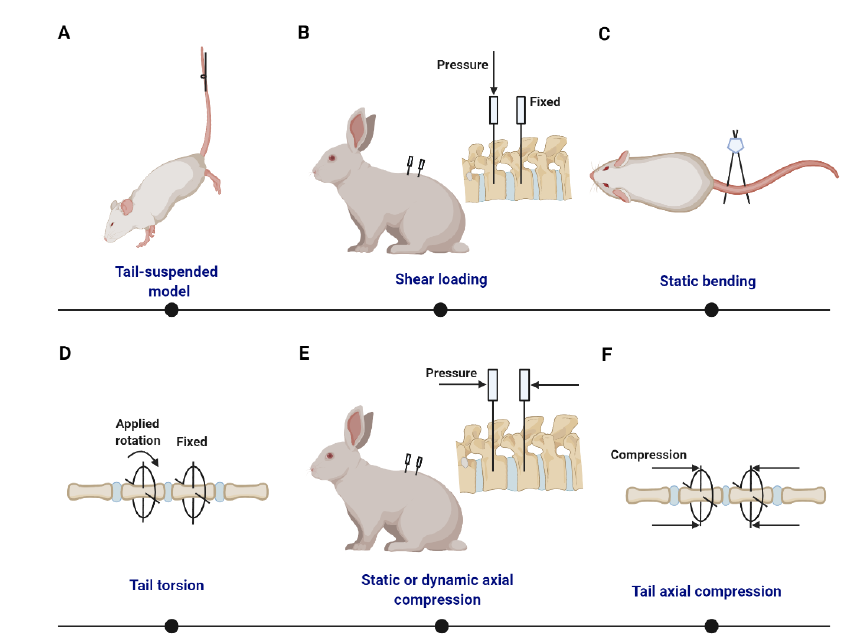
Figure 4. Summary of static/dynamic compression models with external apparatus. (A) Tail-suspended rat with its hind limbs off the floor.272 (B) Shear loading is generated from forces of different magnitudes on the adjecant vertebrae.281, 282 (C) Static disc bending model based on pins and an alignment jig.283 (D) Ilizarov-type apparatus is used to produce tail torsion. (E) Surgically implanted transfixing pins and percutaneous posts allow the application of static or dynamic axial compression and distraction loading at a single level of the rabbit lumbar spine.284 (F) Ilizarov-type apparatus is used to produce tail axial compression.
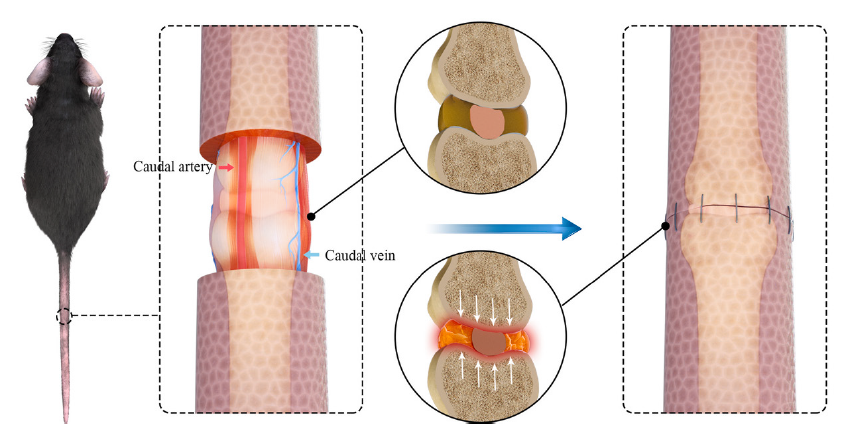
Figure 5. Compressive suture-induced rat IDD model. Circumcising the skin around index discs with a width of 2 mm and anastomosing the skin impose axial compression on the tail. Reprinted from Liu et al.292 Copyright ? 2021 with permission from Elsevier. IDD: intervertebral disc degeneration.
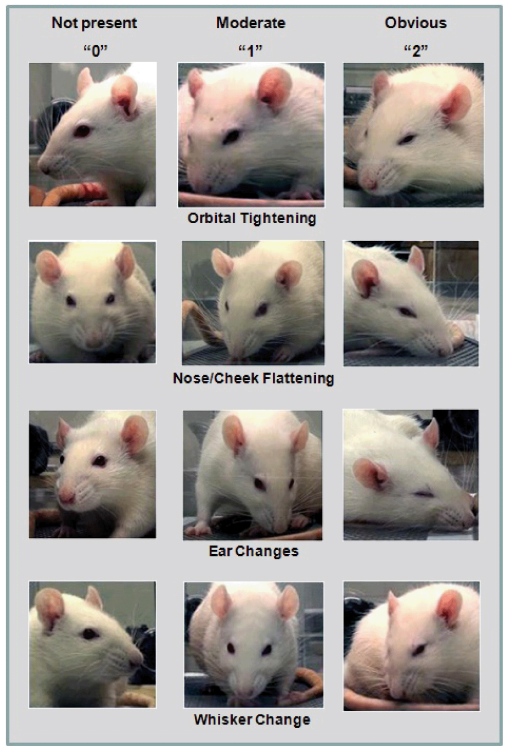
Figure 6. Scales for spontaneous pain evaluation based on rodent facial expressions. Copyright ?2011 Sotocinal et al.675 Reprinted from BioMed Central Ltd.
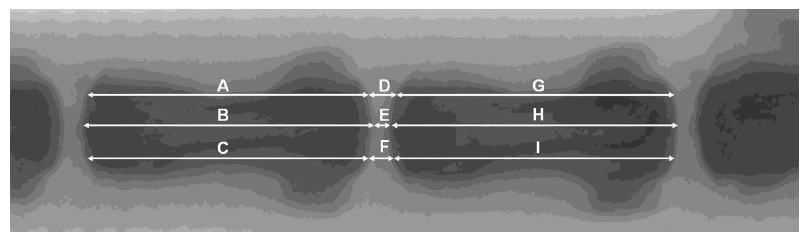
Figure 7. Measurements and calculations of vertebral body height and IVD height based on radiographs. The IVD height should be quantified using a relative value, %DHI, which measures changes in the DHI of punctured discs. DHI = 2 × (D + E + F) / (A + B + C + G + H + I); %DHI = post-punctured DHI / pre-punctured DHI × 100. DHI: disc height index; IVD: intervertebral disc.313
| Model type | Species | Manipulation | Reference |
|---|---|---|---|
| Disc disruption | |||
| Spontaneous | Mouse | Aging | |
| Ercc1 mutation | |||
| Cmd aggrecan knockout | |||
| Inherited kyphoscoliosis | |||
| Collagen II mutation | |||
| Collagen IX mutation | |||
| Myostatin knockout | |||
| Defect at ank locus, ankylosing spondylitis | |||
| twy mouse—IVD calcification and ankylosis | |||
| SPARC null | |||
| HLA B27 transgenic, spondylolisthesis | |||
| Rat | Aging | ||
| Sand rat | Chondrodystrophy, aging, breed | ||
| Dog | Spondylosis; aging | ||
| Chinese hamster | Aging | ||
| Baboon | Aging | ||
| Mechanical alteration | Mouse | Lumbar spine instability mouse model with/without ovariectomy | |
| Mouse, rat | Static compression | ||
| Rabbit | Shear stress | ||
| Rabbit | Compression injury, lumbar spine and caudal disc compression | ||
| Rat | Tail suspension | ||
| Shear stress | |||
| Amputation of upper limbs and tail | |||
| Mouse | Amputation of upper limbs | ||
| Rabbit | Resection of the cervical supraspinous and interspinous ligaments and detachment of the posterior paravertebral muscles from the cervical vertebrae; the removal of facet joints | ||
| Dog | Static compression | ||
| Pig | Resection of facet joint, interspinous and anterior ligament injury | ||
| Rabbit | Facetectomy/capsulotomy torsional lumbar injury | ||
| Disc herniation | Cavine | A partial laminectomy of the caudal part of the | |
| Rat | NP obtained from tail amputation and placed on nerve root | ||
| Rabbit | Bilateral facet joint resection at L7﹣S1 and rotational manipulation | ||
| External annular wound (2 mm) | |||
| Rat | Flexion, lateral bending and rotational forces | ||
| Disc lesions | Rabbit | Multiple 5 mm stab incisions using 16, 18 or 21G needles | |
| NP removal | |||
| 3﹣5 mm outer anterolateral annular incision (rim-lesion) | |||
| Ovine | Circumferential annular tear (delamellation) | ||
| A lateral retroperitoneal drill bit injury | |||
| Anular lesion by surgical incision through the left anterolateral AF | |||
| Pig | Combined lesions in AF (1.2 cm), NP (1.5 cm), facet joint and capsule | ||
| Rat | 5 mm stab by 18﹣30G needles | ||
| Dog | 4 mm posterior annulotomy | ||
| Local chemical stimulation | Rat | Chondroitinase ABC | |
| Rabbit | |||
| Sheep | |||
| Macaque | Chymopapain | ||
| Rabbit | Chymopapain | ||
| Rhesus monkeys | Pingyangmycin | ||
| Bleomycin | |||
| Dog | Fibronectin fragments | ||
| Rabbit | Fibronectin fragments | ||
| Chymopapain, krill proteases | |||
| Rat | Complete Freund's adjuvant | ||
| Rat | IL-1β | ||
| Rat | AGE | ||
| Systematic reagents stimulation | Mouse | Immunized with aggrecan and/or versican, develops spondylitis | |
| Dietary AGE | |||
| Diabetic | |||
| Fusion | Rabbit | Lumbar arthrodesis | |
| Sheep | Lumbar arthrodesis | ||
| Rat | Lumbar arthrodesis | ||
| Rabbit | Controlled dynamic distraction | ||
| Pinealectomy models of scoliosis | Chicken | Pinealectomy | |
| Rat | Pinealectomy + bipedal | ||
| Appendix | |||
| Loss of nutrient supply | Mouse, rat | Endplate perforation | |
| Pig | Disc allograft transplantation | ||
| Endplate perforation and cryoinjury | |||
| Goat | Ethanol injection to bone marrow vertebrae body | ||
| Cement injection to the adjecant vertebrae body | |||
| Rat | Nd: YAG laser on the CEP of the degenerated IVD | ||
| Nerves and vessels ingrowth | Pig | Annulus fibrosus puncture and poly(lactic-co-glycolic acid)/fibrin gel sealing | |
| Mouse | Disc puncture and nucleus pulposus removal | ||
| Sheep | Annulus fibrosus puncture | ||
| Nerve associated degeneration | Rabbit | Surgical narrowing of intervertebral neural foramen, vibrational stimulation of dorsal root ganglia | |
| Others | |||
| Hyperactivity | Dog | Long distance running training |
Additional Table 1 Animal models used to study disc degeneration
| Model type | Species | Manipulation | Reference |
|---|---|---|---|
| Disc disruption | |||
| Spontaneous | Mouse | Aging | |
| Ercc1 mutation | |||
| Cmd aggrecan knockout | |||
| Inherited kyphoscoliosis | |||
| Collagen II mutation | |||
| Collagen IX mutation | |||
| Myostatin knockout | |||
| Defect at ank locus, ankylosing spondylitis | |||
| twy mouse—IVD calcification and ankylosis | |||
| SPARC null | |||
| HLA B27 transgenic, spondylolisthesis | |||
| Rat | Aging | ||
| Sand rat | Chondrodystrophy, aging, breed | ||
| Dog | Spondylosis; aging | ||
| Chinese hamster | Aging | ||
| Baboon | Aging | ||
| Mechanical alteration | Mouse | Lumbar spine instability mouse model with/without ovariectomy | |
| Mouse, rat | Static compression | ||
| Rabbit | Shear stress | ||
| Rabbit | Compression injury, lumbar spine and caudal disc compression | ||
| Rat | Tail suspension | ||
| Shear stress | |||
| Amputation of upper limbs and tail | |||
| Mouse | Amputation of upper limbs | ||
| Rabbit | Resection of the cervical supraspinous and interspinous ligaments and detachment of the posterior paravertebral muscles from the cervical vertebrae; the removal of facet joints | ||
| Dog | Static compression | ||
| Pig | Resection of facet joint, interspinous and anterior ligament injury | ||
| Rabbit | Facetectomy/capsulotomy torsional lumbar injury | ||
| Disc herniation | Cavine | A partial laminectomy of the caudal part of the | |
| Rat | NP obtained from tail amputation and placed on nerve root | ||
| Rabbit | Bilateral facet joint resection at L7﹣S1 and rotational manipulation | ||
| External annular wound (2 mm) | |||
| Rat | Flexion, lateral bending and rotational forces | ||
| Disc lesions | Rabbit | Multiple 5 mm stab incisions using 16, 18 or 21G needles | |
| NP removal | |||
| 3﹣5 mm outer anterolateral annular incision (rim-lesion) | |||
| Ovine | Circumferential annular tear (delamellation) | ||
| A lateral retroperitoneal drill bit injury | |||
| Anular lesion by surgical incision through the left anterolateral AF | |||
| Pig | Combined lesions in AF (1.2 cm), NP (1.5 cm), facet joint and capsule | ||
| Rat | 5 mm stab by 18﹣30G needles | ||
| Dog | 4 mm posterior annulotomy | ||
| Local chemical stimulation | Rat | Chondroitinase ABC | |
| Rabbit | |||
| Sheep | |||
| Macaque | Chymopapain | ||
| Rabbit | Chymopapain | ||
| Rhesus monkeys | Pingyangmycin | ||
| Bleomycin | |||
| Dog | Fibronectin fragments | ||
| Rabbit | Fibronectin fragments | ||
| Chymopapain, krill proteases | |||
| Rat | Complete Freund's adjuvant | ||
| Rat | IL-1β | ||
| Rat | AGE | ||
| Systematic reagents stimulation | Mouse | Immunized with aggrecan and/or versican, develops spondylitis | |
| Dietary AGE | |||
| Diabetic | |||
| Fusion | Rabbit | Lumbar arthrodesis | |
| Sheep | Lumbar arthrodesis | ||
| Rat | Lumbar arthrodesis | ||
| Rabbit | Controlled dynamic distraction | ||
| Pinealectomy models of scoliosis | Chicken | Pinealectomy | |
| Rat | Pinealectomy + bipedal | ||
| Appendix | |||
| Loss of nutrient supply | Mouse, rat | Endplate perforation | |
| Pig | Disc allograft transplantation | ||
| Endplate perforation and cryoinjury | |||
| Goat | Ethanol injection to bone marrow vertebrae body | ||
| Cement injection to the adjecant vertebrae body | |||
| Rat | Nd: YAG laser on the CEP of the degenerated IVD | ||
| Nerves and vessels ingrowth | Pig | Annulus fibrosus puncture and poly(lactic-co-glycolic acid)/fibrin gel sealing | |
| Mouse | Disc puncture and nucleus pulposus removal | ||
| Sheep | Annulus fibrosus puncture | ||
| Nerve associated degeneration | Rabbit | Surgical narrowing of intervertebral neural foramen, vibrational stimulation of dorsal root ganglia | |
| Others | |||
| Hyperactivity | Dog | Long distance running training |
| Gauge number | Needle nominal O.D. (mm) | Needle nominal I.D. (mm) | Needle wall thickness (mm) |
|---|---|---|---|
| 10G | 3.404 | 2.693 | 0.356 |
| 11G | 3.048 | 2.388 | 0.33 |
| 12G | 2.769 | 2.159 | 0.305 |
| 13G | 2.413 | 1.804 | 0.305 |
| 14G | 2.109 | 1.6 | 0.254 |
| 15G | 1.829 | 1.372 | 0.229 |
| 16G | 1.651 | 1.194 | 0.229 |
| 17G | 1.473 | 1.067 | 0.203 |
| 18G | 1.27 | 0.838 | 0.216 |
| 19G | 1.067 | 0.686 | 0.191 |
| 20G | 0.908 | 0.603 | 0.152 |
| 21G | 0.819 | 0.514 | 0.152 |
| 22G | 0.718 | 0.413 | 0.152 |
| 23G | 0.642 | 0.337 | 0.152 |
| 24G | 0.566 | 0.311 | 0.127 |
| 25G | 0.515 | 0.26 | 0.127 |
| 26G | 0.464 | 0.26 | 0.102 |
| 27G | 0.413 | 0.21 | 0.102 |
| 28G | 0.362 | 0.184 | 0.089 |
| 29G | 0.337 | 0.184 | 0.076 |
| 30G | 0.312 | 0.159 | 0.076 |
| 31G | 0.261 | 0.133 | 0.064 |
| 32G | 0.235 | 0.108 | 0.064 |
| 33G | 0.21 | 0.108 | 0.051 |
| 34G | 0.159 | 0.051 | 0.051 |
Additional Table 2 Needle gauge and corresponding size
| Gauge number | Needle nominal O.D. (mm) | Needle nominal I.D. (mm) | Needle wall thickness (mm) |
|---|---|---|---|
| 10G | 3.404 | 2.693 | 0.356 |
| 11G | 3.048 | 2.388 | 0.33 |
| 12G | 2.769 | 2.159 | 0.305 |
| 13G | 2.413 | 1.804 | 0.305 |
| 14G | 2.109 | 1.6 | 0.254 |
| 15G | 1.829 | 1.372 | 0.229 |
| 16G | 1.651 | 1.194 | 0.229 |
| 17G | 1.473 | 1.067 | 0.203 |
| 18G | 1.27 | 0.838 | 0.216 |
| 19G | 1.067 | 0.686 | 0.191 |
| 20G | 0.908 | 0.603 | 0.152 |
| 21G | 0.819 | 0.514 | 0.152 |
| 22G | 0.718 | 0.413 | 0.152 |
| 23G | 0.642 | 0.337 | 0.152 |
| 24G | 0.566 | 0.311 | 0.127 |
| 25G | 0.515 | 0.26 | 0.127 |
| 26G | 0.464 | 0.26 | 0.102 |
| 27G | 0.413 | 0.21 | 0.102 |
| 28G | 0.362 | 0.184 | 0.089 |
| 29G | 0.337 | 0.184 | 0.076 |
| 30G | 0.312 | 0.159 | 0.076 |
| 31G | 0.261 | 0.133 | 0.064 |
| 32G | 0.235 | 0.108 | 0.064 |
| 33G | 0.21 | 0.108 | 0.051 |
| 34G | 0.159 | 0.051 | 0.051 |
| Animal | Needle size | Needle diameter/disc height (%) | Approach | Depth | Puncture position | Segements | Additional | Degenerated time point/longest recorded time | Mechanical | Biochemical | Height (longest recorded time) | Histologic and gross | Radiograph and MRI | Neuropathic pain | Reference | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Rat | 18G | 128% | Open/percutaneous puncture | Needle bevel completely inserted | Tail | C3/4 | - | 1/4 months | - | - | - | Yes, degenerated, NP herniation (more severe in open puncture) | Yes, progressed (more severe in open puncture) | |||||
| 20G | 95% | Percutaneous puncture | 5 mm (through the annulus fibrosus); 10 mm (full penetration) | Tail | C6/7﹣C9/10 | - | 2﹣4/4﹣8 weeks | - | Decreased GAG (by ~11% for 5 mm, by ~16% for 10 mm) | Decreased (by ~10% for 5 mm; by ~20% for 10 mm) | Yes, degenerated, NP herniation (more severe in full penetration) | Yes, progressed (more severe in full penetration) | Yes | |||||
| 20G | 95% | Percutaneous puncture | Through the annulus fibrosus | Tail | C6/7﹣C8/9 | - | 1﹣4/4﹣24 weeks | - | Decreased water, GAG and type I collagen expression | Decreased (by 25-75%) | Yes, degenerated | Yes, progressed | - | |||||
| 21G | 85% | Open puncture | 3 mm (through the annulus fibrosus) | Posterial approach | L4/5 | - | 4/8 weeks | - | Altered collagens expression | - | - | Yes, progressed | - | |||||
| 21G | 85% | Open puncture | 3 mm (through the annulus fibrosus) | Posterior/anterior approach | L4/5 | - | 2/6 weeks | - | - | - | Yes, degenerated | Yes, progressed | Yes (more significant for posterior puncture) | |||||
| 21G | 85% | Open puncture | 3 mm (through the annulus fibrosus) | Tail | C4/5, C8/9 | - | 2 weeks | - | - | - | Yes, degenerated, NP herniation | Yes, progressed | Yes | |||||
| 21G | 85% | Open puncture | 5 mm (through the annulus fibrosus) | Tail | C5/6, C7/8 | - | 4 weeks | - | Altered collagens expression | - | Yes, degenerated | Yes, progressed | - | |||||
| 21G | 85% | Percutaneous puncture | Through the annulus fibrosus | Tail | C4/5﹣C8/9 | - | 1﹣2/14﹣42 days | - | - | - | Yes, degenerated | Yes, progressed | - | |||||
| 23G | 64% | Open puncture | Through the annulus fibrosus | Lateral approach | L5/6 | Repetitive puncture for five times | 1/2 weeks | - | - | - | - | - | Yes, increased neurons staining | |||||
| 27G | 51% | Open puncture | Through the annulus fibrosus | Dorsal approach | L4/5, L5/6 | - | 2/8 weeks | - | Altered collagens, SOX9, aggrecan expression | Yes, degenerated, NP herniation | Yes, progressed | - | ||||||
| 31G | 26% | Percutaneous puncture | 1.5 mm (through the annulus fibrosus) | Tail | C6/7 | - | 4 weeks | - | Altered collagens, aggrecan, MMP13, Adamts4 expression | - | - | - | ||||||
| 18G/22G | 128%/74% | Percutaneous puncture | Through the annulus fibrosus | Tail | C6/7, C8/9 | - | 2/4 weeks | - | - | - | Yes, degenerated | - | - | |||||
| 18G/22G/26G | 128%/74%/20% | Percutaneous puncture | 2 mm (through the annulus fibrosus) | Tail | C6/7, C8/9 | - | 1/4 weeks | Altered creep behavior (for 18G) | - | Yes, degenerated (more severe for 18G) | Yes, progressed | - | ||||||
| 18/20/22G | 128%/95%/74% | Percutaneous puncture | 5 mm (through the annulus fibrosus) | Tail | C6/7, C8/9 | - | 2/8 weeks | - | Increased proteoglycan (for 18G, 20G) | Decreased (for 18G) | Yes, degenerated, NP herniation (more severe in 18G) | Yes, progressed (more severe in 18G) | ||||||
| 18/21/23/ 25/27/29G | 128%/85%/64%/ 53%/51%/36% | Percutaneous puncture | 5 cmm (through the annulus fibrosus) | Tail | NA | - | 2/8﹣12 weeks | - | Altered collagens, SOX9, aggrecan expression | Decreased (by ~10% for 29/27/25G, by ~30% for 23/21G, by ~35% for 18G) | Yes, degenerated (more severe in 18G) | Yes, progressed (more severe in 18G) | ||||||
| 16G/18G/26G | 170%/128%/50% | Percutaneous puncture | Full penetration | Tail | C8/9 | - | 2/4 weeks | - | Altered Collagens expression | Yes, degenerated, NP herniation (more severe in 16G, 18G) | Yes, progressed (more severe in 16G, 18G) | |||||||
| Mice | 26G | 100% | Percutaneous puncture | 2/3 of the disc thickness | Tail | C3/4﹣C6/7 | - | 4﹣8/4﹣32 weeks | - | Altered Collagens, MMPs, Adam8, Cxcl-1 expression | - | Yes, degenerated, NP herniation | - | - | ||||
| 27G | 90% | Open puncture | Through the annulus fibrosus | Anterior approach | L3/4, L4/5 | - | 1/7 days | - | - | - | Yes, degenerated, NP herniation | - | - | |||||
| 30G | 63% | Percutaneous puncture | Needle bevel completely inserted | Tail | NA | - | 14 weeks | - | - | Decreased (by ~25%) | Yes, degenerated | - | - | |||||
| 31G | 55% | Open puncture | 1 mm (through the annulus fibrosus) | Tail | C9/10 | - | 1/12 weeks | - | Altered collagens, GAG, aggrecan expression | Decreased (by ~30%) | Yes, degenerated | - | - | |||||
| 26G/29G | 100%/65% | Percutaneous puncture | 1.75 mm or 90% of the dorsoventral width | Dorsal approach | C6/7-C8/9 | - | 8 weeks | Decreased compressive stiffness, torsional stiffness, torque range, nc compressive ROM, increased creep displacement (for 26G) | Decreased GAG (by ~30% for 26G) | Decreased (by ~30% for 26G) | Yes, degenerated, NP herniation | - | - | |||||
| 27G/29G/31G | 90%/70%/55% | Percutaneous puncture | Through the annulus fibrosus/full penetration | Tail | C7/8, C9/10 | - | 4 weeks | - | - | NS | Yes, degenerated, NP herniation (more severe in full penetration) | Yes, progressed (more severe in full penetration) | ||||||
| 27/30/33G | 90%/63%/40% | Open puncture | NA | Ventral approach | L4/5﹣L6/S1 | - | 1/8 weeks | - | Decreased GAG expression (for 27G and 30G) | Yes, degenerated (more severe for 27G and 30G) | - | |||||||
| 33G/35G | 50%/42% | Open puncture | Through the annulus fibrosus | Ventral/central/dorsal approach | L4/5 | - | 1/12 weeks | - | - | - | Yes, degenerated (more severe in central/dorsal approach) | Yes, progressed (more severe in central/dorsal approach) | ||||||
| Rabbit | 16G | 66% | Percutaneous puncture | Through the annulus fibrosus | Lateral approach | L2/3﹣L4/5 | NP removal with negative pressure | 6/12 weeks | - | Decreased collagen X expression | Decreased (by ~25%) | Yes, degenerated | - | - | ||||
| 16G | 66% | Open puncture | 5 mm (through the annulus fibrosus) | Posterolateral approach | L3/4, L5/6 | - | 4/12 weeks | - | - | Decreased (by ~45%) | Yes, degenerated, NP herniation | Yes, progressed | - | |||||
| 16G | 66% | Open puncture | 5 mm (through the annulus fibrosus) | Anterolateral approach | L2/3﹣L6/7 | NP removal with negative pressure | 2﹣8/12﹣24 weeks | Decreased ROM, increased creep displacement | Altered GAG, collagens, aggrecan, MMP3, SOX9 expression | Decreased (by ~25%) | Yes, degenerated, NP herniation | Yes, progressed | - | |||||
| 18G | 50% | Open puncture | 5 mm (through the annulus fibrosus) | Anterior/lateral approach | L2/3﹣L6/7 | NP removal with negative pressure | 1﹣4/4﹣14 weeks | - | Decreased GAG, proteoglycan (by ~30%) | Decreased (by ~30%) | Yes, degenerated | Yes, progressed | - | |||||
| 18G | 50% | Percutaneous puncture | Through the annulus fibrosus | Lateral approach | L5/6 | NP removal with negative pressure | 1﹣4/4﹣12 weeks | - | Decreased GAG, collagens expression | Decreased (by ~50%) | Yes, degenerated, NP herniation | Yes, progressed | - | |||||
| 18G | 50% | Open puncture | 1 mm (superfical annulus defect); 5 mm (through the annulus fibrosus) | Anterior approach | L2/3﹣L4/5 | - | 2/12﹣24 weeks | - | - | Decreased (NS for 1 mm puncture, by ~25% for 5 mm puncture) | Yes, degenerated, NP herniation (for 5 mm puncture) | Yes, progressed (for 5 mm puncture) | ||||||
| 19G | 44% | Percutaneous puncture | 5 mm (through the annulus fibrosus) | Posterolateral approach | L2/3﹣L4/5 | NP removal with negative pressure | 9/20 weeks | - | - | Decreased (by ~40%) | Yes, degenerated | Yes, progressed | - | |||||
| 21G | 27% | Open puncture | 5 mm (through the annulus fibrosus) | Anterior approach | L3/4﹣L5/6 | NP removal with negative pressure | 4/12﹣28 weeks | - | Decreased proteoglycan expression | - | Yes, degenerated | - | - | |||||
| 16/18/21G | 66%/50%/27% | Open puncture | 5 mm (through the annulus fibrosus) | Anterior approach | L2/3﹣L5/6 | - | 4/8 weeks | - | - | Decreased (by ~30% for 16G, by ~10% for 18G/21G) | Yes, degenerated, NP herniation (for 16G/18G) | Yes, progressed (for 16G/18G) | ||||||
| Pig | 3.2 mm diameter trephine | 62% | Open puncture | NA | Anterolateral approach | NA | - | 8/39 weeks | - | - | Decreased (by ~15%) | Yes, degenerated | - | - | ||||
| 16G | 30% | Open puncture | Through the annulus fibrosus | Anterolateral approach | L2/5 | NP removal with negative pressure | 3/12﹣24 weeks | - | Altered Collagens, MMPs, aggrecan, TIMPs expression | Yes, degenerated | Yes, progressed | - | ||||||
| 20G | 17% | Open puncture | Through the annulus fibrosus | NA | L2/3, L4/5 | NP removal with negative pressure | 12/24 weeks | - | - | - | Yes, degenerated | Yes, progressed | - | |||||
| Rhesus monkeys | 15G/20G | 41%/20% | Percutaneous puncture | Through the annulus fibrosus | Anterolateral approach | L1/2﹣L5/6 | - | 4/12 weeks | - | - | - | Yes, degenerated (more severe in 15G) | Yes, progressed (more severe in 15G) | |||||
| Ovine | 3.2-4.5 mm drill | 94﹣100% | Open puncture | 9﹣15 mm (through the annulus fibrosus) | Lateral approach | L1/2﹣L5/6 | - | 16 weeks | - | - | - | Yes, degenerated | Yes, progressed | - | ||||
| CD-Canine | NA | 30﹣50% | Open puncture | Through the annulus fibrosus | Dorsal approach | L1/2, L3/4, L5/6 | - | 14 weeks | - | Altered aggrecan, collagens expression | - | Yes, degenerated | Yes, progressed | - | ||||
Additional Table 3 Parameters for needle puncture-induced intervertebral disc degeneration models
| Animal | Needle size | Needle diameter/disc height (%) | Approach | Depth | Puncture position | Segements | Additional | Degenerated time point/longest recorded time | Mechanical | Biochemical | Height (longest recorded time) | Histologic and gross | Radiograph and MRI | Neuropathic pain | Reference | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Rat | 18G | 128% | Open/percutaneous puncture | Needle bevel completely inserted | Tail | C3/4 | - | 1/4 months | - | - | - | Yes, degenerated, NP herniation (more severe in open puncture) | Yes, progressed (more severe in open puncture) | |||||
| 20G | 95% | Percutaneous puncture | 5 mm (through the annulus fibrosus); 10 mm (full penetration) | Tail | C6/7﹣C9/10 | - | 2﹣4/4﹣8 weeks | - | Decreased GAG (by ~11% for 5 mm, by ~16% for 10 mm) | Decreased (by ~10% for 5 mm; by ~20% for 10 mm) | Yes, degenerated, NP herniation (more severe in full penetration) | Yes, progressed (more severe in full penetration) | Yes | |||||
| 20G | 95% | Percutaneous puncture | Through the annulus fibrosus | Tail | C6/7﹣C8/9 | - | 1﹣4/4﹣24 weeks | - | Decreased water, GAG and type I collagen expression | Decreased (by 25-75%) | Yes, degenerated | Yes, progressed | - | |||||
| 21G | 85% | Open puncture | 3 mm (through the annulus fibrosus) | Posterial approach | L4/5 | - | 4/8 weeks | - | Altered collagens expression | - | - | Yes, progressed | - | |||||
| 21G | 85% | Open puncture | 3 mm (through the annulus fibrosus) | Posterior/anterior approach | L4/5 | - | 2/6 weeks | - | - | - | Yes, degenerated | Yes, progressed | Yes (more significant for posterior puncture) | |||||
| 21G | 85% | Open puncture | 3 mm (through the annulus fibrosus) | Tail | C4/5, C8/9 | - | 2 weeks | - | - | - | Yes, degenerated, NP herniation | Yes, progressed | Yes | |||||
| 21G | 85% | Open puncture | 5 mm (through the annulus fibrosus) | Tail | C5/6, C7/8 | - | 4 weeks | - | Altered collagens expression | - | Yes, degenerated | Yes, progressed | - | |||||
| 21G | 85% | Percutaneous puncture | Through the annulus fibrosus | Tail | C4/5﹣C8/9 | - | 1﹣2/14﹣42 days | - | - | - | Yes, degenerated | Yes, progressed | - | |||||
| 23G | 64% | Open puncture | Through the annulus fibrosus | Lateral approach | L5/6 | Repetitive puncture for five times | 1/2 weeks | - | - | - | - | - | Yes, increased neurons staining | |||||
| 27G | 51% | Open puncture | Through the annulus fibrosus | Dorsal approach | L4/5, L5/6 | - | 2/8 weeks | - | Altered collagens, SOX9, aggrecan expression | Yes, degenerated, NP herniation | Yes, progressed | - | ||||||
| 31G | 26% | Percutaneous puncture | 1.5 mm (through the annulus fibrosus) | Tail | C6/7 | - | 4 weeks | - | Altered collagens, aggrecan, MMP13, Adamts4 expression | - | - | - | ||||||
| 18G/22G | 128%/74% | Percutaneous puncture | Through the annulus fibrosus | Tail | C6/7, C8/9 | - | 2/4 weeks | - | - | - | Yes, degenerated | - | - | |||||
| 18G/22G/26G | 128%/74%/20% | Percutaneous puncture | 2 mm (through the annulus fibrosus) | Tail | C6/7, C8/9 | - | 1/4 weeks | Altered creep behavior (for 18G) | - | Yes, degenerated (more severe for 18G) | Yes, progressed | - | ||||||
| 18/20/22G | 128%/95%/74% | Percutaneous puncture | 5 mm (through the annulus fibrosus) | Tail | C6/7, C8/9 | - | 2/8 weeks | - | Increased proteoglycan (for 18G, 20G) | Decreased (for 18G) | Yes, degenerated, NP herniation (more severe in 18G) | Yes, progressed (more severe in 18G) | ||||||
| 18/21/23/ 25/27/29G | 128%/85%/64%/ 53%/51%/36% | Percutaneous puncture | 5 cmm (through the annulus fibrosus) | Tail | NA | - | 2/8﹣12 weeks | - | Altered collagens, SOX9, aggrecan expression | Decreased (by ~10% for 29/27/25G, by ~30% for 23/21G, by ~35% for 18G) | Yes, degenerated (more severe in 18G) | Yes, progressed (more severe in 18G) | ||||||
| 16G/18G/26G | 170%/128%/50% | Percutaneous puncture | Full penetration | Tail | C8/9 | - | 2/4 weeks | - | Altered Collagens expression | Yes, degenerated, NP herniation (more severe in 16G, 18G) | Yes, progressed (more severe in 16G, 18G) | |||||||
| Mice | 26G | 100% | Percutaneous puncture | 2/3 of the disc thickness | Tail | C3/4﹣C6/7 | - | 4﹣8/4﹣32 weeks | - | Altered Collagens, MMPs, Adam8, Cxcl-1 expression | - | Yes, degenerated, NP herniation | - | - | ||||
| 27G | 90% | Open puncture | Through the annulus fibrosus | Anterior approach | L3/4, L4/5 | - | 1/7 days | - | - | - | Yes, degenerated, NP herniation | - | - | |||||
| 30G | 63% | Percutaneous puncture | Needle bevel completely inserted | Tail | NA | - | 14 weeks | - | - | Decreased (by ~25%) | Yes, degenerated | - | - | |||||
| 31G | 55% | Open puncture | 1 mm (through the annulus fibrosus) | Tail | C9/10 | - | 1/12 weeks | - | Altered collagens, GAG, aggrecan expression | Decreased (by ~30%) | Yes, degenerated | - | - | |||||
| 26G/29G | 100%/65% | Percutaneous puncture | 1.75 mm or 90% of the dorsoventral width | Dorsal approach | C6/7-C8/9 | - | 8 weeks | Decreased compressive stiffness, torsional stiffness, torque range, nc compressive ROM, increased creep displacement (for 26G) | Decreased GAG (by ~30% for 26G) | Decreased (by ~30% for 26G) | Yes, degenerated, NP herniation | - | - | |||||
| 27G/29G/31G | 90%/70%/55% | Percutaneous puncture | Through the annulus fibrosus/full penetration | Tail | C7/8, C9/10 | - | 4 weeks | - | - | NS | Yes, degenerated, NP herniation (more severe in full penetration) | Yes, progressed (more severe in full penetration) | ||||||
| 27/30/33G | 90%/63%/40% | Open puncture | NA | Ventral approach | L4/5﹣L6/S1 | - | 1/8 weeks | - | Decreased GAG expression (for 27G and 30G) | Yes, degenerated (more severe for 27G and 30G) | - | |||||||
| 33G/35G | 50%/42% | Open puncture | Through the annulus fibrosus | Ventral/central/dorsal approach | L4/5 | - | 1/12 weeks | - | - | - | Yes, degenerated (more severe in central/dorsal approach) | Yes, progressed (more severe in central/dorsal approach) | ||||||
| Rabbit | 16G | 66% | Percutaneous puncture | Through the annulus fibrosus | Lateral approach | L2/3﹣L4/5 | NP removal with negative pressure | 6/12 weeks | - | Decreased collagen X expression | Decreased (by ~25%) | Yes, degenerated | - | - | ||||
| 16G | 66% | Open puncture | 5 mm (through the annulus fibrosus) | Posterolateral approach | L3/4, L5/6 | - | 4/12 weeks | - | - | Decreased (by ~45%) | Yes, degenerated, NP herniation | Yes, progressed | - | |||||
| 16G | 66% | Open puncture | 5 mm (through the annulus fibrosus) | Anterolateral approach | L2/3﹣L6/7 | NP removal with negative pressure | 2﹣8/12﹣24 weeks | Decreased ROM, increased creep displacement | Altered GAG, collagens, aggrecan, MMP3, SOX9 expression | Decreased (by ~25%) | Yes, degenerated, NP herniation | Yes, progressed | - | |||||
| 18G | 50% | Open puncture | 5 mm (through the annulus fibrosus) | Anterior/lateral approach | L2/3﹣L6/7 | NP removal with negative pressure | 1﹣4/4﹣14 weeks | - | Decreased GAG, proteoglycan (by ~30%) | Decreased (by ~30%) | Yes, degenerated | Yes, progressed | - | |||||
| 18G | 50% | Percutaneous puncture | Through the annulus fibrosus | Lateral approach | L5/6 | NP removal with negative pressure | 1﹣4/4﹣12 weeks | - | Decreased GAG, collagens expression | Decreased (by ~50%) | Yes, degenerated, NP herniation | Yes, progressed | - | |||||
| 18G | 50% | Open puncture | 1 mm (superfical annulus defect); 5 mm (through the annulus fibrosus) | Anterior approach | L2/3﹣L4/5 | - | 2/12﹣24 weeks | - | - | Decreased (NS for 1 mm puncture, by ~25% for 5 mm puncture) | Yes, degenerated, NP herniation (for 5 mm puncture) | Yes, progressed (for 5 mm puncture) | ||||||
| 19G | 44% | Percutaneous puncture | 5 mm (through the annulus fibrosus) | Posterolateral approach | L2/3﹣L4/5 | NP removal with negative pressure | 9/20 weeks | - | - | Decreased (by ~40%) | Yes, degenerated | Yes, progressed | - | |||||
| 21G | 27% | Open puncture | 5 mm (through the annulus fibrosus) | Anterior approach | L3/4﹣L5/6 | NP removal with negative pressure | 4/12﹣28 weeks | - | Decreased proteoglycan expression | - | Yes, degenerated | - | - | |||||
| 16/18/21G | 66%/50%/27% | Open puncture | 5 mm (through the annulus fibrosus) | Anterior approach | L2/3﹣L5/6 | - | 4/8 weeks | - | - | Decreased (by ~30% for 16G, by ~10% for 18G/21G) | Yes, degenerated, NP herniation (for 16G/18G) | Yes, progressed (for 16G/18G) | ||||||
| Pig | 3.2 mm diameter trephine | 62% | Open puncture | NA | Anterolateral approach | NA | - | 8/39 weeks | - | - | Decreased (by ~15%) | Yes, degenerated | - | - | ||||
| 16G | 30% | Open puncture | Through the annulus fibrosus | Anterolateral approach | L2/5 | NP removal with negative pressure | 3/12﹣24 weeks | - | Altered Collagens, MMPs, aggrecan, TIMPs expression | Yes, degenerated | Yes, progressed | - | ||||||
| 20G | 17% | Open puncture | Through the annulus fibrosus | NA | L2/3, L4/5 | NP removal with negative pressure | 12/24 weeks | - | - | - | Yes, degenerated | Yes, progressed | - | |||||
| Rhesus monkeys | 15G/20G | 41%/20% | Percutaneous puncture | Through the annulus fibrosus | Anterolateral approach | L1/2﹣L5/6 | - | 4/12 weeks | - | - | - | Yes, degenerated (more severe in 15G) | Yes, progressed (more severe in 15G) | |||||
| Ovine | 3.2-4.5 mm drill | 94﹣100% | Open puncture | 9﹣15 mm (through the annulus fibrosus) | Lateral approach | L1/2﹣L5/6 | - | 16 weeks | - | - | - | Yes, degenerated | Yes, progressed | - | ||||
| CD-Canine | NA | 30﹣50% | Open puncture | Through the annulus fibrosus | Dorsal approach | L1/2, L3/4, L5/6 | - | 14 weeks | - | Altered aggrecan, collagens expression | - | Yes, degenerated | Yes, progressed | - | ||||
| Subtype | Method | Protocol | Reference |
|---|---|---|---|
| Mechanical | Tactile responses | Rats are placed in individual plexiglass boxes on a stainless-steel mesh floor and are allowed to adjust for at least 20 minutes. | |
| A series of calibrated von Frey filaments (range 4﹣28 g) is applied perpendicularly to the plantar surface of a hindpaw with sufficient force to bend the filament for 6 seconds. | |||
| Brisk withdrawal or paw flinching is considered as a positive response. | |||
| Once a positive response is seen, the previous filament is applied. | |||
| If positive, the lower filament is determined to be the 50% paw-withdrawal threshold. | |||
| If negative, the next ascending filament is applied. | |||
| If that next filament provokes a positive response, the original filament is considered to be the 50% withdrawal threshold. | |||
| If the next ascending filament is negative, further ascending filaments are applied until a response is provoked. | |||
| Cautions: Avoid obscure foot pads and surgical incisions, and ensure that the position of the pain measurement is fixed in the central area of the foot; repeat the test four to five times at 5-min intervals on each animal. | |||
| Mechanical algesia | A von Frey anesthesiometer and rigid von Frey filaments are used to quantifying the withdrawal threshold of the hindpaw in response to mechanical stimulation. | ||
| Rats are placed in individual plexiglass boxes on a stainless-steel mesh floor and are allowed to acclimate for at least 20 minutes. | |||
| A 0.5-mm diameter polypropylene rigid tip is used to apply a force to the plantar surface of the hindpaw. | |||
| The force causing the withdrawal response is recorded by the anesthesiometer. | |||
| The anesthesiometer is calibrated before each recording. | |||
| The test is repeated four to five times at 5-minute intervals on each animal, and the mean value is calculated. | |||
| Mechanical hyperalgesia/pressure hyperalgesia | The vocalization threshold based on the force of an applied force gauge is measured by pressing the 0.5-c | ||
| The force was slowly increased 100 g/s until an audible vocalization is heard. | |||
| A cut off force of 1000 g is used to prevent tissue trauma. | |||
| The tests should be carried out in duplicate, and the mean value is taken as the nociceptive threshold. | |||
| Caution: Postoperative testing should be delayed until one week after surgery to allow the abdominal tissue to heal. | |||
| Thermal | Hot algesia (plate) | Rats were placed within a plexiglass chamber on a transparent glass surface and allowed to acclimate for at least 20 minutes. | |
| A thermal stimulation meter is used with the temperature set to 50°C and the stimulating time set to 30 seconds. | |||
| Brisk withdrawal or paw flinching is considered as a positive response. | |||
| The duration from stimulation to positive responses is recorded and noted as paw withdrawal latency. | |||
| Individual measurements were repeated four to five times. The intermittent period for repetitive measurements of each rat is 15 minutes. | |||
| The mean value was calculated as the thermal threshold. | |||
| Cautions: The tests should be restricted to a certain period in a day, like 8-12 a.m., to avoid the influence of memorial reflex. Data from scalded rats should be eliminated to avoid bias. | |||
| Hot algesia (tail flick test) | Animal are calmed by enclosing their heads with a towel on the apparatus, and acclimate to the test environment for 30 minutes. | ||
| Radiant heat is applied to the tail 5 cm from the tip using a tail-fick analgesia meter. | |||
| Record baseline latencies of the animals. Test the animals’ tail-flick response using a tail-flick apparatus, and adjust the intensity of the heat source to produce tail-flick latencies of 3 to 4 seconds. For mice, focus the light beam ~15 mm from the tip of the tail. For rats, stimulate an area ~50 mm from the tip of the tail. In the absence of a withdrawal reflex, set the stimulus cutoff to 10 seconds to avoid possible tissue damage. | |||
| Record the time for the animal to show a tail-flick response, or assign a value of 10 seconds (cutoff time) if no tail-flick is observed. | |||
| After sufficient data collection (n = 8 per group and dose), perform statistical analysis and calculate the means and standard errors for data presentation. | |||
| Cold algesia (hindpaw and back) | The total duration of acetone-evoked behaviours (e.g. flinching, licking or biting) are measured in seconds for 1 minute after a drop of acetone (25 μL) is applied to the plantar surface of the hindpaw using a blunt needle connected to a 1 mL-syringe. | ||
| Increased behavioural response to acetone suggests the development of cold hypersensitivity. | |||
| The grades are recorded as follow: 0, static; 1, slow flinching or paw movement; 2, fast flinching with paw shaking; 3, fast flinching, biting and paw remaining off the ground. | |||
| Cold algesia (tail) | Animals were placed individually in the test chamber for 60 minutes prior to testing. | ||
| Half of the length of the tail was dipped into the cold water, and the latency to tail withdrawal was measured. | |||
| A maximum cut-off of 30 seconds was set to avoid tissue damage. |
Additional Table 4 Stimuli-evoked hypersensitivity measurement in rodent model
| Subtype | Method | Protocol | Reference |
|---|---|---|---|
| Mechanical | Tactile responses | Rats are placed in individual plexiglass boxes on a stainless-steel mesh floor and are allowed to adjust for at least 20 minutes. | |
| A series of calibrated von Frey filaments (range 4﹣28 g) is applied perpendicularly to the plantar surface of a hindpaw with sufficient force to bend the filament for 6 seconds. | |||
| Brisk withdrawal or paw flinching is considered as a positive response. | |||
| Once a positive response is seen, the previous filament is applied. | |||
| If positive, the lower filament is determined to be the 50% paw-withdrawal threshold. | |||
| If negative, the next ascending filament is applied. | |||
| If that next filament provokes a positive response, the original filament is considered to be the 50% withdrawal threshold. | |||
| If the next ascending filament is negative, further ascending filaments are applied until a response is provoked. | |||
| Cautions: Avoid obscure foot pads and surgical incisions, and ensure that the position of the pain measurement is fixed in the central area of the foot; repeat the test four to five times at 5-min intervals on each animal. | |||
| Mechanical algesia | A von Frey anesthesiometer and rigid von Frey filaments are used to quantifying the withdrawal threshold of the hindpaw in response to mechanical stimulation. | ||
| Rats are placed in individual plexiglass boxes on a stainless-steel mesh floor and are allowed to acclimate for at least 20 minutes. | |||
| A 0.5-mm diameter polypropylene rigid tip is used to apply a force to the plantar surface of the hindpaw. | |||
| The force causing the withdrawal response is recorded by the anesthesiometer. | |||
| The anesthesiometer is calibrated before each recording. | |||
| The test is repeated four to five times at 5-minute intervals on each animal, and the mean value is calculated. | |||
| Mechanical hyperalgesia/pressure hyperalgesia | The vocalization threshold based on the force of an applied force gauge is measured by pressing the 0.5-c | ||
| The force was slowly increased 100 g/s until an audible vocalization is heard. | |||
| A cut off force of 1000 g is used to prevent tissue trauma. | |||
| The tests should be carried out in duplicate, and the mean value is taken as the nociceptive threshold. | |||
| Caution: Postoperative testing should be delayed until one week after surgery to allow the abdominal tissue to heal. | |||
| Thermal | Hot algesia (plate) | Rats were placed within a plexiglass chamber on a transparent glass surface and allowed to acclimate for at least 20 minutes. | |
| A thermal stimulation meter is used with the temperature set to 50°C and the stimulating time set to 30 seconds. | |||
| Brisk withdrawal or paw flinching is considered as a positive response. | |||
| The duration from stimulation to positive responses is recorded and noted as paw withdrawal latency. | |||
| Individual measurements were repeated four to five times. The intermittent period for repetitive measurements of each rat is 15 minutes. | |||
| The mean value was calculated as the thermal threshold. | |||
| Cautions: The tests should be restricted to a certain period in a day, like 8-12 a.m., to avoid the influence of memorial reflex. Data from scalded rats should be eliminated to avoid bias. | |||
| Hot algesia (tail flick test) | Animal are calmed by enclosing their heads with a towel on the apparatus, and acclimate to the test environment for 30 minutes. | ||
| Radiant heat is applied to the tail 5 cm from the tip using a tail-fick analgesia meter. | |||
| Record baseline latencies of the animals. Test the animals’ tail-flick response using a tail-flick apparatus, and adjust the intensity of the heat source to produce tail-flick latencies of 3 to 4 seconds. For mice, focus the light beam ~15 mm from the tip of the tail. For rats, stimulate an area ~50 mm from the tip of the tail. In the absence of a withdrawal reflex, set the stimulus cutoff to 10 seconds to avoid possible tissue damage. | |||
| Record the time for the animal to show a tail-flick response, or assign a value of 10 seconds (cutoff time) if no tail-flick is observed. | |||
| After sufficient data collection (n = 8 per group and dose), perform statistical analysis and calculate the means and standard errors for data presentation. | |||
| Cold algesia (hindpaw and back) | The total duration of acetone-evoked behaviours (e.g. flinching, licking or biting) are measured in seconds for 1 minute after a drop of acetone (25 μL) is applied to the plantar surface of the hindpaw using a blunt needle connected to a 1 mL-syringe. | ||
| Increased behavioural response to acetone suggests the development of cold hypersensitivity. | |||
| The grades are recorded as follow: 0, static; 1, slow flinching or paw movement; 2, fast flinching with paw shaking; 3, fast flinching, biting and paw remaining off the ground. | |||
| Cold algesia (tail) | Animals were placed individually in the test chamber for 60 minutes prior to testing. | ||
| Half of the length of the tail was dipped into the cold water, and the latency to tail withdrawal was measured. | |||
| A maximum cut-off of 30 seconds was set to avoid tissue damage. |
| Method | Protocol | Reference |
|---|---|---|
| Grip Force assay | The mice grip a metal bar attached to a Grip Strength Meter (Stoelting Co., Wood Dale, IL, USA) with their forepaws. | |
| The mice are slowly pulled back by the tail, exerting a stretching force. | ||
| The peak force in grams at the point of release is recorded twice at a 10 minutes interval. | ||
| A decrease in grip force is interpreted as a measure of hypersensitivity to axial stretching. | ||
| Tail suspension | Mice are suspended individually underneath a platform by the tail with adhesive tape attached 0.5 to 1 cm from the base of the tail and are videotaped for 180 seconds. | |
| The duration of time spent in (a) immobility (not moving but stretched out) and (b) escape behaviours (rearing to reach the underside of the platform, extending to reach the floor, or self-supported at the base of the tail or the suspension tape) are determined. | ||
| The duration of immobility reflects the animal’s willingness to stretch its main body axis. | ||
| Deceased immobility is indicative of axial discomfort. | ||
| FlexMaze assay | The FlexMaze apparatus consists of a long (8 cm × 80 cm) transparent corridor with regularly spaced staggered doors and neutral (beige) 15 cm × 15 cm compartments with 6 cm × 6 cm openings on either side | |
| The FlexMaze apparatus is placed in a quiet room illuminated with white light. | ||
| Mice are placed into one of the neutral compartments and are allowed to explore the apparatus freely for 10 minutes. | ||
| Videotapes are analyzed for total distance covered and average velocity. |
Additional Table 5 Movement-evoked hypersensitivity measurement in rodent model
| Method | Protocol | Reference |
|---|---|---|
| Grip Force assay | The mice grip a metal bar attached to a Grip Strength Meter (Stoelting Co., Wood Dale, IL, USA) with their forepaws. | |
| The mice are slowly pulled back by the tail, exerting a stretching force. | ||
| The peak force in grams at the point of release is recorded twice at a 10 minutes interval. | ||
| A decrease in grip force is interpreted as a measure of hypersensitivity to axial stretching. | ||
| Tail suspension | Mice are suspended individually underneath a platform by the tail with adhesive tape attached 0.5 to 1 cm from the base of the tail and are videotaped for 180 seconds. | |
| The duration of time spent in (a) immobility (not moving but stretched out) and (b) escape behaviours (rearing to reach the underside of the platform, extending to reach the floor, or self-supported at the base of the tail or the suspension tape) are determined. | ||
| The duration of immobility reflects the animal’s willingness to stretch its main body axis. | ||
| Deceased immobility is indicative of axial discomfort. | ||
| FlexMaze assay | The FlexMaze apparatus consists of a long (8 cm × 80 cm) transparent corridor with regularly spaced staggered doors and neutral (beige) 15 cm × 15 cm compartments with 6 cm × 6 cm openings on either side | |
| The FlexMaze apparatus is placed in a quiet room illuminated with white light. | ||
| Mice are placed into one of the neutral compartments and are allowed to explore the apparatus freely for 10 minutes. | ||
| Videotapes are analyzed for total distance covered and average velocity. |
| Grade | Structure | Distinction of nucleus and annulus | Signal intensity | Height of intervertebral disc |
|---|---|---|---|---|
| I | Homogeneous, bright white | Clear | Hyperintense, isointense to cerebrospinal fluid | Normal |
| II | Inhomogeneous with or without horizontal bands | Clear | Hyperintense, isointense to cerebrospinal fluid | Normal |
| III | Inhomogeneous, gray | Unclear | Intermediate | Normal to slightly decreased |
| IV | Inhomogeneous, gray to black | Lost | Intermediate to hypointense | Normal to moderately decreased |
| V | Inhomogeneous, black | Lost | Hypointense | Collapsed disc space |
Additional Table 6 Pfirrmann et al.’s classification of disc degeneration
| Grade | Structure | Distinction of nucleus and annulus | Signal intensity | Height of intervertebral disc |
|---|---|---|---|---|
| I | Homogeneous, bright white | Clear | Hyperintense, isointense to cerebrospinal fluid | Normal |
| II | Inhomogeneous with or without horizontal bands | Clear | Hyperintense, isointense to cerebrospinal fluid | Normal |
| III | Inhomogeneous, gray | Unclear | Intermediate | Normal to slightly decreased |
| IV | Inhomogeneous, gray to black | Lost | Intermediate to hypointense | Normal to moderately decreased |
| V | Inhomogeneous, black | Lost | Hypointense | Collapsed disc space |
| Grade | Annulus fibrosus | Nucleus pulposus |
|---|---|---|
| 0 | Normal structure | Normal structure |
| 1 | Mildly serpentine appearance of the annulus fibrosus | No proliferative connective tissue but a honey-comb appearance of the extracellular matrix |
| 2 | Moderately serpentine appearance of the annulus fibrosus with rupture | As much as 24% of the nucleus pulposus occupied by proliferative connective tissue |
| 3 | Severely serpentine appearance of the annulus fibrosus with mildly reversed contour | 25% to 50% of the nucleus pulposus occupied by proliferative connective tissue |
| 4 | Severely reversed contour | More than 50% occupied by proliferative connective tissue |
| 5 | Indistinct | Complete replacement of normal architecture by proliferative connective tissue |
Additional Table 7 Nomura et al.’s histological grading system
| Grade | Annulus fibrosus | Nucleus pulposus |
|---|---|---|
| 0 | Normal structure | Normal structure |
| 1 | Mildly serpentine appearance of the annulus fibrosus | No proliferative connective tissue but a honey-comb appearance of the extracellular matrix |
| 2 | Moderately serpentine appearance of the annulus fibrosus with rupture | As much as 24% of the nucleus pulposus occupied by proliferative connective tissue |
| 3 | Severely serpentine appearance of the annulus fibrosus with mildly reversed contour | 25% to 50% of the nucleus pulposus occupied by proliferative connective tissue |
| 4 | Severely reversed contour | More than 50% occupied by proliferative connective tissue |
| 5 | Indistinct | Complete replacement of normal architecture by proliferative connective tissue |
| Grade | Structure | Scale |
|---|---|---|
| I | Annulus fibrosus | 1. Normal pattern of fibrocartilage lamellae (U-shaped in the posterior aspect and slightly convex in the anterior aspect), without ruptured fibers and a serpentine appearance anywhere within the annulus |
| 2. Ruptured or serpentine patterned fibers in less than 30% of the annulus | ||
| 3. Ruptured or serpentine patterned fibers in more than 30% of the annulus | ||
| II | Border between the annulus fibrosus and nucleus pulposus | 1. Normal |
| 2. Minimal interruption | ||
| 3. Moderate or severe interruption | ||
| III | Cellularity of the nucleus pulposus | 1. Normal cellularity with large vacuoles in the gelatinous structure of the matrix |
| 2. Slight decrease in the number of cells and fewer vacuoles | ||
| 3. Moderate/severe decrease (> 50%) in the number of cells and no vacuoles | ||
| IV | Morphology of the nucleus pulposus | 1. Normal gelatinous appearance |
| 2. Slight condensation of the extracellular matrix | ||
| 3. Moderate/severe condensation of the extracellular matrix |
Additional Table 8 Masuda et al.’s histological grading scale
| Grade | Structure | Scale |
|---|---|---|
| I | Annulus fibrosus | 1. Normal pattern of fibrocartilage lamellae (U-shaped in the posterior aspect and slightly convex in the anterior aspect), without ruptured fibers and a serpentine appearance anywhere within the annulus |
| 2. Ruptured or serpentine patterned fibers in less than 30% of the annulus | ||
| 3. Ruptured or serpentine patterned fibers in more than 30% of the annulus | ||
| II | Border between the annulus fibrosus and nucleus pulposus | 1. Normal |
| 2. Minimal interruption | ||
| 3. Moderate or severe interruption | ||
| III | Cellularity of the nucleus pulposus | 1. Normal cellularity with large vacuoles in the gelatinous structure of the matrix |
| 2. Slight decrease in the number of cells and fewer vacuoles | ||
| 3. Moderate/severe decrease (> 50%) in the number of cells and no vacuoles | ||
| IV | Morphology of the nucleus pulposus | 1. Normal gelatinous appearance |
| 2. Slight condensation of the extracellular matrix | ||
| 3. Moderate/severe condensation of the extracellular matrix |
| Grade | Structure | Scale |
|---|---|---|
| I | Cellularity of the annulus fibrosus | 1. Fibroblasts comprise more than 75% of the cells |
| 2. Neither fibroblasts nor chondrocytes comprise more than 75% of the cells | ||
| 3. Chondrocytes comprise more than 75% of the cells | ||
| II | Morphology of the annulus fibrosus | 1. Well-organized collagen lamellae without ruptured or serpentine fibers |
| 2. Inward bulging, ruptured or serpentine fibers in less than one-third of the annulus | ||
| 3. Inward bulging, ruptured or serpentine fibers in more than one-third of the annulus | ||
| III | Border between the annulus fibrosus and nucleus pulposus | 1. Normal, without any interruption |
| 2. Minimal interruption | ||
| 3. Moderate or severe interruption | ||
| IV | Cellularity of the nucleus pulposus | 1. Normal cellularity with stellar shaped nuclear cells evenly distributed throughout the nucleus |
| 2. Slight decrease in the number of cells with some clustering | ||
| 3. Moderate or severe decrease (> 50%) in the number of cells with all remaining cells clustered and separated by dense areas of proteoglycans | ||
| V | Morphology of the nucleus pulposus | 1. Round, comprising at least half of the disc area in midsagittal sections |
| 2. Rounded or irregularly shaped, comprising one-quarter to half of the disc area in midsagittal sections | ||
| 3. Irregularly shaped, comprising less than one-quarter of the disc area in midsagittal sections |
Additional Table 9 Han et al.’s histological grading scale
| Grade | Structure | Scale |
|---|---|---|
| I | Cellularity of the annulus fibrosus | 1. Fibroblasts comprise more than 75% of the cells |
| 2. Neither fibroblasts nor chondrocytes comprise more than 75% of the cells | ||
| 3. Chondrocytes comprise more than 75% of the cells | ||
| II | Morphology of the annulus fibrosus | 1. Well-organized collagen lamellae without ruptured or serpentine fibers |
| 2. Inward bulging, ruptured or serpentine fibers in less than one-third of the annulus | ||
| 3. Inward bulging, ruptured or serpentine fibers in more than one-third of the annulus | ||
| III | Border between the annulus fibrosus and nucleus pulposus | 1. Normal, without any interruption |
| 2. Minimal interruption | ||
| 3. Moderate or severe interruption | ||
| IV | Cellularity of the nucleus pulposus | 1. Normal cellularity with stellar shaped nuclear cells evenly distributed throughout the nucleus |
| 2. Slight decrease in the number of cells with some clustering | ||
| 3. Moderate or severe decrease (> 50%) in the number of cells with all remaining cells clustered and separated by dense areas of proteoglycans | ||
| V | Morphology of the nucleus pulposus | 1. Round, comprising at least half of the disc area in midsagittal sections |
| 2. Rounded or irregularly shaped, comprising one-quarter to half of the disc area in midsagittal sections | ||
| 3. Irregularly shaped, comprising less than one-quarter of the disc area in midsagittal sections |
| Grade | Nucleus | Annulus | Endplate | Vertebral body |
|---|---|---|---|---|
| I | Bulging gel | Annulus | Hyaline, uniformly thick | Margins rounded |
| II | White fibrous tissue peripherally | Discrete fibrosus lamellas | Thickness irregular | Margins pointed |
| III | Consolidated fibrous tissue | Mucinous material between lamellas | Focal defects in cartilage | Early chondrophytes or osteophytes at margins |
| IV | Horizontal clefts parallel to endplate | Extensive mucinous infiltration; loss of annular-nuclear demarcation | Fibrocartilage extending from subchondral bone; irregularity and focal sclerosis in subchondral bone | Osteophytes less than 2 mm |
| V | Clefts extend through nucleus and annulus | Focal disruptions | Diffuse sclerosis | Osteophytes greater than 2 mm |
Additional Table 10. Thompson et al.’s description of morphologic grades
| Grade | Nucleus | Annulus | Endplate | Vertebral body |
|---|---|---|---|---|
| I | Bulging gel | Annulus | Hyaline, uniformly thick | Margins rounded |
| II | White fibrous tissue peripherally | Discrete fibrosus lamellas | Thickness irregular | Margins pointed |
| III | Consolidated fibrous tissue | Mucinous material between lamellas | Focal defects in cartilage | Early chondrophytes or osteophytes at margins |
| IV | Horizontal clefts parallel to endplate | Extensive mucinous infiltration; loss of annular-nuclear demarcation | Fibrocartilage extending from subchondral bone; irregularity and focal sclerosis in subchondral bone | Osteophytes less than 2 mm |
| V | Clefts extend through nucleus and annulus | Focal disruptions | Diffuse sclerosis | Osteophytes greater than 2 mm |
| Global disc appearance | Grade |
|---|---|
| Macroscopic assessment IVD, endplate, and adjacent bone) | Grade 1 = normal juvenile disc; Grade 2 = normal adult disc; Grade 3 = mild disc degeneration; Grade 4 = moderate disc degeneration; Grade 5 = severe disc degeneration |
| IVD | |
| Cells (chondrocyte proliferation) | 0 = no proliferation; 1 = increased cell density; 2 = connection of two chondrocytes; 3 = small size clones (several chondrocytes, grouped together, 3﹣7 cells); 4 = moderate size clones (8﹣15 cells); 5 = huge clones (> 15 cells) |
| Multiple chondrocytes growing in small, rounded groups or clusters sharply demarcated by a rim of territorial matrix | |
| Granular changes | 0 = absent; 1 = rarely present; 2 = present in intermediate amounts of 1 to 3; 3 = abundantly present |
| Eosinophilic-staining amorphous granules within the fibrocartilage matrix | |
| Mucous degeneration | 0 = absent; 1 = rarely present; 2 = present in intermediate amounts of 1 to 3; 3 = abundantly present |
| Cystic, oval, or irregular areas with an intense deposition of acid mucopolysaccharides (i.e., sulfated glycosaminoglycans) staining dark blue with Alcian blue/PAS | |
| Edge neovascularity | 0 = absent; 1 = rarely present; 2 = present in intermediate amounts of 1 to 3; 3 = abundantly present |
| Newly formed blood vessels with reparative alteration | |
| Rim lesions | 0 = absent; 1 = rarely present; 2 = present in intermediate amounts of 1 to 3; 3 = abundantly present |
| Radial tears adjacent to the endplates | |
| Concentric tears | 0 = absent; 1 = rarely present; 2 = present in intermediate amounts of 1 to 3; 3 = abundantly present |
| Tears after the orientation of collagen fiber bundles in the annulus fibrosus | |
| Radial tears | 0 = absent; 1 = rarely present; 2 = present in intermediate amounts of 1 to 3; 3 = abundantly present |
| Radiating defects extending from the nucleus pulposus to the outer annulus lamellae parallel or oblique to the endplate (clefts) | |
| Notochordal cells | 0 = absent; 1 = present |
| Embryonic disc cells | |
| Cell death | 0 = absent; 1 = rarely present; 2 = present in intermediate amounts of 1 to 3; 3 = abundantly present |
| Altered phenotype | |
| Scar formation | 0 = absent; 1 = present |
| Amorphous fibrosus tissue without any differentiation | |
| Tissue defects | 0 = absent; 1 = present |
| Voids within the tissue (e.g., resulting from tissue resorption, probably filled with fluid in vivo) | |
| Endplate | |
| Cells | 0 = absent; 1 = rarely present; 2 = present in intermediate amounts of 1 to 3; 3 = abundantly present |
| Number of cells (chondrocyte clusters) | |
| Structural disorganization | 0 = absent; 1 = rarely present; 2 = present in intermediate amounts of 1 to 3; 3 = abundantly present |
| Focal disorganization of the cartilaginous matrix with clumping of chondrocytes | |
| Clefts | 0 = absent; 1 = rarely present; 2 = present in intermediate amounts of 1 to 3; 3 = abundantly present |
| Tears in the endplate | |
| Microfracture | 0 = absent; 1 = rarely present; 2 = present in intermediate amounts of 1 to 3; 3 = abundantly present |
| Disruption of the subchondral bone | |
| Neovascularization | 0 = absent; 1 = rarely present; 2 = present in intermediate amounts of 1 to 3; 3 = abundantly present |
| Vessels penetrating from the bone marrow into the endplate in conjunction with microfractures | |
| New bone formation | 0 = absent; 1 = present |
| Bone islands within the cartilage | |
| Bony sclerosis | 0 = absent; 1 = present |
| Formation of new bone | |
| Physiologic vessels | 0 = absent; 1 = present |
| Obliterated vessels | 0 = absent; 1 = present |
| Scar formation | 0 = absent; 1 = present |
| Amorphous fibrosus tissue without any differentiation | |
| Tissue defects | 0 = absent; 1 = present |
| Voids within the tissue (e.g., resulting from tissue resorption, probably filled with fluid in vivo) |
Additional Table 11. Boos et al.’s variables of macroscopic and histological assessment
| Global disc appearance | Grade |
|---|---|
| Macroscopic assessment IVD, endplate, and adjacent bone) | Grade 1 = normal juvenile disc; Grade 2 = normal adult disc; Grade 3 = mild disc degeneration; Grade 4 = moderate disc degeneration; Grade 5 = severe disc degeneration |
| IVD | |
| Cells (chondrocyte proliferation) | 0 = no proliferation; 1 = increased cell density; 2 = connection of two chondrocytes; 3 = small size clones (several chondrocytes, grouped together, 3﹣7 cells); 4 = moderate size clones (8﹣15 cells); 5 = huge clones (> 15 cells) |
| Multiple chondrocytes growing in small, rounded groups or clusters sharply demarcated by a rim of territorial matrix | |
| Granular changes | 0 = absent; 1 = rarely present; 2 = present in intermediate amounts of 1 to 3; 3 = abundantly present |
| Eosinophilic-staining amorphous granules within the fibrocartilage matrix | |
| Mucous degeneration | 0 = absent; 1 = rarely present; 2 = present in intermediate amounts of 1 to 3; 3 = abundantly present |
| Cystic, oval, or irregular areas with an intense deposition of acid mucopolysaccharides (i.e., sulfated glycosaminoglycans) staining dark blue with Alcian blue/PAS | |
| Edge neovascularity | 0 = absent; 1 = rarely present; 2 = present in intermediate amounts of 1 to 3; 3 = abundantly present |
| Newly formed blood vessels with reparative alteration | |
| Rim lesions | 0 = absent; 1 = rarely present; 2 = present in intermediate amounts of 1 to 3; 3 = abundantly present |
| Radial tears adjacent to the endplates | |
| Concentric tears | 0 = absent; 1 = rarely present; 2 = present in intermediate amounts of 1 to 3; 3 = abundantly present |
| Tears after the orientation of collagen fiber bundles in the annulus fibrosus | |
| Radial tears | 0 = absent; 1 = rarely present; 2 = present in intermediate amounts of 1 to 3; 3 = abundantly present |
| Radiating defects extending from the nucleus pulposus to the outer annulus lamellae parallel or oblique to the endplate (clefts) | |
| Notochordal cells | 0 = absent; 1 = present |
| Embryonic disc cells | |
| Cell death | 0 = absent; 1 = rarely present; 2 = present in intermediate amounts of 1 to 3; 3 = abundantly present |
| Altered phenotype | |
| Scar formation | 0 = absent; 1 = present |
| Amorphous fibrosus tissue without any differentiation | |
| Tissue defects | 0 = absent; 1 = present |
| Voids within the tissue (e.g., resulting from tissue resorption, probably filled with fluid in vivo) | |
| Endplate | |
| Cells | 0 = absent; 1 = rarely present; 2 = present in intermediate amounts of 1 to 3; 3 = abundantly present |
| Number of cells (chondrocyte clusters) | |
| Structural disorganization | 0 = absent; 1 = rarely present; 2 = present in intermediate amounts of 1 to 3; 3 = abundantly present |
| Focal disorganization of the cartilaginous matrix with clumping of chondrocytes | |
| Clefts | 0 = absent; 1 = rarely present; 2 = present in intermediate amounts of 1 to 3; 3 = abundantly present |
| Tears in the endplate | |
| Microfracture | 0 = absent; 1 = rarely present; 2 = present in intermediate amounts of 1 to 3; 3 = abundantly present |
| Disruption of the subchondral bone | |
| Neovascularization | 0 = absent; 1 = rarely present; 2 = present in intermediate amounts of 1 to 3; 3 = abundantly present |
| Vessels penetrating from the bone marrow into the endplate in conjunction with microfractures | |
| New bone formation | 0 = absent; 1 = present |
| Bone islands within the cartilage | |
| Bony sclerosis | 0 = absent; 1 = present |
| Formation of new bone | |
| Physiologic vessels | 0 = absent; 1 = present |
| Obliterated vessels | 0 = absent; 1 = present |
| Scar formation | 0 = absent; 1 = present |
| Amorphous fibrosus tissue without any differentiation | |
| Tissue defects | 0 = absent; 1 = present |
| Voids within the tissue (e.g., resulting from tissue resorption, probably filled with fluid in vivo) |
| Criteria | Range |
|---|---|
| Intervertebral disc region | |
| Chondrocyte proliferation/density | 0﹣6 |
| Mucous degeneration | 0﹣4 |
| Cell death | 0﹣4 |
| Tear/cleft formation | 0﹣4 |
| Granular changes | 0﹣4 |
| Vertebral endplate region | |
| Cell proliferation | 0﹣4 |
| Cartilage disorganization | 0﹣4 |
| Cartilage cracks | 0﹣4 |
| Microfracture | 0﹣2 |
| New bone formation | 0﹣2 |
| Bony sclerosis | 0﹣2 |
Additional Table 12. Boyd et al.’s grading for intervertebral disc and endplate regions
| Criteria | Range |
|---|---|
| Intervertebral disc region | |
| Chondrocyte proliferation/density | 0﹣6 |
| Mucous degeneration | 0﹣4 |
| Cell death | 0﹣4 |
| Tear/cleft formation | 0﹣4 |
| Granular changes | 0﹣4 |
| Vertebral endplate region | |
| Cell proliferation | 0﹣4 |
| Cartilage disorganization | 0﹣4 |
| Cartilage cracks | 0﹣4 |
| Microfracture | 0﹣2 |
| New bone formation | 0﹣2 |
| Bony sclerosis | 0﹣2 |
| Method | Protocol | Reference | |
|---|---|---|---|
| In vitro | ASTM F2256-05 (T-Peel by Tension Loading) | At least 10 specimens of each type are to be tested. | |
| Tissue specimen thickness should be uniform and less than 5 mm. | |||
| The specimen width is 2.5 ± 0.1 cm, and the specimen length is 15 ± 0.2 cm (2.5 cm unbonded, 12.5 cm bonded). | |||
| A bond force of 5﹣10 N is applied until the experimental adhesive sets. | |||
| The specimens are conditioned for 1 hour ± 15 minutes in phosphate buffered saline at 37 ± 1°C. | |||
| After conditioning, samples are acclimated to the test temperature for 15 minutes. | |||
| The sample apparatus is loaded into the tensile test machine and at a constant cross-head speed of 250 mm/min. | |||
| The load as a function of displacement and the type of failure (percentage cohesive, adhesive, or substrate failure) are recorded | |||
| ASTM F2258-05 (Tension) | At least ten specimens of each type are to be tested. | ||
| The bond area of 2.5 ± 0.005 cm by 2.5 ± 0.005 cm. | |||
| A bond force of 1﹣2 N is applied until the experimental adhesive sets. | |||
| The specimens are conditioned for 1 hour ± 15 minutes in phosphate buffered saline at 37 ± 1°C. | |||
| After conditioning, samples are acclimated to the test temperature for 15 minutes. | |||
| The sample apparatus is loaded into the tensile test machine and at a constant cross-head speed of 2 mm/min. | |||
| The load at failure (maximum load sustained) and the type of failure (percentage cohesive, adhesive, or substrate failure) are recorded. | |||
| ASTM F2255-05 (Lap-Shear by Tension Loading) | At least 10 specimens of each type are to be tested. | ||
| The length of the tissue substrate attached to each specimen holder should be 1.5 times the length of the bond area (1.0 ± 0.1 cm). | |||
| The tissue specimens should be 1﹣2 mm thick. | |||
| A bond force of 1﹣2 N is placed on the bond area between the two tissue specimens (1.0 ± 0.1 cm by 2.5 ± 0.1 cm) until the experimental adhesive sets. | |||
| The specimens are conditioned for 1 hour ± 15 minutes in phosphate buffered saline at 37 ± 1°C. | |||
| After conditioning, samples are acclimated to the test temperature for 15 minutes. | |||
| The sample is loaded into the testing apparatus such that the load coincides with the long axis of the sample. | |||
| The sample is loaded to failure at a constant crosshead speed of 5 mm/min. The load at failure (maximum load sustained) and the type of failure (percentage cohesive, adhesive, or substrate failure) are recorded. | |||
| ASTM F2458-05 (Wound Closure Strength of Tissue Adhesives and Sealants) | At least ten specimens of each type are to be tested. | ||
| Two tissue samples of identical size (10 ± 0.2 cm by 2.5 ± 0.1 cm) are bonded using the experimental adhesive on the 2.5 cm side, with a bonding length of 0.5 cm on either side of the join line. | |||
| The thickness of the specimens should be uniform and less than 5 mm. | |||
| The specimens are conditioned for 1 hour ± 15 minutes in phosphate buffered saline at 37 ± 1°C. | |||
| After conditioning, samples are acclimated to the test temperature for 15 minutes. | |||
| The sample is loaded into the testing apparatus such that the load coincides with the long axis of the sample. | |||
| The distance from the grip to the midline of each sample is 5 cm, with the remaining 5 cm held between the grips. | |||
| The specimen is loaded to failure at a constant speed of 50 mm/min. | |||
| The time from application to testing (cure time), force at failure (maximum force required to disrupt substrate), and the type of failure (percentage cohesive, adhesive, or substrate failure) are recorded. | |||
| ASTM F2392-04 (Burst Strength of Surgical Sealants) | At least 10 specimens of each type are to be tested. | ||
| This test employs an apparatus that clamps down on a substrate to prevent leakage. | |||
| The thickness of the tissue should be uniform and not exceed 5 mm. | |||
| Tissue samples should be circles 3.0 ± 0.1 cm in diameter, in which a 3.0 mm diameter hole is created using a biopsy punch. | |||
| The specimens are conditioned for 1 hour ± 15 minutes in phosphate buffered saline at 37 ± 1°C. | |||
| After conditioning, samples are acclimated to the test temperature for 15 minutes. | |||
| This test uses a stationary fixture containing test substrate and the sealant to be tested. | |||
| A 1.0 mm thick PTFE mask with a 15 mm diameter hole is secured over the sample, with the hole in the mask centered with the hole in the sample. | |||
| Saline is pumped into the fixture at a constant rate of 2 mL/min, and pressures are measured at all time points. | |||
| Peak pressure and failure type (cohesive, adhesive, or substrate) are recorded. | |||
| Ex vivo (risk of herniation) | Ramp-to-Failure Testing | Herniation risk was evaluated through failure testing using a MTS Bionix Servohydraulic Test System (MTS, Eden Parairie, MN, USA). | |
| Specimens were placed on the MTS stage at an offset of 5° from the normal axis, with the postero-lateral portion of the disc at the outside of the bend to simulate postero-lateral flexion. | |||
| A force of ~20 N was applied as a pre-load. | |||
| The samples were then compressed in displacement-control mode using a ramp function at 2 mm/min until failure. | |||
| Biomechanical output measures that quantitatively describe IVD herniation risk include failure strength, failure strain, subsidence-to-failure, maximum stiffness, work-to-failure, yield strength, ultimate strength, and the ratios of the ultimate or yield strength to the failure strength of the motion segment. | |||
| Fatigue Endurance Testing | The fatigue loading protocol consisted of cyclic eccentric compression between 50 N and 300 N at 1 Hz and at an offset of 20 mm to induce a physiological bending moment of 6 N·m. | ||
| The loading indenter cyclically rotated from -135° to +135° from the axis opposite of the incision site at 15° increments with 1 minute of cyclic loading at each location. | |||
| This test setup was considered to mimic the “worst-case scenario” as loading opposite of the injury site was expected to aggravate NP extrusion. | |||
| Failure was defined by significant NP protrusion greater than 2 mm. | |||
| The main outcome measure from the fatigue tests was cycles-to-failure, which was indicative of fatigue endurance. |
Additional Table 13. Methods for the evaluation of adhesive properties
| Method | Protocol | Reference | |
|---|---|---|---|
| In vitro | ASTM F2256-05 (T-Peel by Tension Loading) | At least 10 specimens of each type are to be tested. | |
| Tissue specimen thickness should be uniform and less than 5 mm. | |||
| The specimen width is 2.5 ± 0.1 cm, and the specimen length is 15 ± 0.2 cm (2.5 cm unbonded, 12.5 cm bonded). | |||
| A bond force of 5﹣10 N is applied until the experimental adhesive sets. | |||
| The specimens are conditioned for 1 hour ± 15 minutes in phosphate buffered saline at 37 ± 1°C. | |||
| After conditioning, samples are acclimated to the test temperature for 15 minutes. | |||
| The sample apparatus is loaded into the tensile test machine and at a constant cross-head speed of 250 mm/min. | |||
| The load as a function of displacement and the type of failure (percentage cohesive, adhesive, or substrate failure) are recorded | |||
| ASTM F2258-05 (Tension) | At least ten specimens of each type are to be tested. | ||
| The bond area of 2.5 ± 0.005 cm by 2.5 ± 0.005 cm. | |||
| A bond force of 1﹣2 N is applied until the experimental adhesive sets. | |||
| The specimens are conditioned for 1 hour ± 15 minutes in phosphate buffered saline at 37 ± 1°C. | |||
| After conditioning, samples are acclimated to the test temperature for 15 minutes. | |||
| The sample apparatus is loaded into the tensile test machine and at a constant cross-head speed of 2 mm/min. | |||
| The load at failure (maximum load sustained) and the type of failure (percentage cohesive, adhesive, or substrate failure) are recorded. | |||
| ASTM F2255-05 (Lap-Shear by Tension Loading) | At least 10 specimens of each type are to be tested. | ||
| The length of the tissue substrate attached to each specimen holder should be 1.5 times the length of the bond area (1.0 ± 0.1 cm). | |||
| The tissue specimens should be 1﹣2 mm thick. | |||
| A bond force of 1﹣2 N is placed on the bond area between the two tissue specimens (1.0 ± 0.1 cm by 2.5 ± 0.1 cm) until the experimental adhesive sets. | |||
| The specimens are conditioned for 1 hour ± 15 minutes in phosphate buffered saline at 37 ± 1°C. | |||
| After conditioning, samples are acclimated to the test temperature for 15 minutes. | |||
| The sample is loaded into the testing apparatus such that the load coincides with the long axis of the sample. | |||
| The sample is loaded to failure at a constant crosshead speed of 5 mm/min. The load at failure (maximum load sustained) and the type of failure (percentage cohesive, adhesive, or substrate failure) are recorded. | |||
| ASTM F2458-05 (Wound Closure Strength of Tissue Adhesives and Sealants) | At least ten specimens of each type are to be tested. | ||
| Two tissue samples of identical size (10 ± 0.2 cm by 2.5 ± 0.1 cm) are bonded using the experimental adhesive on the 2.5 cm side, with a bonding length of 0.5 cm on either side of the join line. | |||
| The thickness of the specimens should be uniform and less than 5 mm. | |||
| The specimens are conditioned for 1 hour ± 15 minutes in phosphate buffered saline at 37 ± 1°C. | |||
| After conditioning, samples are acclimated to the test temperature for 15 minutes. | |||
| The sample is loaded into the testing apparatus such that the load coincides with the long axis of the sample. | |||
| The distance from the grip to the midline of each sample is 5 cm, with the remaining 5 cm held between the grips. | |||
| The specimen is loaded to failure at a constant speed of 50 mm/min. | |||
| The time from application to testing (cure time), force at failure (maximum force required to disrupt substrate), and the type of failure (percentage cohesive, adhesive, or substrate failure) are recorded. | |||
| ASTM F2392-04 (Burst Strength of Surgical Sealants) | At least 10 specimens of each type are to be tested. | ||
| This test employs an apparatus that clamps down on a substrate to prevent leakage. | |||
| The thickness of the tissue should be uniform and not exceed 5 mm. | |||
| Tissue samples should be circles 3.0 ± 0.1 cm in diameter, in which a 3.0 mm diameter hole is created using a biopsy punch. | |||
| The specimens are conditioned for 1 hour ± 15 minutes in phosphate buffered saline at 37 ± 1°C. | |||
| After conditioning, samples are acclimated to the test temperature for 15 minutes. | |||
| This test uses a stationary fixture containing test substrate and the sealant to be tested. | |||
| A 1.0 mm thick PTFE mask with a 15 mm diameter hole is secured over the sample, with the hole in the mask centered with the hole in the sample. | |||
| Saline is pumped into the fixture at a constant rate of 2 mL/min, and pressures are measured at all time points. | |||
| Peak pressure and failure type (cohesive, adhesive, or substrate) are recorded. | |||
| Ex vivo (risk of herniation) | Ramp-to-Failure Testing | Herniation risk was evaluated through failure testing using a MTS Bionix Servohydraulic Test System (MTS, Eden Parairie, MN, USA). | |
| Specimens were placed on the MTS stage at an offset of 5° from the normal axis, with the postero-lateral portion of the disc at the outside of the bend to simulate postero-lateral flexion. | |||
| A force of ~20 N was applied as a pre-load. | |||
| The samples were then compressed in displacement-control mode using a ramp function at 2 mm/min until failure. | |||
| Biomechanical output measures that quantitatively describe IVD herniation risk include failure strength, failure strain, subsidence-to-failure, maximum stiffness, work-to-failure, yield strength, ultimate strength, and the ratios of the ultimate or yield strength to the failure strength of the motion segment. | |||
| Fatigue Endurance Testing | The fatigue loading protocol consisted of cyclic eccentric compression between 50 N and 300 N at 1 Hz and at an offset of 20 mm to induce a physiological bending moment of 6 N·m. | ||
| The loading indenter cyclically rotated from -135° to +135° from the axis opposite of the incision site at 15° increments with 1 minute of cyclic loading at each location. | |||
| This test setup was considered to mimic the “worst-case scenario” as loading opposite of the injury site was expected to aggravate NP extrusion. | |||
| Failure was defined by significant NP protrusion greater than 2 mm. | |||
| The main outcome measure from the fatigue tests was cycles-to-failure, which was indicative of fatigue endurance. |
| Items | Purpose | Reference for protocals |
|---|---|---|
| Adhesion evaluation (in vitro and ex vivo) | To determine the tissue integrating strength after implantation | |
| Tension/compression/shear evaluation (in vitro) | To determine whether the mechanical properties of biomaterials match with that of human tissue | |
| Swelling (in vitro) | To determine whether biomaterials will swelling and its potential damage to surrounding tissue | |
| Gelation kinetics (in vitro) | To determine whether the gelation time is suitable for clinical application | |
| Failure test & fatigue failure test (ex vivo) | To determine the herniation risk under extensive and prolonged mechanical loadings | |
| Biomechanics test (ex vivo) | To determine whether biomaterials will maintain the motion segment biomechanics | |
| Compressive/torsional/tensile stiffness; creep displacement; torque range; axial range of motion (ex vivo) | To determine the biomechanical reparative effects of implanted biomaterials |
Additional Table 14. A paradigm for testing intervertebral disc mechanical properties.
| Items | Purpose | Reference for protocals |
|---|---|---|
| Adhesion evaluation (in vitro and ex vivo) | To determine the tissue integrating strength after implantation | |
| Tension/compression/shear evaluation (in vitro) | To determine whether the mechanical properties of biomaterials match with that of human tissue | |
| Swelling (in vitro) | To determine whether biomaterials will swelling and its potential damage to surrounding tissue | |
| Gelation kinetics (in vitro) | To determine whether the gelation time is suitable for clinical application | |
| Failure test & fatigue failure test (ex vivo) | To determine the herniation risk under extensive and prolonged mechanical loadings | |
| Biomechanics test (ex vivo) | To determine whether biomaterials will maintain the motion segment biomechanics | |
| Compressive/torsional/tensile stiffness; creep displacement; torque range; axial range of motion (ex vivo) | To determine the biomechanical reparative effects of implanted biomaterials |
| Parameter | Recommended value |
|---|---|
| Disc pressure, after implantation | 1.50 MPa |
| Disc pressure, maximal (till failure) | 2.30 MPa |
| Tensile modulus, axial | 0.5-1 MPa |
| Compressive/tensile strain | 28%/65% |
| Axial stiffness of restored intervertebral disc | 1.5-2 kN/mm |
| Torsional stiffness of restored intervertebral disc | 3.2 N·m/° |
| Tensile modulus, circumferential | 11-29 MPa |
| Aggregate modulus | 0.4-6 MPa |
| Shear modulus | 0.1-0.28 MPa |
Additional Table 15. Recommended parameters for disc regeneration
| Parameter | Recommended value |
|---|---|
| Disc pressure, after implantation | 1.50 MPa |
| Disc pressure, maximal (till failure) | 2.30 MPa |
| Tensile modulus, axial | 0.5-1 MPa |
| Compressive/tensile strain | 28%/65% |
| Axial stiffness of restored intervertebral disc | 1.5-2 kN/mm |
| Torsional stiffness of restored intervertebral disc | 3.2 N·m/° |
| Tensile modulus, circumferential | 11-29 MPa |
| Aggregate modulus | 0.4-6 MPa |
| Shear modulus | 0.1-0.28 MPa |
| 1. |
Cieza, A.; Causey, K.; Kamenov, K.; Hanson, S. W.; Chatterji, S.; Vos, T. Global estimates of the need for rehabilitation based on the Global Burden of Disease study 2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet. 2021, 396, 2006-2017.
doi: 10.1016/S0140-6736(20)32340-0 URL |
| 2. |
Cheung, K. M.; Samartzis, D.; Karppinen, J.; Luk, K. D. Are “patterns” of lumbar disc degeneration associated with low back pain?: new insights based on skipped level disc pathology. Spine (Phila Pa 1976). 2012, 37, E430-438.
doi: 10.1097/BRS.0b013e3182304dfc URL |
| 3. |
Hunter, D. J.; Bierma-Zeinstra, S. Osteoarthritis. Lancet. 2019, 393, 1745-1759.
doi: 10.1016/S0140-6736(19)30417-9 URL |
| 4. | Guevar, J.; Olby, N. Minimally invasive microsurgical decompression of an intervertebral disc protrusion in a dog. Vet Surg. 2020, 49 Suppl 1, O86-O92. |
| 5. |
Geisler, F. H.; McAfee, P. C.; Banco, R. J.; Blumenthal, S. L.; Guyer, R. D.; Holt, R. T.; Majd, M. E. Prospective, randomized, multicenter FDA IDE study of CHARITÉ artificial disc versus lumbar fusion: effect at 5-year follow-up of prior surgery and prior discectomy on clinical outcomes following lumbar arthroplasty. SAS J. 2009, 3, 17-25.
doi: 10.1016/S1935-9810(09)70003-9 URL |
| 6. |
Zou, S.; Gao, J.; Xu, B.; Lu, X.; Han, Y.; Meng, H. Anterior cervical discectomy and fusion (ACDF) versus cervical disc arthroplasty (CDA) for two contiguous levels cervical disc degenerative disease: a meta-analysis of randomized controlled trials. Eur Spine J. 2017, 26, 985-997.
doi: 10.1007/s00586-016-4655-5 URL |
| 7. |
Parker, S. L.; Mendenhall, S. K.; Godil, S. S.; Sivasubramanian, P.; Cahill, K.; Ziewacz, J.; McGirt, M. J. Incidence of low back pain after lumbar discectomy for herniated disc and its effect on patient-reported outcomes. Clin Orthop Relat Res. 2015, 473, 1988-1999.
doi: 10.1007/s11999-015-4193-1 URL |
| 8. |
Hilibrand, A. S.; Robbins, M. Adjacent segment degeneration and adjacent segment disease: the consequences of spinal fusion? Spine J. 2004, 4, 190s-194s.
doi: 10.1016/j.spinee.2004.07.007 URL |
| 9. |
Mukherjee, D.; Nissen, S. E.; Topol, E. J. Risk of cardiovascular events associated with selective COX-2 inhibitors. JAMA. 2001, 286, 954-959.
doi: 10.1001/jama.286.8.954 URL |
| 10. | Rosenzweig, D. H.; Fairag, R.; Mathieu, A. P.; Li, L.; Eglin, D.; D’Este, M.; Steffen, T.; Weber, M. H.; Ouellet, J. A.; Haglund, L. Thermoreversible hyaluronan-hydrogel and autologous nucleus pulposus cell delivery regenerates human intervertebral discs in an ex vivo, physiological organ culture model. Eur Cell Mater. 2018, 36, 200-217. |
| 11. |
Noriega, D. C.; Ardura, F.; Hernández-Ramajo, R.; Martín-Ferrero, M.; Sánchez-Lite, I.; Toribio, B.; Alberca, M.; García, V.; Moraleda, J. M.; Sánchez, A.; García-Sancho, J. Intervertebral disc repair by allogeneic mesenchymal bone marrow cells: a randomized controlled trial. Transplantation. 2017, 101, 1945-1951.
doi: 10.1097/TP.0000000000001484 URL |
| 12. |
Orozco, L.; Soler, R.; Morera, C.; Alberca, M.; Sánchez, A.; García-Sancho, J. Intervertebral disc repair by autologous mesenchymal bone marrow cells: a pilot study. Transplantation. 2011, 92, 822-828.
doi: 10.1097/TP.0b013e3182298a15 URL |
| 13. |
Pettine, K.; Suzuki, R.; Sand, T.; Murphy, M. Treatment of discogenic back pain with autologous bone marrow concentrate injection with minimum two year follow-up. Int Orthop. 2016, 40, 135-140.
doi: 10.1007/s00264-015-2886-4 URL |
| 14. |
Pettine, K. A.; Suzuki, R. K.; Sand, T. T.; Murphy, M. B. Autologous bone marrow concentrate intradiscal injection for the treatment of degenerative disc disease with three-year follow-up. Int Orthop. 2017, 41, 2097-2103.
doi: 10.1007/s00264-017-3560-9 URL |
| 15. |
Elabd, C.; Centeno, C. J.; Schultz, J. R.; Lutz, G.; Ichim, T.; Silva, F. J. Intra-discal injection of autologous, hypoxic cultured bone marrow-derived mesenchymal stem cells in five patients with chronic lower back pain: a long-term safety and feasibility study. J Transl Med. 2016, 14, 253.
doi: 10.1186/s12967-016-1015-5 URL |
| 16. |
Centeno, C.; Markle, J.; Dodson, E.; Stemper, I.; Williams, C. J.; Hyzy, M.; Ichim, T.; Freeman, M. Treatment of lumbar degenerative disc disease-associated radicular pain with culture-expanded autologous mesenchymal stem cells: a pilot study on safety and efficacy. J Transl Med. 2017, 15, 197.
doi: 10.1186/s12967-017-1300-y URL |
| 17. |
Laslett, M.; Oberg, B.; Aprill, C. N.; McDonald, B. Centralization as a predictor of provocation discography results in chronic low back pain, and the influence of disability and distress on diagnostic power. Spine J. 2005, 5, 370-380.
doi: 10.1016/j.spinee.2004.11.007 URL |
| 18. |
Pettine, K. A.; Murphy, M. B.; Suzuki, R. K.; Sand, T. T. Percutaneous injection of autologous bone marrow concentrate cells significantly reduces lumbar discogenic pain through 12 months. Stem Cells. 2015, 33, 146-156.
doi: 10.1002/stem.1845 URL |
| 19. |
Fayad, F.; Lefevre-Colau, M. M.; Rannou, F.; Quintero, N.; Nys, A.; Macé, Y.; Poiraudeau, S.; Drapé, J. L.; Revel, M. Relation of inflammatory modic changes to intradiscal steroid injection outcome in chronic low back pain. Eur Spine J. 2007, 16, 925-931.
doi: 10.1007/s00586-006-0301-y URL |
| 20. |
Bedard, N. A.; Pugely, A. J.; Elkins, J. M.; Duchman, K. R.; Westermann, R. W.; Liu, S. S.; Gao, Y.; Callaghan, J. J. The John N. Insall award: do Intraarticular injections increase the risk of infection after TKA? Clin Orthop Relat Res. 2017, 475, 45-52.
doi: 10.1007/s11999-016-4757-8 URL |
| 21. |
Chambers, A. W.; Lacy, K. W.; Liow, M. H. L.; Manalo, J. P. M.; Freiberg, A. A.; Kwon, Y. M. Multiple hip intra-articular steroid injections increase risk of periprosthetic joint infection compared with single injections. J Arthroplasty. 2017, 32, 1980-1983.
doi: 10.1016/j.arth.2017.01.030 URL |
| 22. |
Pal, B.; Morris, J. Perceived risks of joint infection following intra-articular corticosteroid injections: a survey of rheumatologists. Clin Rheumatol. 1999, 18, 264-265.
doi: 10.1007/s100670050098 URL |
| 23. |
Teixeira, G. Q.; Leite Pereira, C.; Castro, F.; Ferreira, J. R.; Gomez-Lazaro, M.; Aguiar, P.; Barbosa, M. A.; Neidlinger-Wilke, C.; Goncalves, R. M. Anti-inflammatory chitosan/poly-γ-glutamic acid nanoparticles control inflammation while remodeling extracellular matrix in degenerated intervertebral disc. Acta Biomater. 2016, 42, 168-179.
doi: 10.1016/j.actbio.2016.06.013 URL |
| 24. |
Zhang, H.; Yu, S.; Zhao, X.; Mao, Z.; Gao, C. Stromal cell-derived factor-1α-encapsulated albumin/heparin nanoparticles for induced stem cell migration and intervertebral disc regeneration in vivo. Acta Biomater. 2018, 72, 217-227.
doi: 10.1016/j.actbio.2018.03.032 URL |
| 25. |
Bowles, R. D.; Setton, L. A. Biomaterials for intervertebral disc regeneration and repair. Biomaterials. 2017, 129, 54-67.
doi: 10.1016/j.biomaterials.2017.03.013 URL |
| 26. |
Harmon, M. D.; Ramos, D. M.; Nithyadevi, D.; Bordett, R.; Rudraiah, S.; Nukavarapu, S. P.; Moss, I. L.; Kumbar, S. G. Growing a backbone - functional biomaterials and structures for intervertebral disc (IVD) repair and regeneration: challenges, innovations, and future directions. Biomater Sci. 2020, 8, 1216-1239.
doi: 10.1039/C9BM01288E URL |
| 27. |
Virk, S.; Chen, T.; Meyers, K. N.; Lafage, V.; Schwab, F.; Maher, S. A. Comparison of biomechanical studies of disc repair devices based on a systematic review . Spine J. 2020, 20, 1344-1355.
doi: 10.1016/j.spinee.2020.02.007 URL |
| 28. |
Long, R. G.; Torre, O. M.; Hom, W. W.; Assael, D. J.; Iatridis, J. C. Design requirements for annulus fibrosus repair: review of forces, displacements, and material properties of the intervertebral disk and a summary of candidate hydrogels for repair. J Biomech Eng. 2016, 138, 021007.
doi: 10.1115/1.4032353 URL |
| 29. |
Krock, E.; Millecamps, M.; Anderson, K. M.; Srivastava, A.; Reihsen, T. E.; Hari, P.; Sun, Y. R.; Jang, S. H.; Wilcox, G. L.; Belani, K. G.; Beebe, D. S.; Ouellet, J.; Pinto, M. R.; Kehl, L. J.; Haglund, L.; Stone, L. S. Interleukin-8 as a therapeutic target for chronic low back pain: Upregulation in human cerebrospinal fluid and pre-clinical validation with chronic reparixin in the SPARC-null mouse model. EBioMedicine. 2019, 43, 487-500.
doi: 10.1016/j.ebiom.2019.04.032 URL |
| 30. |
Millecamps, M.; Czerminski, J. T.; Mathieu, A. P.; Stone, L. S. Behavioral signs of axial low back pain and motor impairment correlate with the severity of intervertebral disc degeneration in a mouse model. Spine J. 2015, 15, 2524-2537.
doi: 10.1016/j.spinee.2015.08.055 URL |
| 31. |
Bian, Q.; Ma, L.; Jain, A.; Crane, J. L.; Kebaish, K.; Wan, M.; Zhang, Z.; Edward Guo, X.; Sponseller, P. D.; Séguin, C. A.; Riley, L. H.; Wang, Y.; Cao, X. Mechanosignaling activation of TGFβ maintains intervertebral disc homeostasis. Bone Res. 2017, 5, 17008.
doi: 10.1038/boneres.2017.8 URL |
| 32. | Huang, D.; Peng, Y.; Ma, K.; Qing, X.; Deng, X.; Li, Z.; Shao, Z. Puerarin relieved compression-induced apoptosis and mitochondrial dysfunction in human nucleus pulposus mesenchymal stem cells via the PI3K/Akt pathway. Stem Cells Int. 2020, 2020, 7126914. |
| 33. |
Sekiguchi, M.; Konno, S.; Kikuchi, S. The effects of a 5-HT2A receptor antagonist on blood flow in lumbar disc herniation: application of nucleus pulposus in a canine model. Eur Spine J. 2008, 17, 307-313.
doi: 10.1007/s00586-007-0534-4 URL |
| 34. |
Bailey, J. F.; Fields, A. J.; Liebenberg, E.; Mattison, J. A.; Lotz, J. C.; Kramer, P. A. Comparison of vertebral and intervertebral disc lesions in aging humans and rhesus monkeys. Osteoarthritis Cartilage. 2014, 22, 980-985.
doi: 10.1016/j.joca.2014.04.027 URL |
| 35. |
Beckstein, J. C.; Sen, S.; Schaer, T. P.; Vresilovic, E. J.; Elliott, D. M. Comparison of animal discs used in disc research to human lumbar disc: axial compression mechanics and glycosaminoglycan content. Spine (Phila Pa 1976). 2008, 33, E166-173.
doi: 10.1097/BRS.0b013e318166e001 URL |
| 36. | Weiler, C.; Schietzsch, M.; Kirchner, T.; Nerlich, A. G.; Boos, N.; Wuertz, K. Age-related changes in human cervical, thoracal and lumbar intervertebral disc exhibit a strong intra-individual correlation. Eur Spine J. 2012, 21 Suppl 6, S810-818. |
| 37. |
Robinson, W. A.; Nassr, A. N.; Sebastian, A. S. Thoracic disc herniation, avoidance, and management of the surgical complications. Int Orthop. 2019, 43, 817-823.
doi: 10.1007/s00264-018-04282-x URL |
| 38. |
Colombier, P.; Camus, A.; Lescaudron, L.; Clouet, J.; Guicheux, J. Intervertebral disc regeneration: a great challenge for tissue engineers. Trends Biotechnol. 2014, 32, 433-435.
doi: 10.1016/j.tibtech.2014.05.006 URL |
| 39. | Stergar, J.; Gradisnik, L.; Velnar, T.; Maver, U. Intervertebral disc tissue engineering: A brief review. Bosn J Basic Med Sci. 2019, 19, 130-137. |
| 40. |
Chuah, Y. J.; Peck, Y.; Lau, J. E.; Hee, H. T.; Wang, D. A. Hydrogel based cartilaginous tissue regeneration: recent insights and technologies. Biomater Sci. 2017, 5, 613-631.
doi: 10.1039/C6BM00863A URL |
| 41. |
Gruber, H. E.; Hoelscher, G. L.; Leslie, K.; Ingram, J. A.; Hanley, E. N., Jr. Three-dimensional culture of human disc cells within agarose or a collagen sponge: assessment of proteoglycan production. Biomaterials. 2006, 27, 371-376.
doi: 10.1016/j.biomaterials.2005.06.032 URL |
| 42. | Silagi, E. S.; Shapiro, I. M.; Risbud, M. V Glycosaminoglycan synthesis in the nucleus pulposus: Dysregulation and the pathogenesis of disc degeneration. Matrix Biol. 2018, 71-72, 368-379. |
| 43. |
Erwin, W. M.; Ashman, K.; O’Donnel, P.; Inman, R. D. Nucleus pulposus notochord cells secrete connective tissue growth factor and up-regulate proteoglycan expression by intervertebral disc chondrocytes. Arthritis Rheum. 2006, 54, 3859-3867.
doi: 10.1002/(ISSN)1529-0131 URL |
| 44. |
Erwin, W. M.; Islam, D.; Inman, R. D.; Fehlings, M. G.; Tsui, F. W. Notochordal cells protect nucleus pulposus cells from degradation and apoptosis: implications for the mechanisms of intervertebral disc degeneration. Arthritis Res Ther. 2011, 13, R215.
doi: 10.1186/ar3548 URL |
| 45. |
McCann, M. R.; Séguin, C. A. Notochord cells in intervertebral disc development and degeneration. J Dev Biol. 2016, 4, 3.
doi: 10.3390/jdb4010003 URL |
| 46. |
Hu, B.; Zhang, S.; Liu, W.; Wang, P.; Chen, S.; Lv, X.; Shi, D.; Ma, K.; Wang, B.; Wu, Y.; Shao, Z. Inhibiting heat shock protein 90 protects nucleus pulposus-derived stem/progenitor cells from compression-induced necroptosis and apoptosis. Front Cell Dev Biol. 2020, 8, 685.
doi: 10.3389/fcell.2020.00685 URL |
| 47. |
Chen, S.; Lv, X.; Hu, B.; Shao, Z.; Wang, B.; Ma, K.; Lin, H.; Cui, M. RIPK1/RIPK3/MLKL-mediated necroptosis contributes to compression-induced rat nucleus pulposus cells death. Apoptosis. 2017, 22, 626-638.
doi: 10.1007/s10495-017-1358-2 URL |
| 48. | Chen, S.; Liu, S.; Zhao, L.; Lin, H.; Ma, K.; Shao, Z. Heme Oxygenase-1-mediated autophagy protects against oxidative damage in rat nucleus pulposus-derived mesenchymal stem cells. Oxid Med Cell Longev. 2020, 2020, 9349762. |
| 49. | Zhang, Y.; Yang, B.; Wang, J.; Cheng, F.; Shi, K.; Ying, L.; Wang, C.; Xia, K.; Huang, X.; Gong, Z.; Yu, C.; Li, F.; Liang, C.; Chen, Q. Cell senescence: a nonnegligible cell state under survival stress in pathology of intervertebral disc degeneration. Oxid Med Cell Longev. 2020, 2020, 9503562. |
| 50. | Di Micco, R.; Krizhanovsky, V.; Baker, D.; d’Adda di Fagagna, F. Cellular senescence in ageing: from mechanisms to therapeutic opportunities. Nat Rev Mol Cell Biol. 2021, 22, 75-95. |
| 51. | Childs, B. G.; Gluscevic, M.; Baker, D. J.; Laberge, R. M.; Marquess, D.; Dananberg, J.; van Deursen, J. M. Senescent cells: an emerging target for diseases of ageing. Nat Rev Drug Discov. 2017, 16, 718-735. |
| 52. |
Wang, F.; Cai, F.; Shi, R.; Wang, X. H.; Wu, X. T. Aging and age related stresses: a senescence mechanism of intervertebral disc degeneration. Osteoarthritis Cartilage. 2016, 24, 398-408.
doi: 10.1016/j.joca.2015.09.019 URL |
| 53. |
Zou, H.; Stoppani, E.; Volonte, D.; Galbiati, F. Caveolin-1, cellular senescence and age-related diseases. Mech Ageing Dev. 2011, 132, 533-542.
doi: 10.1016/j.mad.2011.11.001 URL |
| 54. |
van Deursen, J. M. The role of senescent cells in ageing. Nature. 2014, 509, 439-446.
doi: 10.1038/nature13193 URL |
| 55. |
Chen, X. J.; Kim, S. R.; Jiang, K.; Ferguson, C. M.; Tang, H.; Zhu, X. Y.; Lerman, A.; Eirin, A.; Lerman, L. O. Renovascular disease induces senescence in renal scattered tubular-like cells and impairs their reparative potency. Hypertension. 2021, 77, 507-518.
doi: 10.1161/HYPERTENSIONAHA.120.16218 URL |
| 56. | Dookun, E.; Walaszczyk, A.; Redgrave, R.; Palmowski, P.; Tual-Chalot, S.; Suwana, A.; Chapman, J.; Jirkovsky, E.; Donastorg Sosa, L.; Gill, E.; Yausep, O. E.; Santin, Y.; Mialet-Perez, J.; Andrew Owens, W.; Grieve, D.; Spyridopoulos, I.; Taggart, M.; Arthur, H. M.; Passos, J. F.; Richardson, G. D. Clearance of senescent cells during cardiac ischemia-reperfusion injury improves recovery. Aging Cell. 2020, 19, e13249. |
| 57. |
Dookun, E.; Passos, J. F.; Arthur, H. M.; Richardson, G. D. Therapeutic Potential of Senolytics in Cardiovascular Disease. Cardiovasc Drugs Ther. 2020. doi: 10.1007/s10557-020-07075-w.
doi: 10.1007/s10557-020-07075-w URL |
| 58. |
Walaszczyk, A.; Dookun, E.; Redgrave, R.; Tual-Chalot, S.; Victorelli, S.; Spyridopoulos, I.; Owens, A.; Arthur, H. M.; Passos, J. F.; Richardson, G. D. Pharmacological clearance of senescent cells improves survival and recovery in aged mice following acute myocardial infarction. Aging Cell. 2019, 18, e12945.
doi: 10.1111/acel.2019.18.issue-3 URL |
| 59. | Ogrodnik, M.; Evans, S. A.; Fielder, E.; Victorelli, S.; Kruger, P.; Salmonowicz, H.; Weigand, B. M.; Patel, A. D.; Pirtskhalava, T.; Inman, C. L.; Johnson, K. O.; Dickinson, S. L.; Rocha, A.; Schafer, M. J.; Zhu, Y.; Allison, D. B.; von Zglinicki, T.; LeBrasseur, N. K.; Tchkonia, T.; Neretti, N.; Passos, J. F.; Kirkland, J. L.; Jurk, D. Whole-body senescent cell clearance alleviates age-related brain inflammation and cognitive impairment in mice. Aging Cell. 2021, 20, e13296. |
| 60. |
Johmura, Y.; Yamanaka, T.; Omori, S.; Wang, T. W.; Sugiura, Y.; Matsumoto, M.; Suzuki, N.; Kumamoto, S.; Yamaguchi, K.; Hatakeyama, S.; Takami, T.; Yamaguchi, R.; Shimizu, E.; Ikeda, K.; Okahashi, N.; Mikawa, R.; Suematsu, M.; Arita, M.; Sugimoto, M.; Nakayama, K. I.; Furukawa, Y.; Imoto, S.; Nakanishi, M. Senolysis by glutaminolysis inhibition ameliorates various age-associated disorders. Science. 2021, 371, 265-270.
doi: 10.1126/science.abb5916 URL |
| 61. |
Hwang, P. Y.; Chen, J.; Jing, L.; Hoffman, B. D.; Setton, L. A. The role of extracellular matrix elasticity and composition in regulating the nucleus pulposus cell phenotype in the intervertebral disc: a narrative review. J Biomech Eng. 2014, 136, 021010.
doi: 10.1115/1.4026360 URL |
| 62. |
Gilbert, H. T. J.; Hodson, N.; Baird, P.; Richardson, S. M.; Hoyland, J. A. Acidic pH promotes intervertebral disc degeneration: Acid-sensing ion channel-3 as a potential therapeutic target. Sci Rep. 2016, 6, 37360.
doi: 10.1038/srep37360 URL |
| 63. |
Quero, L.; Klawitter, M.; Schmaus, A.; Rothley, M.; Sleeman, J.; Tiaden, A. N.; Klasen, J.; Boos, N.; Hottiger, M. O.; Wuertz, K.; Richards, P. J. Hyaluronic acid fragments enhance the inflammatory and catabolic response in human intervertebral disc cells through modulation of toll-like receptor 2 signalling pathways. Arthritis Res Ther. 2013, 15, R94.
doi: 10.1186/ar4274 URL |
| 64. | Zhao, K.; An, R.; Xiang, Q.; Li, G.; Wang, K.; Song, Y.; Liao, Z.; Li, S.; Hua, W.; Feng, X.; Wu, X.; Zhang, Y.; Das, A.; Yang, C. Acid-sensing ion channels regulate nucleus pulposus cell inflammation and pyroptosis via the NLRP3 inflammasome in intervertebral disc degeneration. Cell Prolif. 2021, 54, e12941. |
| 65. |
Bachmeier, B. E.; Nerlich, A.; Mittermaier, N.; Weiler, C.; Lumenta, C.; Wuertz, K.; Boos, N. Matrix metalloproteinase expression levels suggest distinct enzyme roles during lumbar disc herniation and degeneration. Eur Spine J. 2009, 18, 1573-1586.
doi: 10.1007/s00586-009-1031-8 URL |
| 66. |
Gruber, H. E.; Hoelscher, G. L.; Ingram, J. A.; Bethea, S.; Hanley, E. N., Jr. Growth and differentiation factor-5 (GDF-5) in the human intervertebral annulus cells and its modulation by IL-1ß and TNF-α in vitro. Exp Mol Pathol. 2014, 96, 225-229.
doi: 10.1016/j.yexmp.2014.02.005 URL |
| 67. |
Weiler, C.; Nerlich, A. G.; Zipperer, J.; Bachmeier, B. E.; Boos, N. 2002 SSE Award Competition in Basic Science: expression of major matrix metalloproteinases is associated with intervertebral disc degradation and resorption. Eur Spine J. 2002, 11, 308-320.
doi: 10.1007/s00586-002-0472-0 URL |
| 68. |
Risbud, M. V.; Shapiro, I. M. Role of cytokines in intervertebral disc degeneration: pain and disc content. Nat Rev Rheumatol. 2014, 10, 44-56.
doi: 10.1038/nrrheum.2013.160 URL |
| 69. |
Willems, N.; Tellegen, A. R.; Bergknut, N.; Creemers, L. B.; Wolfswinkel, J.; Freudigmann, C.; Benz, K.; Grinwis, G. C.; Tryfonidou, M. A.; Meij, B. P. Inflammatory profiles in canine intervertebral disc degeneration. BMC Vet Res. 2016, 12, 10.
doi: 10.1186/s12917-016-0635-6 URL |
| 70. |
Zhang, Y.; Liu, L.; Wang, S.; Zhao, Y.; Liu, Y.; Li, J.; Nie, L.; Cheng, L. Production of CCL20 on nucleus pulposus cells recruits IL-17-producing cells to degenerated IVD tissues in rat models. J Mol Histol. 2016, 47, 81-89.
doi: 10.1007/s10735-015-9651-2 URL |
| 71. |
Wang, J.; Chen, H.; Cao, P.; Wu, X.; Zang, F.; Shi, L.; Liang, L.; Yuan, W. Inflammatory cytokines induce caveolin-1/β-catenin signalling in rat nucleus pulposus cell apoptosis through the p38 MAPK pathway. Cell Prolif. 2016, 49, 362-372.
doi: 10.1111/cpr.2016.49.issue-3 URL |
| 72. |
Guo, H. Y.; Guo, M. K.; Wan, Z. Y.; Song, F.; Wang, H. Q. Emerging evidence on noncoding-RNA regulatory machinery in intervertebral disc degeneration: a narrative review. Arthritis Res Ther. 2020, 22, 270.
doi: 10.1186/s13075-020-02353-2 URL |
| 73. | Hernandez, P. A.; Jacobsen, T. D.; Chahine, N. O. Actomyosin contractility confers mechanoprotection against TNFα-induced disruption of the intervertebral disc. Sci Adv. 2020, 6, eaba2368. |
| 74. |
Qi, W.; Ren, D.; Wang, P.; Song, Z.; Wu, H.; Yao, S.; Geng, L.; Su, Y.; Bai, X. Upregulation of Sirt1 by tyrosol suppresses apoptosis and inflammation and modulates extracellular matrix remodeling in interleukin-1β-stimulated human nucleus pulposus cells through activation of PI3K/Akt pathway. Int Immunopharmacol. 2020, 88, 106904.
doi: 10.1016/j.intimp.2020.106904 URL |
| 75. |
Molinos, M.; Almeida, C. R.; Caldeira, J.; Cunha, C.; Gonçalves, R. M.; Barbosa, M. A. Inflammation in intervertebral disc degeneration and regeneration. J R Soc Interface. 2015, 12, 20141191.
doi: 10.1098/rsif.2014.1191 URL |
| 76. |
Bian, J.; Cai, F.; Chen, H.; Tang, Z.; Xi, K.; Tang, J.; Wu, L.; Xu, Y.; Deng, L.; Gu, Y.; Cui, W.; Chen, L. Modulation of local overactive inflammation via injectable hydrogel microspheres. Nano Lett. 2021, 21, 2690-2698.
doi: 10.1021/acs.nanolett.0c04713 URL |
| 77. |
Liu, L.; He, J.; Liu, C.; Yang, M.; Fu, J.; Yi, J.; Ai, X.; Liu, M.; Zhuang, Y.; Zhang, Y.; Huang, B.; Li, C.; Zhou, Y.; Feng, C. Cartilage intermediate layer protein affects the progression of intervertebral disc degeneration by regulating the extracellular microenvironment (review). Int J Mol Med. 2021, 47, 475-484.
doi: 10.3892/ijmm URL |
| 78. |
Tsingas, M.; Ottone, O. K.; Haseeb, A.; Barve, R. A.; Shapiro, I. M.; Lefebvre, V.; Risbud, M. V. Sox9 deletion causes severe intervertebral disc degeneration characterized by apoptosis, matrix remodeling, and compartment-specific transcriptomic changes. Matrix Biol. 2020, 94, 110-133.
doi: 10.1016/j.matbio.2020.09.003 URL |
| 79. |
McCann, M. R.; Patel, P.; Pest, M. A.; Ratneswaran, A.; Lalli, G.; Beaucage, K. L.; Backler, G. B.; Kamphuis, M. P.; Esmail, Z.; Lee, J.; Barbalinardo, M.; Mort, J. S.; Holdsworth, D. W.; Beier, F.; Dixon, S. J.; Séguin, C. A. Repeated exposure to high-frequency low-amplitude vibration induces degeneration of murine intervertebral discs and knee joints. Arthritis Rheumatol. 2015, 67, 2164-2175.
doi: 10.1002/art.39154 URL |
| 80. |
Bergknut, N.; Grinwis, G.; Pickee, E.; Auriemma, E.; Lagerstedt, A. S.; Hagman, R.; Hazewinkel, H. A.; Meij, B. P. Reliability of macroscopic grading of intervertebral disk degeneration in dogs by use of the Thompson system and comparison with low-field magnetic resonance imaging findings. Am J Vet Res. 2011, 72, 899-904.
doi: 10.2460/ajvr.72.7.899 URL |
| 81. |
Chen, S.; Liu, S.; Ma, K.; Zhao, L.; Lin, H.; Shao, Z. TGF-β signaling in intervertebral disc health and disease. Osteoarthritis Cartilage. 2019, 27, 1109-1117.
doi: 10.1016/j.joca.2019.05.005 URL |
| 82. |
Wang, S. Z.; Chang, Q.; Lu, J.; Wang, C. Growth factors and platelet-rich plasma: promising biological strategies for early intervertebral disc degeneration. Int Orthop. 2015, 39, 927-934.
doi: 10.1007/s00264-014-2664-8 URL |
| 83. |
Bian, Q.; Jain, A.; Xu, X.; Kebaish, K.; Crane, J. L.; Zhang, Z.; Wan, M.; Ma, L.; Riley, L. H.; Sponseller, P. D.; Guo, X. E.; Lu, W. W.; Wang, Y.; Cao, X. Excessive activation of TGFβ by spinal instability causes vertebral endplate sclerosis. Sci Rep. 2016, 6, 27093.
doi: 10.1038/srep27093 URL |
| 84. |
Zieba, J.; Forlenza, K. N.; Khatra, J. S.; Sarukhanov, A.; Duran, I.; Rigueur, D.; Lyons, K. M.; Cohn, D. H.; Merrill, A. E.; Krakow, D. TGFβ and BMP dependent cell fate changes due to loss of filamin B produces disc degeneration and progressive vertebral fusions. PLoS Genet. 2016, 12, e1005936.
doi: 10.1371/journal.pgen.1005936 URL |
| 85. |
Hu, Y.; Tang, J. S.; Hou, S. X.; Shi, X. X.; Qin, J.; Zhang, T. S.; Wang, X. J. Neuroprotective effects of curcumin alleviate lumbar intervertebral disc degeneration through regulating the expression of iNOS, COX-2, TGF-β1/2, MMP-9 and BDNF in a rat model. Mol Med Rep. 2017, 16, 6864-6869.
doi: 10.3892/mmr.2017.7464 URL |
| 86. |
Li, H.; Li, W.; Liang, B.; Wei, J.; Yin, D.; Fan, Q. Role of AP-2α/TGF-β1/Smad3 axis in rats with intervertebral disc degeneration. Life Sci. 2020, 263, 118567.
doi: 10.1016/j.lfs.2020.118567 URL |
| 87. |
Finnson, K. W.; Parker, W. L.; ten Dijke, P.; Thorikay, M.; Philip, A. ALK1 opposes ALK5/Smad3 signaling and expression of extracellular matrix components in human chondrocytes. J Bone Miner Res. 2008, 23, 896-906.
doi: 10.1359/jbmr.080209 URL |
| 88. |
Than, K. D.; Rahman, S. U.; Vanaman, M. J.; Wang, A. C.; Lin, C. Y.; Zhang, H.; La Marca, F.; Park, P. Bone morphogenetic proteins and degenerative disk disease. Neurosurgery. 2012, 70, 996-1002; discussion 1002.
doi: 10.1227/NEU.0b013e318235d65f URL |
| 89. |
Enochson, L.; Stenberg, J.; Brittberg, M.; Lindahl, A. GDF5 reduces MMP13 expression in human chondrocytes via DKK1 mediated canonical Wnt signaling inhibition. Osteoarthritis Cartilage. 2014, 22, 566-577.
doi: 10.1016/j.joca.2014.02.004 URL |
| 90. |
Ghezelbash, F.; Eskandari, A. H.; Shirazi-Adl, A.; Kazempour, M.; Tavakoli, J.; Baghani, M.; Costi, J. J. Modeling of human intervertebral disc annulus fibrosus with complex multi-fiber networks. Acta Biomater. 2021, 123, 208-221.
doi: 10.1016/j.actbio.2020.12.062 URL |
| 91. |
Hayes, A. J.; Benjamin, M.; Ralphs, J. R. Extracellular matrix in development of the intervertebral disc. Matrix Biol. 2001, 20, 107-121.
doi: 10.1016/S0945-053X(01)00125-1 URL |
| 92. |
Tavakoli, J.; Diwan, A. D.; Tipper, J. L. Advanced strategies for the regeneration of lumbar disc annulus fibrosus. Int J Mol Sci. 2020, 21, 4889.
doi: 10.3390/ijms21144889 URL |
| 93. |
Torre, O. M.; Mroz, V.; Bartelstein, M. K.; Huang, A. H.; Iatridis, J. C. Annulus fibrosus cell phenotypes in homeostasis and injury: implications for regenerative strategies. Ann N Y Acad Sci. 2019, 1442, 61-78.
doi: 10.1111/nyas.2019.1442.issue-1 URL |
| 94. |
Bruehlmann, S. B.; Rattner, J. B.; Matyas, J. R.; Duncan, N. A. Regional variations in the cellular matrix of the annulus fibrosus of the intervertebral disc. J Anat. 2002, 201, 159-171.
doi: 10.1046/j.1469-7580.2002.00080.x URL |
| 95. |
Tavakoli, J.; Elliott, D. M.; Costi, J. J. Structure and mechanical function of the inter-lamellar matrix of the annulus fibrosus in the disc. J Orthop Res. 2016, 34, 1307-1315.
doi: 10.1002/jor.23306 URL |
| 96. | Fernandez-Moure, J.; Moore, C. A.; Kim, K.; Karim, A.; Smith, K.; Barbosa, Z.; Van Eps, J.; Rameshwar, P.; Weiner, B. Novel therapeutic strategies for degenerative disc disease: Review of cell biology and intervertebral disc cell therapy. SAGE open medicine. 2018, 6, 2050312118761674. |
| 97. | Pritzker, K. P. Aging and degeneration in the lumbar intervertebral disc. Orthop Clin North Am. 1977, 8, 66-77. |
| 98. |
Bron, J. L.; Helder, M. N.; Meisel, H. J.; Van Royen, B. J.; Smit, T. H. Repair, regenerative and supportive therapies of the annulus fibrosus: achievements and challenges. Eur Spine J. 2009, 18, 301-313.
doi: 10.1007/s00586-008-0856-x URL |
| 99. |
Chu, G.; Shi, C.; Wang, H.; Zhang, W.; Yang, H.; Li, B. Strategies for annulus fibrosus regeneration: from biological therapies to tissue engineering. Front Bioeng Biotechnol. 2018, 6, 90.
doi: 10.3389/fbioe.2018.00090 URL |
| 100. | Chu, G.; Shi, C.; Lin, J.; Wang, S.; Wang, H.; Liu, T.; Yang, H.; Li, B. Biomechanics in annulus fibrosus degeneration and regeneration. Adv Exp Med Biol. 2018, 1078, 409-420. |
| 101. |
Vergroesen, P. P.; Kingma, I.; Emanuel, K. S.; Hoogendoorn, R. J.; Welting, T. J.; van Royen, B. J.; van Dieën, J. H.; Smit, T. H. Mechanics and biology in intervertebral disc degeneration: a vicious circle. Osteoarthritis Cartilage. 2015, 23, 1057-1070.
doi: 10.1016/j.joca.2015.03.028 URL |
| 102. |
Zhou, Z.; Cui, S.; Du, J.; Richards, R. G.; Alini, M.; Grad, S.; Li, Z. One strike loading organ culture model to investigate the post-traumatic disc degenerative condition. J Orthop Translat. 2021, 26, 141-150.
doi: 10.1016/j.jot.2020.08.003 URL |
| 103. |
Michalek, A. J.; Funabashi, K. L.; Iatridis, J. C. Needle puncture injury of the rat intervertebral disc affects torsional and compressive biomechanics differently. Eur Spine J. 2010, 19, 2110-2116.
doi: 10.1007/s00586-010-1473-z URL |
| 104. | Guterl, C. C.; See, E. Y.; Blanquer, S. B.; Pandit, A.; Ferguson, S. J.; Benneker, L. M.; Grijpma, D. W.; Sakai, D.; Eglin, D.; Alini, M.; Iatridis, J. C.; Grad, S. Challenges and strategies in the repair of ruptured annulus fibrosus. Eur Cell Mater. 2013, 25, 1-21. |
| 105. |
Miyamoto, H.; Doita, M.; Nishida, K.; Yamamoto, T.; Sumi, M.; Kurosaka, M. Effects of cyclic mechanical stress on the production of inflammatory agents by nucleus pulposus and anulus fibrosus derived cells in vitro. Spine (Phila Pa 1976). 2006, 31, 4-9.
doi: 10.1097/01.brs.0000192682.87267.2a URL |
| 106. |
Pratsinis, H.; Papadopoulou, A.; Neidlinger-Wilke, C.; Brayda-Bruno, M.; Wilke, H. J.; Kletsas, D. Cyclic tensile stress of human annulus fibrosus cells induces MAPK activation: involvement in proinflammatory gene expression. Osteoarthritis Cartilage. 2016, 24, 679-687.
doi: 10.1016/j.joca.2015.11.022 URL |
| 107. |
Kanerva, A.; Kommonen, B.; Grönblad, M.; Tolonen, J.; Habtemariam, A.; Virri, J.; Karaharju, E. Inflammatory cells in experimental intervertebral disc injury. Spine (Phila Pa 1976). 1997, 22, 2711-2715.
doi: 10.1097/00007632-199712010-00002 URL |
| 108. |
Wiet, M. G.; Piscioneri, A.; Khan, S. N.; Ballinger, M. N.; Hoyland, J. A.; Purmessur, D. Mast Cell-Intervertebral disc cell interactions regulate inflammation, catabolism and angiogenesis in discogenic back pain. Sci Rep. 2017, 7, 12492.
doi: 10.1038/s41598-017-12666-z URL |
| 109. |
Sainoh, T.; Orita, S.; Miyagi, M.; Sakuma, Y.; Yamauchi, K.; Suzuki, M.; Kubota, G.; Oikawa, Y.; Inage, K.; Sato, J.; Fujimoto, K.; Shiga, Y.; Inoue, G.; Aoki, Y.; Takahashi, K.; Ohtori, S. Interleukin-6 and interleukin-6 receptor expression, localization, and involvement in pain-sensing neuron activation in a mouse intervertebral disc injury model. J Orthop Res. 2015, 33, 1508-1514.
doi: 10.1002/jor.22925 URL |
| 110. | Osti, O. L.; Vernon-Roberts, B.; Fraser, R. D. 1990 Volvo Award in experimental studies. Anulus tears and intervertebral disc degeneration. An experimental study using an animal model. Spine (Phila Pa 1976). 1990, 15, 762-767. |
| 111. |
You, C.; Zhu, K.; Liu, X.; Xi, C.; Zhang, Z.; Xu, G.; Yan, J. Tumor necrosis factor-α-dependent infiltration of macrophages into the dorsal root ganglion in a rat disc herniation model. Spine (Phila Pa 1976). 2013, 38, 2003-2007.
doi: 10.1097/BRS.0b013e3182a84701 URL |
| 112. |
Rand, N. S.; Dawson, J. M.; Juliao, S. F.; Spengler, D. M.; Floman, Y. In vivo macrophage recruitment by murine intervertebral disc cells. J Spinal Disord. 2001, 14, 339-342.
doi: 10.1097/00002517-200108000-00010 URL |
| 113. |
Nakazawa, K. R.; Walter, B. A.; Laudier, D. M.; Krishnamoorthy, D.; Mosley, G. E.; Spiller, K. L.; Iatridis, J. C. Accumulation and localization of macrophage phenotypes with human intervertebral disc degeneration. Spine J. 2018, 18, 343-356.
doi: 10.1016/j.spinee.2017.09.018 URL |
| 114. |
Park, J. J.; Moon, H. J.; Park, J. H.; Kwon, T. H.; Park, Y. K.; Kim, J. H. Induction of proinflammatory cytokine production in intervertebral disc cells by macrophage-like THP-1 cells requires mitogen-activated protein kinase activity. J Neurosurg Spine. 2016, 24, 167-175.
doi: 10.3171/2015.3.SPINE14729 URL |
| 115. | Bai, J.; Zhang, Y.; Fan, Q.; Xu, J.; Shan, H.; Gao, X.; Ma, Q.; Sheng, L.; Zheng, X.; Cheng, W.; Li, D.; Zhang, M.; Hao, Y.; Feng, L.; Chen, Q.; Zhou, X.; Wang, C. Reactive oxygen species-scavenging scaffold with rapamycin for treatment of intervertebral disk degeneration. Adv Healthc Mater. 2020, 9, e1901186. |
| 116. |
Takeoka, Y.; Yurube, T.; Morimoto, K.; Kunii, S.; Kanda, Y.; Tsujimoto, R.; Kawakami, Y.; Fukase, N.; Takemori, T.; Omae, K.; Kakiuchi, Y.; Miyazaki, S.; Kakutani, K.; Takada, T.; Nishida, K.; Fukushima, M.; Kuroda, R. Reduced nucleotomy-induced intervertebral disc disruption through spontaneous spheroid formation by the low adhesive scaffold collagen (LASCol). Biomaterials. 2020, 235, 119781.
doi: 10.1016/j.biomaterials.2020.119781 URL |
| 117. |
Lin, H. A.; Varma, D. M.; Hom, W. W.; Cruz, M. A.; Nasser, P. R.; Phelps, R. G.; Iatridis, J. C.; Nicoll, S. B. Injectable cellulose-based hydrogels as nucleus pulposus replacements: Assessment of in vitro structural stability, ex vivo herniation risk, and in vivo biocompatibility. J Mech Behav Biomed Mater. 2019, 96, 204-213.
doi: 10.1016/j.jmbbm.2019.04.021 URL |
| 118. | Moore, R. J. The vertebral endplate: disc degeneration, disc regeneration. Eur Spine J. 2006, 15 Suppl 3, S333-337. |
| 119. | Lakshmanan, P.; Purushothaman, B.; Dvorak, V.; Schratt, W.; Thambiraj, S.; Boszczyk, M. Sagittal endplate morphology of the lower lumbar spine. Eur Spine J. 2012, 21 Suppl 2, S160-164. |
| 120. |
Che-Nordin, N.; Deng, M.; Griffith, J. F.; Leung, J. C. S.; Kwok, A. W. L.; Zhu, Y. Q.; So, R. H. Y.; Kwok, T. C. Y.; Leung, P. C.; Wáng, Y. X. J. Prevalent osteoporotic vertebral fractures more likely involve the upper endplate than the lower endplate and even more so in males. Ann Transl Med. 2018, 6, 442.
doi: 10.21037/atm URL |
| 121. |
Nagaraja, S.; Awada, H. K.; Dreher, M. L.; Bouck, J. T.; Gupta, S. Effects of vertebroplasty on endplate subsidence in elderly female spines. J Neurosurg Spine. 2015, 22, 273-282.
doi: 10.3171/2014.10.SPINE14195 URL |
| 122. |
Ferguson, S. J.; Steffen, T. Biomechanics of the aging spine. Eur Spine J. 2003, 12 Suppl 2, S97-S103.
doi: 10.1007/s00586-003-0621-0 URL |
| 123. |
Adams, M. A.; Dolan, P. Intervertebral disc degeneration: evidence for two distinct phenotypes. J Anat. 2012, 221, 497-506.
doi: 10.1111/j.1469-7580.2012.01551.x URL |
| 124. |
Braithwaite, I.; White, J.; Saifuddin, A.; Renton, P.; Taylor, B. A. Vertebral end-plate (Modic) changes on lumbar spine MRI: correlation with pain reproduction at lumbar discography. Eur Spine J. 1998, 7, 363-368.
doi: 10.1007/s005860050091 URL |
| 125. |
Kuisma, M.; Karppinen, J.; Niinimäki, J.; Ojala, R.; Haapea, M.; Heliövaara, M.; Korpelainen, R.; Taimela, S.; Natri, A.; Tervonen, O. Modic changes in endplates of lumbar vertebral bodies: prevalence and association with low back and sciatic pain among middle-aged male workers. Spine (Phila Pa 1976). 2007, 32, 1116-1122.
doi: 10.1097/01.brs.0000261561.12944.ff URL |
| 126. |
Albert, H. B.; Manniche, C. Modic changes following lumbar disc herniation. Eur Spine J. 2007, 16, 977-982.
doi: 10.1007/s00586-007-0336-8 URL |
| 127. |
Rahme, R.; Moussa, R. The modic vertebral endplate and marrow changes: pathologic significance and relation to low back pain and segmental instability of the lumbar spine. AJNR Am J Neuroradiol. 2008, 29, 838-842.
doi: 10.3174/ajnr.A0925 URL |
| 128. |
Dudli, S.; Fields, A. J.; Samartzis, D.; Karppinen, J.; Lotz, J. C. Pathobiology of modic changes. Eur Spine J. 2016, 25, 3723-3734.
doi: 10.1007/s00586-016-4459-7 URL |
| 129. |
Crockett, M. T.; Kelly, B. S.; van Baarsel, S.; Kavanagh, E. C. Modic type 1 vertebral endplate changes: injury, inflammation, or infection?. AJR Am J Roentgenol. 2017, 209, 167-170.
doi: 10.2214/AJR.16.17403 URL |
| 130. |
Hanımoğlu, H.; Çevik, S.; Yılmaz, H.; Kaplan, A.; Çalış, F.; Katar, S.; Evran, Ş.; Akkaya, E.; Karaca, O. Effects of modic type 1 changes in the vertebrae on low back pain. World Neurosurg. 2019, 121, e426-e432.
doi: 10.1016/j.wneu.2018.09.132 URL |
| 131. |
Ni, S.; Ling, Z.; Wang, X.; Cao, Y.; Wu, T.; Deng, R.; Crane, J. L.; Skolasky, R.; Demehri, S.; Zhen, G.; Jain, A.; Wu, P.; Pan, D.; Hu, B.; Lyu, X.; Li, Y.; Chen, H.; Qi, H.; Guan, Y.; Dong, X.; Wan, M.; Zou, X.; Lu, H.; Hu, J.; Cao, X. Sensory innervation in porous endplates by Netrin-1 from osteoclasts mediates PGE2-induced spinal hypersensitivity in mice. Nat Commun. 2019, 10, 5643.
doi: 10.1038/s41467-019-13476-9 URL |
| 132. |
Freemont, A. J.; Peacock, T. E.; Goupille, P.; Hoyland, J. A.; O’Brien, J.; Jayson, M. I. Nerve ingrowth into diseased intervertebral disc in chronic back pain. Lancet. 1997, 350, 178-181.
doi: 10.1016/S0140-6736(97)02135-1 URL |
| 133. |
Fields, A. J.; Liebenberg, E. C.; Lotz, J. C. Innervation of pathologies in the lumbar vertebral end plate and intervertebral disc. Spine J. 2014, 14, 513-521.
doi: 10.1016/j.spinee.2013.06.075 URL |
| 134. |
Ohtori, S.; Miyagi, M.; Inoue, G. Sensory nerve ingrowth, cytokines, and instability of discogenic low back pain: A review. Spine Surg Relat Res. 2018, 2, 11-17.
doi: 10.22603/ssrr.2016-0018 URL |
| 135. |
Lama, P.; Le Maitre, C. L.; Harding, I. J.; Dolan, P.; Adams, M. A. Nerves and blood vessels in degenerated intervertebral discs are confined to physically disrupted tissue. J Anat. 2018, 233, 86-97.
doi: 10.1111/joa.2018.233.issue-1 URL |
| 136. |
Zhang, S.; Hu, B.; Liu, W.; Wang, P.; Lv, X.; Chen, S.; Shao, Z. The role of structure and function changes of sensory nervous system in intervertebral disc-related low back pain. Osteoarthritis Cartilage. 2021, 29, 17-27.
doi: 10.1016/j.joca.2020.09.002 URL |
| 137. |
Johnson, W. E.; Caterson, B.; Eisenstein, S. M.; Hynds, D. L.; Snow, D. M.; Roberts, S. Human intervertebral disc aggrecan inhibits nerve growth in vitro. Arthritis Rheum. 2002, 46, 2658-2664.
doi: 10.1002/(ISSN)1529-0131 URL |
| 138. |
Lee, J. M.; Song, J. Y.; Baek, M.; Jung, H. Y.; Kang, H.; Han, I. B.; Kwon, Y. D.; Shin, D. E. Interleukin-1β induces angiogenesis and innervation in human intervertebral disc degeneration. J Orthop Res. 2011, 29, 265-269.
doi: 10.1002/jor.v29.2 URL |
| 139. |
Binch, A. L.; Cole, A. A.; Breakwell, L. M.; Michael, A. L.; Chiverton, N.; Cross, A. K.; Le Maitre, C. L. Expression and regulation of neurotrophic and angiogenic factors during human intervertebral disc degeneration. Arthritis Res Ther. 2014, 16, 416.
doi: 10.1186/s13075-014-0416-1 URL |
| 140. |
Obata, K.; Tsujino, H.; Yamanaka, H.; Yi, D.; Fukuoka, T.; Hashimoto, N.; Yonenobu, K.; Yoshikawa, H.; Noguchi, K. Expression of neurotrophic factors in the dorsal root ganglion in a rat model of lumbar disc herniation. Pain. 2002, 99, 121-132.
doi: 10.1016/S0304-3959(02)00068-4 URL |
| 141. |
Henry, N.; Clouet, J.; Le Bideau, J.; Le Visage, C.; Guicheux, J. Innovative strategies for intervertebral disc regenerative medicine: From cell therapies to multiscale delivery systems. Biotechnol Adv. 2018, 36, 281-294.
doi: 10.1016/j.biotechadv.2017.11.009 URL |
| 142. | Alkhatib, B.; Rosenzweig, D. H.; Krock, E.; Roughley, P. J.; Beckman, L.; Steffen, T.; Weber, M. H.; Ouellet, J. A.; Haglund, L. Acute mechanical injury of the human intervertebral disc: link to degeneration and pain. Eur Cell Mater. 2014, 28, 98-110; discussion 110-111. |
| 143. | Fournier, D. E.; Kiser, P. K.; Shoemaker, J. K.; Battié, M. C.; Séguin, C. A. Vascularization of the human intervertebral disc: A scoping review. JOR Spine. 2020, 3, e1123. |
| 144. |
Hassler, O. The human intervertebral disc. A micro-angiographical study on its vascular supply at various ages. Acta Orthop Scand. 1969, 40, 765-772.
doi: 10.3109/17453676908989540 URL |
| 145. |
Hee, H. T.; Chuah, Y. J.; Tan, B. H.; Setiobudi, T.; Wong, H. K. Vascularization and morphological changes of the endplate after axial compression and distraction of the intervertebral disc. Spine (Phila Pa 1976). 2011, 36, 505-511.
doi: 10.1097/BRS.0b013e3181d32410 URL |
| 146. |
Binch, A. L.; Cole, A. A.; Breakwell, L. M.; Michael, A. L.; Chiverton, N.; Creemers, L. B.; Cross, A. K.; Le Maitre, C. L. Nerves are more abundant than blood vessels in the degenerate human intervertebral disc. Arthritis Res Ther. 2015, 17, 370.
doi: 10.1186/s13075-015-0889-6 URL |
| 147. |
Lyu, F. J.; Cheung, K. M.; Zheng, Z.; Wang, H.; Sakai, D.; Leung, V. Y. IVD progenitor cells: a new horizon for understanding disc homeostasis and repair. Nat Rev Rheumatol. 2019, 15, 102-112.
doi: 10.1038/s41584-018-0154-x URL |
| 148. | Wang, B.; Ke, W.; Wang, K.; Li, G.; Ma, L.; Lu, S.; Xiang, Q.; Liao, Z.; Luo, R.; Song, Y.; Hua, W.; Wu, X.; Zhang, Y.; Zeng, X.; Yang, C. Mechanosensitive ion channel piezo1 activated by matrix stiffness regulates oxidative stress-induced senescence and apoptosis in human intervertebral disc degeneration. Oxid Med Cell Longev. 2021, 2021, 8884922. |
| 149. |
Lin, J.; Du, J.; Wu, X.; Xu, C.; Liu, J.; Jiang, L.; Cheng, X.; Ge, G.; Chen, L.; Pang, Q.; Geng, D.; Mao, H. SIRT3 mitigates intervertebral disc degeneration by delaying oxidative stress-induced senescence of nucleus pulposus cells. J Cell Physiol. 2021. doi: 10.1002/jcp.30319.
doi: 10.1002/jcp.30319 URL |
| 150. |
Tang, N.; Dong, Y.; Chen, C.; Zhao, H. Anisodamine maintains the stability of intervertebral disc tissue by inhibiting the senescence of nucleus pulposus cells and degradation of extracellular matrix via interleukin-6/janus kinases/signal transducer and activator of transcription 3 pathway. Front Pharmacol. 2020, 11, 519172.
doi: 10.3389/fphar.2020.519172 URL |
| 151. |
Lotz, J. C.; Haughton, V.; Boden, S. D.; An, H. S.; Kang, J. D.; Masuda, K.; Freemont, A.; Berven, S.; Sengupta, D. K.; Tanenbaum, L.; Maurer, P.; Ranganathan, A.; Alavi, A.; Marinelli, N. L. New treatments and imaging strategies in degenerative disease of the intervertebral disks. Radiology. 2012, 264, 6-19.
doi: 10.1148/radiol.12110339 URL |
| 152. |
Che, H.; Li, J.; Li, Y.; Ma, C.; Liu, H.; Qin, J.; Dong, J.; Zhang, Z.; Xian, C. J.; Miao, D.; Wang, L.; Ren, Y. p16 deficiency attenuates intervertebral disc degeneration by adjusting oxidative stress and nucleus pulposus cell cycle. Elife. 2020, 9, e52570.
doi: 10.7554/eLife.52570 URL |
| 153. |
Bárdos, T.; Szabó, Z.; Czipri, M.; Vermes, C.; Tunyogi-Csapó, M.; Urban, R. M.; Mikecz, K.; Glant, T. T. A longitudinal study on an autoimmune murine model of ankylosing spondylitis. Ann Rheum Dis. 2005, 64, 981-987.
doi: 10.1136/ard.2004.029710 URL |
| 154. |
Mohanty, S.; Dahia, C. L. Defects in intervertebral disc and spine during development, degeneration, and pain: New research directions for disc regeneration and therapy. Wiley Interdiscip Rev Dev Biol. 2019, 8, e343.
doi: 10.1002/wdev.2019.8.issue-4 URL |
| 155. | Hartman, R.; Patil, P.; Tisherman, R.; St Croix, C.; Niedernhofer, L. J.; Robbins, P. D.; Ambrosio, F.; Van Houten, B.; Sowa, G.; Vo, N. Age-dependent changes in intervertebral disc cell mitochondria and bioenergetics. Eur Cell Mater. 2018, 36, 171-183. |
| 156. | Vadalà, G.; Ambrosio, L.; Russo, F.; Papalia, R.; Denaro, V. Interaction between mesenchymal stem cells and intervertebral disc microenvironment: from cell therapy to tissue engineering. Stem Cells Int. 2019, 2019, 2376172. |
| 157. |
Vo, N. V.; Hartman, R. A.; Patil, P. R.; Risbud, M. V.; Kletsas, D.; Iatridis, J. C.; Hoyland, J. A.; Le Maitre, C. L.; Sowa, G. A.; Kang, J. D. Molecular mechanisms of biological aging in intervertebral discs. J Orthop Res. 2016, 34, 1289-1306.
doi: 10.1002/jor.23195 URL |
| 158. |
Risbud, M. V.; Schipani, E.; Shapiro, I. M. Hypoxic regulation of nucleus pulposus cell survival: from niche to notch. Am J Pathol. 2010, 176, 1577-1583.
doi: 10.2353/ajpath.2010.090734 URL |
| 159. |
Peck, S. H.; Bendigo, J. R.; Tobias, J. W.; Dodge, G. R.; Malhotra, N. R.; Mauck, R. L.; Smith, L. J. Hypoxic preconditioning enhances bone marrow-derived mesenchymal stem cell survival in a low oxygen and nutrient-limited 3D microenvironment. Cartilage. 2019. doi: 10.1177/1947603519841675.
doi: 10.1177/1947603519841675 URL |
| 160. |
Potier, E.; Ferreira, E.; Meunier, A.; Sedel, L.; Logeart-Avramoglou, D.; Petite, H. Prolonged hypoxia concomitant with serum deprivation induces massive human mesenchymal stem cell death. Tissue Eng. 2007, 13, 1325-1331.
doi: 10.1089/ten.2006.0325 URL |
| 161. |
Tsai, T. T.; Danielson, K. G.; Guttapalli, A.; Oguz, E.; Albert, T. J.; Shapiro, I. M.; Risbud, M. V. TonEBP/OREBP is a regulator of nucleus pulposus cell function and survival in the intervertebral disc. J Biol Chem. 2006, 281, 25416-25424.
doi: 10.1074/jbc.M601969200 URL |
| 162. |
van Dijk, B.; Potier, E.; Ito, K. Culturing bovine nucleus pulposus explants by balancing medium osmolarity. Tissue Eng Part C Methods. 2011, 17, 1089-1096.
doi: 10.1089/ten.tec.2011.0215 URL |
| 163. |
Li, P.; Gan, Y.; Wang, H.; Xu, Y.; Li, S.; Song, L.; Zhang, C.; Ou, Y.; Wang, L.; Zhou, Q. Role of the ERK1/2 pathway in osmolarity effects on nucleus pulposus cell apoptosis in a disc perfusion culture. J Orthop Res. 2017, 35, 86-92.
doi: 10.1002/jor.v35.1 URL |
| 164. |
Wuertz, K.; Godburn, K.; Neidlinger-Wilke, C.; Urban, J.; Iatridis, J. C. Behavior of mesenchymal stem cells in the chemical microenvironment of the intervertebral disc. Spine (Phila Pa 1976). 2008, 33, 1843-1849.
doi: 10.1097/BRS.0b013e31817b8f53 URL |
| 165. |
Liang, C.; Li, H.; Tao, Y.; Zhou, X.; Li, F.; Chen, G.; Chen, Q. Responses of human adipose-derived mesenchymal stem cells to chemical microenvironment of the intervertebral disc. J Transl Med. 2012, 10, 49.
doi: 10.1186/1479-5876-10-49 URL |
| 166. |
Li, H.; Wang, J.; Li, F.; Chen, G.; Chen, Q. The influence of hyperosmolarity in the intervertebral disc on the proliferation and chondrogenic differentiation of nucleus pulposus-derived mesenchymal stem cells. Cells Tissues Organs. 2018, 205, 178-188.
doi: 10.1159/000490760 URL |
| 167. | Sandor, Z.; Rathonyi, G. K.; Dinya, E. Assessment of lumbar lordosis distribution with a novel mathematical approach and its adaptation for lumbar intervertebral disc degeneration. Comput Math Methods Med. 2020, 2020, 7312125. |
| 168. |
Zhou, C.; Willing, R. Alterations in the geometry, fiber orientation, and mechanical behavior of the lumbar intervertebral disc by nucleus swelling. J Biomech Eng. 2020, 142, 084502.
doi: 10.1115/1.4046362 URL |
| 169. |
Weber, C. I.; Hwang, C. T.; van Dillen, L. R.; Tang, S. Y. Effects of standing on lumbar spine alignment and intervertebral disc geometry in young, healthy individuals determined by positional magnetic resonance imaging. Clin Biomech (Bristol, Avon). 2019, 65, 128-134.
doi: 10.1016/j.clinbiomech.2019.04.010 URL |
| 170. |
O’Connell, G. D.; Vresilovic, E. J.; Elliott, D. M. Comparison of animals used in disc research to human lumbar disc geometry. Spine (Phila Pa 1976). 2007, 32, 328-333.
doi: 10.1097/01.brs.0000253961.40910.c1 URL |
| 171. |
Kandil, K.; Zaïri, F.; Messager, T.; Zaïri, F. A microstructure-based modeling approach to assess aging-sensitive mechanics of human intervertebral disc. Comput Methods Programs Biomed. 2021, 200, 105890.
doi: 10.1016/j.cmpb.2020.105890 URL |
| 172. | Rosenzweig, D. H.; Gawri, R.; Moir, J.; Beckman, L.; Eglin, D.; Steffen, T.; Roughley, P. J.; Ouellet, J. A.; Haglund, L. Dynamic loading, matrix maintenance and cell injection therapy of human intervertebral discs cultured in a bioreactor. Eur Cell Mater. 2016, 31, 26-39. |
| 173. |
Hartman, R. A.; Yurube, T.; Ngo, K.; Merzlak, N. E.; Debski, R. E.; Brown, B. N.; Kang, J. D.; Sowa, G. A. Biological responses to flexion/extension in spinal segments ex-vivo. J Orthop Res. 2015, 33, 1255-1264.
doi: 10.1002/jor.22900 URL |
| 174. |
Wilke, H. J.; Rohlmann, A.; Neller, S.; Graichen, F.; Claes, L.; Bergmann, G. ISSLS prize winner: A novel approach to determine trunk muscle forces during flexion and extension: a comparison of data from an in vitro experiment and in vivo measurements. Spine (Phila Pa 1976). 2003, 28, 2585-2593.
doi: 10.1097/01.BRS.0000096673.16363.C7 URL |
| 175. |
Alini, M.; Eisenstein, S. M.; Ito, K.; Little, C.; Kettler, A. A.; Masuda, K.; Melrose, J.; Ralphs, J.; Stokes, I.; Wilke, H. J. Are animal models useful for studying human disc disorders/degeneration? Eur Spine J. 2008, 17, 2-19.
doi: 10.1007/s00586-007-0414-y URL |
| 176. |
Motaghinasab, S.; Shirazi-Adl, A.; Urban, J. P.; Parnianpour, M. Computational pharmacokinetics of solute penetration into human intervertebral discs - effects of endplate permeability, solute molecular weight and disc size. J Biomech. 2012, 45, 2195-2202.
doi: 10.1016/j.jbiomech.2012.06.033 URL |
| 177. |
Jackson, A. R.; Yuan, T. Y.; Huang, C. Y.; Brown, M. D.; Gu, W. Y. Nutrient transport in human annulus fibrosus is affected by compressive strain and anisotropy. Ann Biomed Eng. 2012, 40, 2551-2558.
doi: 10.1007/s10439-012-0606-4 URL |
| 178. |
Malandrino, A.; Noailly, J.; Lacroix, D. The effect of sustained compression on oxygen metabolic transport in the intervertebral disc decreases with degenerative changes. PLoS Comput Biol. 2011, 7, e1002112.
doi: 10.1371/journal.pcbi.1002112 URL |
| 179. |
Soukane, D. M.; Shirazi-Adl, A.; Urban, J. P. Computation of coupled diffusion of oxygen, glucose and lactic acid in an intervertebral disc. J Biomech. 2007, 40, 2645-2654.
doi: 10.1016/j.jbiomech.2007.01.003 URL |
| 180. |
DiStefano, T. J.; Shmukler, J. O.; Danias, G.; Iatridis, J. C. The functional role of interface tissue engineering in annulus fibrosus repair: bridging mechanisms of hydrogel integration with regenerative outcomes. ACS Biomater Sci Eng. 2020, 6, 6556-6586.
doi: 10.1021/acsbiomaterials.0c01320 URL |
| 181. | Kim, K. W.; Lim, T. H.; Kim, J. G.; Jeong, S. T.; Masuda, K.; An, H. S. The origin of chondrocytes in the nucleus pulposus and histologic findings associated with the transition of a notochordal nucleus pulposus to a fibrocartilaginous nucleus pulposus in intact rabbit intervertebral discs. Spine (Phila Pa 1976). 2003, 28, 982-990. |
| 182. | Zhao, X.; Liu, H.; Feng, G.; Deng, L.; Li, X.; Liang, T. Notochord cells enhance proliferation and phenotype-keeping of intervertebral disc chondroid cells. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2008, 22, 939-943. |
| 183. |
Sun, Z.; Liu, B.; Liu, Z. H.; Song, W.; Wang, D.; Chen, B. Y.; Fan, J.; Xu, Z.; Geng, D.; Luo, Z. J. Notochordal-cell-derived exosomes induced by compressive load inhibit angiogenesis via the miR-140-5p/Wnt/β-Catenin axis. Mol Ther Nucleic Acids. 2020, 22, 1092-1106.
doi: 10.1016/j.omtn.2020.10.021 URL |
| 184. |
Yin, X.; Motorwala, A.; Vesvoranan, O.; Levene, H. B.; Gu, W.; Huang, C. Y. Effects of glucose deprivation on ATP and proteoglycan production of intervertebral disc cells under hypoxia. Sci Rep. 2020, 10, 8899.
doi: 10.1038/s41598-020-65691-w URL |
| 185. |
Tong, W.; Lu, Z.; Qin, L.; Mauck, R. L.; Smith, H. E.; Smith, L. J.; Malhotra, N. R.; Heyworth, M. F.; Caldera, F.; Enomoto-Iwamoto, M.; Zhang, Y. Cell therapy for the degenerating intervertebral disc. Transl Res. 2017, 181, 49-58.
doi: 10.1016/j.trsl.2016.11.008 URL |
| 186. | Hansen, H. J. Comparative views of the pathology of disk degeneration in animals. Lab Invest. 1959, 8, 1242-1265. |
| 187. |
Souter, W. A.; Taylor, T. K. Sulphated acid mucopolysaccharide metabolism in the rabbit intervertebral disc. J Bone Joint Surg Br. 1970, 52, 371-384.
doi: 10.2106/00004623-197052020-00022 URL |
| 188. | Scott, N. A.; Harris, P. F.; Bagnall, K. M. A morphological and histological study of the postnatal development of intervertebral discs in the lumbar spine of the rabbit. J Anat. 1980, 130, 75-81. |
| 189. | Daly, C.; Ghosh, P.; Jenkin, G.; Oehme, D.; Goldschlager, T. A review of animal models of intervertebral disc degeneration: pathophysiology, regeneration, and translation to the clinic. Biomed Res Int. 2016, 2016, 5952165. |
| 190. |
Cho, H.; Park, S. H.; Lee, S.; Kang, M.; Hasty, K. A.; Kim, S. J. Snapshot of degenerative aging of porcine intervertebral disc: a model to unravel the molecular mechanisms. Exp Mol Med. 2011, 43, 334-340.
doi: 10.3858/emm.2011.43.6.036 URL |
| 191. |
Bach, F. C.; Willems, N.; Penning, L. C.; Ito, K.; Meij, B. P.; Tryfonidou, M. A. Potential regenerative treatment strategies for intervertebral disc degeneration in dogs. BMC Vet Res. 2014, 10, 3.
doi: 10.1186/1746-6148-10-3 URL |
| 192. |
Li, R.; Wang, Z.; Ma, L.; Yang, D.; Xie, D.; Zhang, B.; Ding, W. Lumbar vertebral endplate defects on magnetic resonance imaging in degenerative spondylolisthesis: novel classification, characteristics, and correlative factor analysis. World Neurosurg. 2020, 141, e423-e430.
doi: 10.1016/j.wneu.2020.05.163 URL |
| 193. | Somovilla-Gómez, F.; Lostado-Lorza, R.; Corral-Bobadilla, M.; Escribano-García, R. Improvement in determining the risk of damage to the human lumbar functional spinal unit considering age, height, weight and sex using a combination of FEM and RSM. Biomech Model Mechanobiol. 2020, 19, 351-387. |
| 194. |
Mosley, G. E.; Hoy, R. C.; Nasser, P.; Kaseta, T.; Lai, A.; Evashwick-Rogler, T. W.; Lee, M.; Iatridis, J. C. Sex differences in rat intervertebral disc structure and function following annular puncture injury. Spine (Phila Pa 1976). 2019, 44, 1257-1269.
doi: 10.1097/BRS.0000000000003055 URL |
| 195. |
Gruber, H. E.; Gordon, B.; Williams, C.; Norton, H. J.; Hanley, E. N., Jr. Vertebral endplate and disc changes in the aging sand rat lumbar spine: cross-sectional analyses of a large male and female population. Spine (Phila Pa 1976). 2007, 32, 2529-2536.
doi: 10.1097/BRS.0b013e318158cd69 URL |
| 196. |
LaCroix-Fralish, M. L.; Rutkowski, M. D.; Weinstein, J. N.; Mogil, J. S.; Deleo, J. A. The magnitude of mechanical allodynia in a rodent model of lumbar radiculopathy is dependent on strain and sex. Spine (Phila Pa 1976). 2005, 30, 1821-1827.
doi: 10.1097/01.brs.0000174122.63291.38 URL |
| 197. |
Fillingim, R. B.; King, C. D.; Ribeiro-Dasilva, M. C.; Rahim-Williams, B.; Riley, J. L., 3rd . Sex, gender, and pain: a review of recent clinical and experimental findings. J Pain. 2009, 10, 447-485.
doi: 10.1016/j.jpain.2008.12.001 URL |
| 198. |
Gautschi, O. P.; Corniola, M. V.; Smoll, N. R.; Joswig, H.; Schaller, K.; Hildebrandt, G.; Stienen, M. N. Sex differences in subjective and objective measures of pain, functional impairment, and health-related quality of life in patients with lumbar degenerative disc disease. Pain. 2016, 157, 1065-1071.
doi: 10.1097/j.pain.0000000000000480 URL |
| 199. |
Yang, S.; Zhang, F.; Ma, J.; Ding, W. Intervertebral disc ageing and degeneration: The antiapoptotic effect of oestrogen. Ageing Res Rev. 2020, 57, 100978.
doi: 10.1016/j.arr.2019.100978 URL |
| 200. |
Lauerman, W. C.; Platenberg, R. C.; Cain, J. E.; Deeney, V. F. Age-related disk degeneration: preliminary report of a naturally occurring baboon model. J Spinal Disord. 1992, 5, 170-174.
doi: 10.1097/00002517-199206000-00004 URL |
| 201. |
Platenberg, R. C.; Hubbard, G. B.; Ehler, W. J.; Hixson, C. J. Spontaneous disc degeneration in the baboon model: magnetic resonance imaging and histopathologic correlation. J Med Primatol. 2001, 30, 268-272.
doi: 10.1034/j.1600-0684.2001.d01-59.x URL |
| 202. |
Nuckley, D. J.; Kramer, P. A.; Del Rosario, A.; Fabro, N.; Baran, S.; Ching, R. P. Intervertebral disc degeneration in a naturally occurring primate model: radiographic and biomechanical evidence. J Orthop Res. 2008, 26, 1283-1288.
doi: 10.1002/jor.v26:9 URL |
| 203. |
Showalter, B. L.; Beckstein, J. C.; Martin, J. T.; Beattie, E. E.; Espinoza Orías, A. A.; Schaer, T. P.; Vresilovic, E. J.; Elliott, D. M. Comparison of animal discs used in disc research to human lumbar disc: torsion mechanics and collagen content. Spine (Phila Pa 1976). 2012, 37, E900-907.
doi: 10.1097/BRS.0b013e31824d911c URL |
| 204. |
Videman, T.; Battié, M. C.; Gill, K.; Manninen, H.; Gibbons, L. E.; Fisher, L. D. Magnetic resonance imaging findings and their relationships in the thoracic and lumbar spine. Insights into the etiopathogenesis of spinal degeneration. Spine (Phila Pa 1976). 1995, 20, 928-935.
doi: 10.1097/00007632-199504150-00009 URL |
| 205. | Battié, M. C.; Videman, T. Lumbar disc degeneration: epidemiology and genetics. J Bone Joint Surg Am. 2006, 88 Suppl 2, 3-9. |
| 206. | Lei, C.; Colangelo, D.; Patil, P.; Li, V.; Ngo, K.; Wang, D.; Dong, Q.; Yousefzadeh, M. J.; Lin, H.; Lee, J.; Kang, J.; Sowa, G.; Wyss-Coray, T.; Niedernhofer, L. J.; Robbins, P. D.; Huffman, D. M.; Vo, N. Influences of circulatory factors on intervertebral disc aging phenotype. Aging (Albany NY). 2020, 12, 12285-12304. |
| 207. | Pfeiffer, M.; Wilke, A.; Goetz, W.; Chaparro, F.; Coetzee, E.; Griss, P. Comparison of two experimental models of intervertebral disc degeneration in mammals. Orthop Proc. 2002, 84-B, 18-d-18. |
| 208. | Jin, L.; Balian, G.; Li, X. J. Animal models for disc degeneration-an update. Histol Histopathol. 2018, 33, 543-554. |
| 209. |
Xi, Y.; Kong, J.; Liu, Y.; Wang, Z.; Ren, S.; Diao, Z.; Hu, Y. Minimally invasive induction of an early lumbar disc degeneration model in rhesus monkeys. Spine (Phila Pa 1976). 2013, 38, E579-586.
doi: 10.1097/BRS.0b013e31828b695b URL |
| 210. |
Wei, F.; Zhong, R.; Wang, L.; Zhou, Z.; Pan, X.; Cui, S.; Sun, H.; Zou, X.; Gao, M.; Jiang, B.; Chen, W.; Zhuang, W.; Sun, H.; Liu, S. Pingyangmycin-induced in vivo lumbar disc degeneration model of rhesus monkeys. Spine (Phila Pa 1976). 2015, 40, E199-210.
doi: 10.1097/BRS.0000000000000736 URL |
| 211. |
Wei, F.; Zhong, R.; Zhou, Z.; Wang, L.; Pan, X.; Cui, S.; Zou, X.; Gao, M.; Sun, H.; Chen, W.; Liu, S. In vivo experimental intervertebral disc degeneration induced by bleomycin in the rhesus monkey. BMC Musculoskelet Disord. 2014, 15, 340.
doi: 10.1186/1471-2474-15-340 URL |
| 212. | Ando, T.; Kato, F.; Mimatsu, K.; Iwata, H. Effects of chondroitinase ABC on degenerative intervertebral discs. Clin Orthop Relat Res. 1995, 214-221. |
| 213. |
Yamada, K.; Tanabe, S.; Ueno, H.; Oinuma, A.; Takahashi, T.; Miyauchi, S.; Shigeno, S.; Hirose, T.; Miyahara, K.; Sato, M. Investigation of the short-term effect of chemonucleolysis with chondroitinase ABC. J Vet Med Sci. 2001, 63, 521-525.
doi: 10.1292/jvms.63.521 URL |
| 214. |
Lu, D. S.; Luk, K. D.; Lu, W. W.; Cheung, K. M.; Leong, J. C. Spinal flexibility increase after chymopapain injection is dose dependent: a possible alternative to anterior release in scoliosis. Spine (Phila Pa 1976). 2004, 29, 123-128.
doi: 10.1097/01.BRS.0000107003.19164.18 URL |
| 215. |
Melrose, J.; Taylor, T. K.; Ghosh, P.; Holbert, C.; Macpherson, C.; Bellenger, C. R. Intervertebral disc reconstitution after chemonucleolysis with chymopapain is dependent on dosage. Spine (Phila Pa 1976). 1996, 21, 9-17.
doi: 10.1097/00007632-199601010-00002 URL |
| 216. |
Melrose, J.; Hall, A.; Macpherson, C.; Bellenger, C. R.; Ghosh, P. Evaluation of digestive proteinases from the Antarctic krill Euphasia superba as potential chemonucleolytic agents. In vitro and in vivo studies. Arch Orthop Trauma Surg. 1995, 114, 145-152.
doi: 10.1007/BF00443388 URL |
| 217. |
Suguro, T.; Oegema, T. R., Jr.; Bradford, D. S Ultrastructural study of the short-term effects of chymopapain on the intervertebral disc. J Orthop Res. 1986, 4, 281-287.
doi: 10.1002/(ISSN)1554-527X URL |
| 218. | Jiang, H.; Wang, J.; Xu, B.; Yang, Q.; Liu, Y. Study on the expression of nerve growth associated protein-43 in rat model of intervertebral disc degeneration. J Musculoskelet Neuronal Interact. 2017, 17, 104-107. |
| 219. |
Wilke, H. J.; Kettler, A.; Claes, L. E. Are sheep spines a valid biomechanical model for human spines? Spine (Phila Pa 1976). 1997, 22, 2365-2374.
doi: 10.1097/00007632-199710150-00009 URL |
| 220. |
Turgut, M.; Başaloğlu, H. K.; Yenisey, C.; Ozsunar, Y. Surgical pinealectomy accelerates intervertebral disc degeneration process in chicken. Eur Spine J. 2006, 15, 605-612.
doi: 10.1007/s00586-005-0972-9 URL |
| 221. |
Machida, M.; Murai, I.; Miyashita, Y.; Dubousset, J.; Yamada, T.; Kimura, J. Pathogenesis of idiopathic scoliosis. Experimental study in rats. Spine (Phila Pa 1976). 1999, 24, 1985-1989.
doi: 10.1097/00007632-199910010-00004 URL |
| 222. |
Turgut, M.; Sargin, H.; Onol, B.; Açikgöz, B. Changes in end-plate vascularity after Nd: YAG laser application to the guinea pig intervertebral disc. Acta Neurochir (Wien). 1998, 140, 819-825; discussion 825-826.
doi: 10.1007/s007010050184 URL |
| 223. |
Pedrini-Mille, A.; Weinstein, J. N.; Found, E. M.; Chung, C. B.; Goel, V. K. Stimulation of dorsal root ganglia and degradation of rabbit annulus fibrosus. Spine (Phila Pa 1976). 1990, 15, 1252-1256.
doi: 10.1097/00007632-199012000-00004 URL |
| 224. |
Puustjärvi, K.; Lammi, M.; Helminen, H.; Inkinen, R.; Tammi, M. Proteoglycans in the intervertebral disc of young dogs following strenuous running exercise. Connect Tissue Res. 1994, 30, 225-240.
doi: 10.3109/03008209409061974 URL |
| 225. |
Puustjärvi, K.; Lammi, M.; Kiviranta, I.; Helminen, H. J.; Tammi, M. Proteoglycan synjournal in canine intervertebral discs after long-distance running training. J Orthop Res. 1993, 11, 738-746.
doi: 10.1002/jor.v11:5 URL |
| 226. |
Säämänen, A. M.; Puustjärvi, K.; Ilves, K.; Lammi, M.; Kiviranta, I.; Jurvelin, J.; Helminen, H. J.; Tammi, M. Effect of running exercise on proteoglycans and collagen content in the intervertebral disc of young dogs. Int J Sports Med. 1993, 14, 48-51.
doi: 10.1055/s-2007-1021145 URL |
| 227. |
Lerer, A.; Nykamp, S. G.; Harriss, A. B.; Gibson, T. W.; Koch, T. G.; Brown, S. H. MRI-based relationships between spine pathology, intervertebral disc degeneration, and muscle fatty infiltration in chondrodystrophic and non-chondrodystrophic dogs. Spine J. 2015, 15, 2433-2439.
doi: 10.1016/j.spinee.2015.08.014 URL |
| 228. |
Smolders, L. A.; Bergknut, N.; Grinwis, G. C.; Hagman, R.; Lagerstedt, A. S.; Hazewinkel, H. A.; Tryfonidou, M. A.; Meij, B. P. Intervertebral disc degeneration in the dog. Part 2: chondrodystrophic and non-chondrodystrophic breeds. Vet J. 2013, 195, 292-299.
doi: 10.1016/j.tvjl.2012.10.011 URL |
| 229. |
Bergknut, N.; Rutges, J. P.; Kranenburg, H. J.; Smolders, L. A.; Hagman, R.; Smidt, H. J.; Lagerstedt, A. S.; Penning, L. C.; Voorhout, G.; Hazewinkel, H. A.; Grinwis, G. C.; Creemers, L. B.; Meij, B. P.; Dhert, W. J. The dog as an animal model for intervertebral disc degeneration? Spine (Phila Pa 1976). 2012, 37, 351-358.
doi: 10.1097/BRS.0b013e31821e5665 URL |
| 230. | Ohnishi, T.; Sudo, H.; Tsujimoto, T.; Iwasaki, N. Age-related spontaneous lumbar intervertebral disc degeneration in a mouse model. J Orthop Res. 2018, 36, 224-232. |
| 231. | Tajerian, M.; Alvarado, S.; Millecamps, M.; Dashwood, T.; Anderson, K. M.; Haglund, L.; Ouellet, J.; Szyf, M.; Stone, L. S. DNA methylation of SPARC and chronic low back pain. Mol Pain. 2011, 7, 65. |
| 232. |
Boyd, L. M.; Richardson, W. J.; Allen, K. D.; Flahiff, C.; Jing, L.; Li, Y.; Chen, J.; Setton, L. A. Early-onset degeneration of the intervertebral disc and vertebral end plate in mice deficient in type IX collagen. Arthritis Rheum. 2008, 58, 164-171.
doi: 10.1002/(ISSN)1529-0131 URL |
| 233. |
van Ooij, A. Analysis of porous ingrowth in intervertebral disc prostheses: a nonhuman primate model. Spine (Phila Pa 1976). 2003, 28, 2304.
doi: 10.1097/01.BRS.0000093257.06297.43 URL |
| 234. |
Modic, M. T.; Masaryk, T. J.; Ross, J. S.; Carter, J. R. Imaging of degenerative disk disease. Radiology. 1988, 168, 177-186.
doi: 10.1148/radiology.168.1.3289089 URL |
| 235. |
Kimura, T.; Nakata, K.; Tsumaki, N.; Miyamoto, S.; Matsui, Y.; Ebara, S.; Ochi, T. Progressive degeneration of articular cartilage and intervertebral discs. An experimental study in transgenic mice bearing a type IX collagen mutation. Int Orthop. 1996, 20, 177-181.
doi: 10.1007/s002640050058 URL |
| 236. |
Watanabe, H.; Nakata, K.; Kimata, K.; Nakanishi, I.; Yamada, Y. Dwarfism and age-associated spinal degeneration of heterozygote cmd mice defective in aggrecan. Proc Natl Acad Sci U S A. 1997, 94, 6943-6947.
doi: 10.1073/pnas.94.13.6943 URL |
| 237. |
Watanabe, H.; Yamada, Y. Chondrodysplasia of gene knockout mice for aggrecan and link protein. Glycoconj J. 2002, 19, 269-273.
doi: 10.1023/A:1025344332099 URL |
| 238. |
Mason, R. M.; Palfrey, A. J. Intervertebral disc degeneration in adult mice with hereditary kyphoscoliosis. J Orthop Res. 1984, 2, 333-338.
doi: 10.1002/(ISSN)1554-527X URL |
| 239. |
Sahlman, J.; Inkinen, R.; Hirvonen, T.; Lammi, M. J.; Lammi, P. E.; Nieminen, J.; Lapveteläinen, T.; Prockop, D. J.; Arita, M.; Li, S. W.; Hyttinen, M. M.; Helminen, H. J.; Puustjärvi, K. Premature vertebral endplate ossification and mild disc degeneration in mice after inactivation of one allele belonging to the Col2a1 gene for type II collagen. Spine (Phila Pa 1976). 2001, 26, 2558-2565.
doi: 10.1097/00007632-200112010-00008 URL |
| 240. |
Hamrick, M. W.; Pennington, C.; Byron, C. D. Bone architecture and disc degeneration in the lumbar spine of mice lacking GDF-8 (myostatin). J Orthop Res. 2003, 21, 1025-1032.
doi: 10.1016/S0736-0266(03)00105-0 URL |
| 241. |
Sweet, H. O.; Green, M. C. Progressive ankylosis, a new skeletal mutation in the mouse. J Hered. 1981, 72, 87-93.
doi: 10.1093/oxfordjournals.jhered.a109459 URL |
| 242. |
Furuya, S.; Ohtsuki, T.; Yabe, Y.; Hosoda, Y. Ultrastructural study on calcification of cartilage: comparing ICR and twy mice. J Bone Miner Metab. 2000, 18, 140-147.
doi: 10.1007/s007740050104 URL |
| 243. |
Hammer, R. E.; Maika, S. D.; Richardson, J. A.; Tang, J. P.; Taurog, J. D. Spontaneous inflammatory disease in transgenic rats expressing HLA-B27 and human beta 2m: an animal model of HLA-B27-associated human disorders. Cell. 1990, 63, 1099-1112.
doi: 10.1016/0092-8674(90)90512-D URL |
| 244. |
Gruber, H. E.; Ashraf, N.; Kilburn, J.; Williams, C.; Norton, H. J.; Gordon, B. E.; Hanley, E. N., Jr. Vertebral endplate architecture and vascularization: application of micro-computerized tomography, a vascular tracer, and immunocytochemistry in analyses of disc degeneration in the aging sand rat. Spine (Phila Pa 1976). 2005, 30, 2593-2600.
doi: 10.1097/01.brs.0000187877.30149.83 URL |
| 245. | Cole, T. C.; Ghosh, P.; Taylor, T. K. Variations of the proteoglycans of the canine intervertebral disc with ageing. Biochim Biophys Acta. 1986, 880, 209-219. |
| 246. |
Ghosh, P.; Taylor, T. K.; Braund, K. G.; Larsen, L. H. The collagenous and non-collagenous protein of the canine intervertebral disc and their variation with age, spinal level and breed. Gerontology. 1976, 22, 124-134.
doi: 10.1159/000212129 URL |
| 247. |
Melrose, J.; Taylor, T. K.; Ghosh, P. Variation in intervertebral disc serine proteinase inhibitory proteins with ageing in a chondrodystrophoid (beagle) and a non-chondrodystrophoid (greyhound) canine breed. Gerontology. 1996, 42, 322-329.
doi: 10.1159/000213810 URL |
| 248. |
Togni, A.; Kranenburg, H. J.; Morgan, J. P.; Steffen, F. Radiographic and MRI characteristics of lumbar disseminated idiopathic spinal hyperostosis and spondylosis deformans in dogs. J Small Anim Pract. 2014, 55, 343-349.
doi: 10.1111/jsap.12218 URL |
| 249. |
Silberberg, R.; Gerritsen, G. Aging changes in intervertebral discs and spondylosis in Chinese hamsters. Diabetes. 1976, 25, 477-483.
doi: 10.2337/diab.25.6.477 URL |
| 250. |
Grootaert, M. O. J.; Finigan, A.; Figg, N. L.; Uryga, A. K.; Bennett, M. R. SIRT6 protects smooth muscle cells from senescence and reduces atherosclerosis. Circ Res. 2021, 128, 474-491.
doi: 10.1161/CIRCRESAHA.120.318353 URL |
| 251. |
Nasto, L. A.; Wang, D.; Robinson, A. R.; Clauson, C. L.; Ngo, K.; Dong, Q.; Roughley, P.; Epperly, M.; Huq, S. M.; Pola, E.; Sowa, G.; Robbins, P. D.; Kang, J.; Niedernhofer, L. J.; Vo, N. V. Genotoxic stress accelerates age-associated degenerative changes in intervertebral discs. Mech Ageing Dev. 2013, 134, 35-42.
doi: 10.1016/j.mad.2012.11.002 URL |
| 252. | Han, Y.; Zhou, C. M.; Shen, H.; Tan, J.; Dong, Q.; Zhang, L.; McGowan, S. J.; Zhao, J.; Sowa, G. A.; Kang, J. D.; Niedernhofer, L. J.; Robbins, P. D.; Vo, N. N. Attenuation of ataxia telangiectasia mutated signalling mitigates age-associated intervertebral disc degeneration. Aging Cell. 2020, 19, e13162. |
| 253. |
Vo, N.; Seo, H. Y.; Robinson, A.; Sowa, G.; Bentley, D.; Taylor, L.; Studer, R.; Usas, A.; Huard, J.; Alber, S.; Watkins, S. C.; Lee, J.; Coehlo, P.; Wang, D.; Loppini, M.; Robbins, P. D.; Niedernhofer, L. J.; Kang, J. Accelerated aging of intervertebral discs in a mouse model of progeria. J Orthop Res. 2010, 28, 1600-1607.
doi: 10.1002/jor.21153 URL |
| 254. |
Bradshaw, A. D.; Sage, E. H. SPARC, a matricellular protein that functions in cellular differentiation and tissue response to injury. J Clin Invest. 2001, 107, 1049-1054.
doi: 10.1172/JCI12939 URL |
| 255. |
Millecamps, M.; Tajerian, M.; Sage, E. H.; Stone, L. S. Behavioral signs of chronic back pain in the SPARC-null mouse. Spine (Phila Pa 1976). 2011, 36, 95-102.
doi: 10.1097/BRS.0b013e3181cd9d75 URL |
| 256. |
Millecamps, M.; Tajerian, M.; Naso, L.; Sage, H. E.; Stone, L. S. Lumbar intervertebral disc degeneration associated with axial and radiating low back pain in ageing SPARC-null mice. Pain. 2012, 153, 1167-1179.
doi: 10.1016/j.pain.2012.01.027 URL |
| 257. |
Taunton, J. E.; Ryan, M. B.; Clement, D. B.; McKenzie, D. C.; Lloyd-Smith, D. R.; Zumbo, B. D. A retrospective case-control analysis of 2002 running injuries. Br J Sports Med. 2002, 36, 95-101.
doi: 10.1136/bjsm.36.2.95 URL |
| 258. |
Luoma, K.; Riihimäki, H.; Luukkonen, R.; Raininko, R.; Viikari-Juntura, E.; Lamminen, A. Low back pain in relation to lumbar disc degeneration. Spine (Phila Pa 1976). 2000, 25, 487-492.
doi: 10.1097/00007632-200002150-00016 URL |
| 259. |
Dubravcic-Simunjak, S.; Pecina, M.; Kuipers, H.; Moran, J.; Haspl, M. The incidence of injuries in elite junior figure skaters. Am J Sports Med. 2003, 31, 511-517.
doi: 10.1177/03635465030310040601 URL |
| 260. |
Chen, S.; Lv, X.; Hu, B.; Zhao, L.; Li, S.; Li, Z.; Qing, X.; Liu, H.; Xu, J.; Shao, Z. Critical contribution of RIPK1 mediated mitochondrial dysfunction and oxidative stress to compression-induced rat nucleus pulposus cells necroptosis and apoptosis. Apoptosis. 2018, 23, 299-313.
doi: 10.1007/s10495-018-1455-x URL |
| 261. | Hu, Y.; Huang, L.; Shen, M.; Liu, Y.; Liu, G.; Wu, Y.; Ding, F.; Ma, K.; Wang, W.; Zhang, Y.; Shao, Z.; Cai, X.; Xiong, L. Pioglitazone protects compression-mediated apoptosis in nucleus pulposus mesenchymal stem cells by suppressing oxidative stress. Oxid Med Cell Longev. 2019, 2019, 4764071. |
| 262. |
Huang, D.; Peng, Y.; Li, Z.; Chen, S.; Deng, X.; Shao, Z.; Ma, K. Compression-induced senescence of nucleus pulposus cells by promoting mitophagy activation via the PINK1/PARKIN pathway. J Cell Mol Med. 2020, 24, 5850-5864.
doi: 10.1111/jcmm.v24.10 URL |
| 263. |
Xiang, Q.; Kang, L.; Wang, J.; Liao, Z.; Song, Y.; Zhao, K.; Wang, K.; Yang, C.; Zhang, Y. CircRNA-CIDN mitigated compression loading-induced damage in human nucleus pulposus cells via miR-34a-5p/SIRT1 axis. EBioMedicine. 2020, 53, 102679.
doi: 10.1016/j.ebiom.2020.102679 URL |
| 264. |
Lundin, O.; Ekström, L.; Hellström, M.; Holm, S.; Swärd, L. Exposure of the porcine spine to mechanical compression: differences in injury pattern between adolescents and adults. Eur Spine J. 2000, 9, 466-471.
doi: 10.1007/s005860000164 URL |
| 265. |
Thoreson, O.; Baranto, A.; Ekström, L.; Holm, S.; Hellström, M.; Swärd, L. The immediate effect of repeated loading on the compressive strength of young porcine lumbar spine. Knee Surg Sports Traumatol Arthrosc. 2010, 18, 694-701.
doi: 10.1007/s00167-009-1001-z URL |
| 266. |
Cassidy, J. D.; Yong-Hing, K.; Kirkaldy-Willis, W. H.; Wilkinson, A. A. A study of the effects of bipedism and upright posture on the lumbosacral spine and paravertebral muscles of the Wistar rat. Spine (Phila Pa 1976). 1988, 13, 301-308.
doi: 10.1097/00007632-198803000-00013 URL |
| 267. | Peng, B.; Hou, S.; Shi, Q.; Jia, L. The relationship between cartilage end-plate calcification and disc degeneration: an experimental study. Chin Med J (Engl). 2001, 114, 308-312. |
| 268. |
Stokes, I. A.; Counts, D. F.; Frymoyer, J. W. Experimental instability in the rabbit lumbar spine. Spine (Phila Pa 1976). 1989, 14, 68-72.
doi: 10.1097/00007632-198901000-00014 URL |
| 269. |
Kaigle, A. M.; Holm, S. H.; Hansson, T. H. Experimental instability in the lumbar spine. Spine (Phila Pa 1976). 1995, 20, 421-430.
doi: 10.1097/00007632-199502001-00004 URL |
| 270. | Hadjipavlou, A. G.; Simmons, J. W.; Yang, J. P.; Bi, L. X.; Ansari, G. A.; Kaphalia, B. S.; Simmons, D. J.; Nicodemus, C. L.; Necessary, J. T.; Lane, R.; Esch, O. Torsional injury resulting in disc degeneration: I. An in vivo rabbit model. J Spinal Disord. 1998, 11, 312-317. |
| 271. | Higuchi, M.; Abe, K.; Kaneda, K. Changes in the nucleus pulposus of the intervertebral disc in bipedal mice. A light and electron microscopic study. Clin Orthop Relat Res. 1983, 251-257. |
| 272. |
Hutton, W. C.; Yoon, S. T.; Elmer, W. A.; Li, J.; Murakami, H.; Minamide, A.; Akamaru, T. Effect of tail suspension (or simulated weightlessness) on the lumbar intervertebral disc: study of proteoglycans and collagen. Spine (Phila Pa 1976). 2002, 27, 1286-1290.
doi: 10.1097/00007632-200206150-00008 URL |
| 273. |
Stokes, I. A.; McBride, C. A.; Aronsson, D. D.; Roughley, P. J. Metabolic effects of angulation, compression, and reduced mobility on annulus fibrosis in a model of altered mechanical environment in scoliosis. Spine Deform. 2013, 1, 161-170.
doi: 10.1016/j.jspd.2013.02.001 URL |
| 274. |
Pedrini-Mille, A.; Maynard, J. A.; Durnova, G. N.; Kaplansky, A. S.; Pedrini, V. A.; Chung, C. B.; Fedler-Troester, J . Effects of microgravity on the composition of the intervertebral disk. J Appl Physiol (1985). 1992, 73, 26S-32S.
doi: 10.1152/jappl.1992.73.2.S26 URL |
| 275. |
Bailey, J. F.; Hargens, A. R.; Cheng, K. K.; Lotz, J. C. Effect of microgravity on the biomechanical properties of lumbar and caudal intervertebral discs in mice. J Biomech. 2014, 47, 2983-2988.
doi: 10.1016/j.jbiomech.2014.07.005 URL |
| 276. |
Zheng, L.; Cao, Y.; Ni, S.; Qi, H.; Ling, Z.; Xu, X.; Zou, X.; Wu, T.; Deng, R.; Hu, B.; Gao, B.; Chen, H.; Li, Y.; Zhu, J.; Tintani, F.; Demehri, S.; Jain, A.; Kebaish, K. M.; Liao, S.; Séguin, C. A.; Crane, J. L.; Wan, M.; Lu, H.; Sponseller, P. D.; Riley, L. H., 3rd; Zhou, X.; Hu, J.; Cao, X. Ciliary parathyroid hormone signaling activates transforming growth factor-β to maintain intervertebral disc homeostasis during aging. Bone Res. 2018, 6, 21.
doi: 10.1038/s41413-018-0022-y URL |
| 277. |
Xiao, Z. F.; Su, G. Y.; Hou, Y.; Chen, S. D.; Zhao, B. D.; He, J. B.; Zhang, J. H.; Chen, Y. J.; Lin, D. K. Mechanics and biology interact in intervertebral disc degeneration: a novel composite mouse model. Calcif Tissue Int. 2020, 106, 401-414.
doi: 10.1007/s00223-019-00644-8 URL |
| 278. |
Fu, F.; Bao, R.; Yao, S.; Zhou, C.; Luo, H.; Zhang, Z.; Zhang, H.; Li, Y.; Yan, S.; Yu, H.; Du, W.; Yang, Y.; Jin, H.; Tong, P.; Sun, Z. T.; Yue, M.; Chen, D.; Wu, C.; Ruan, H. Aberrant spinal mechanical loading stress triggers intervertebral disc degeneration by inducing pyroptosis and nerve ingrowth. Sci Rep. 2021, 11, 772.
doi: 10.1038/s41598-020-80756-6 URL |
| 279. | Chen, L. H.; Lai, P. L.; Tai, C. L.; Niu, C. C.; Fu, T. S.; Chen, W. J. The effect of interspinous ligament integrity on adjacent segment instability after lumbar instrumentation and laminectomy--an experimental study in porcine model. Biomed Mater Eng. 2006, 16, 261-267. |
| 280. |
Tai, C. L.; Hsieh, P. H.; Chen, W. P.; Chen, L. H.; Chen, W. J.; Lai, P. L. Biomechanical comparison of lumbar spine instability between laminectomy and bilateral laminotomy for spinal stenosis syndrome - an experimental study in porcine model. BMC Musculoskelet Disord. 2008, 9, 84.
doi: 10.1186/1471-2474-9-84 URL |
| 281. |
Kim, J.; Yang, S. J.; Kim, H.; Kim, Y.; Park, J. B.; Dubose, C.; Lim, T. H. Effect of shear force on intervertebral disc (IVD) degeneration: an in vivo rat study. Ann Biomed Eng. 2012, 40, 1996-2004.
doi: 10.1007/s10439-012-0570-z URL |
| 282. |
Xia, D. D.; Lin, S. L.; Wang, X. Y.; Wang, Y. L.; Xu, H. M.; Zhou, F.; Tan, J. Effects of shear force on intervertebral disc: an in vivo rabbit study. Eur Spine J. 2015, 24, 1711-1719.
doi: 10.1007/s00586-015-3816-2 URL |
| 283. |
Court, C.; Colliou, O. K.; Chin, J. R.; Liebenberg, E.; Bradford, D. S.; Lotz, J. C. The effect of static in vivo bending on the murine intervertebral disc. Spine J. 2001, 1, 239-245.
doi: 10.1016/S1529-9430(01)00056-0 URL |
| 284. |
Gullbrand, S. E.; Peterson, J.; Ahlborn, J.; Mastropolo, R.; Fricker, A.; Roberts, T. T.; Abousayed, M.; Lawrence, J. P.; Glennon, J. C.; Ledet, E. H. ISSLS Prize Winner: dynamic loading-induced convective transport enhances intervertebral disc nutrition. Spine (Phila Pa 1976). 2015, 40, 1158-1164.
doi: 10.1097/BRS.0000000000001012 URL |
| 285. |
Holguin, N.; Uzer, G.; Chiang, F. P.; Rubin, C.; Judex, S. Brief daily exposure to low-intensity vibration mitigates the degradation of the intervertebral disc in a frequency-specific manner. J Appl Physiol (1985). 2011, 111, 1846-1853.
doi: 10.1152/japplphysiol.00846.2011 URL |
| 286. |
Yasuoka, H.; Asazuma, T.; Nakanishi, K.; Yoshihara, Y.; Sugihara, A.; Tomiya, M.; Okabayashi, T.; Nemoto, K. Effects of reloading after simulated microgravity on proteoglycan metabolism in the nucleus pulposus and anulus fibrosus of the lumbar intervertebral disc: an experimental study using a rat tail suspension model. Spine (Phila Pa 1976). 2007, 32, E734-740.
doi: 10.1097/BRS.0b013e31815b7e51 URL |
| 287. |
Jensen, G. M. Biomechanics of the lumbar intervertebral disk: a review. Phys Ther. 1980, 60, 765-773.
doi: 10.1093/ptj/60.6.765 URL |
| 288. |
Court, C.; Chin, J. R.; Liebenberg, E.; Colliou, O. K.; Lotz, J. C. Biological and mechanical consequences of transient intervertebral disc bending. Eur Spine J. 2007, 16, 1899-1906.
doi: 10.1007/s00586-007-0476-x URL |
| 289. |
Barbir, A.; Godburn, K. E.; Michalek, A. J.; Lai, A.; Monsey, R. D.; Iatridis, J. C. Effects of torsion on intervertebral disc gene expression and biomechanics, using a rat tail model. Spine (Phila Pa 1976). 2011, 36, 607-614.
doi: 10.1097/BRS.0b013e3181d9b58b URL |
| 290. |
Lotz, J. C.; Colliou, O. K.; Chin, J. R.; Duncan, N. A.; Liebenberg, E. Compression-induced degeneration of the intervertebral disc: an in vivo mouse model and finite-element study. Spine (Phila Pa 1976). 1998, 23, 2493-2506.
doi: 10.1097/00007632-199812010-00004 URL |
| 291. |
Kawakami, M.; Hashizume, H.; Nishi, H.; Matsumoto, T.; Tamaki, T.; Kuribayashi, K. Comparison of neuropathic pain induced by the application of normal and mechanically compressed nucleus pulposus to lumbar nerve roots in the rat. J Orthop Res. 2003, 21, 535-539.
doi: 10.1016/S0736-0266(02)00192-4 URL |
| 292. |
Liu, Z.; Zhou, Q.; Zheng, J.; Li, C.; Zhang, W.; Zhang, X. A novel in vivo mouse intervertebral disc degeneration model induced by compressive suture. Exp Cell Res. 2021, 398, 112359.
doi: 10.1016/j.yexcr.2020.112359 URL |
| 293. |
Hutton, W. C.; Toribatake, Y.; Elmer, W. A.; Ganey, T. M.; Tomita, K.; Whitesides, T. E. The effect of compressive force applied to the intervertebral disc in vivo. A study of proteoglycans and collagen. Spine (Phila Pa 1976). 1998, 23, 2524-2537.
doi: 10.1097/00007632-199812010-00007 URL |
| 294. | Osterman, K.; Osterman, H. Experimental lumbar spondylolisthesis in growing rabbits. Clin Orthop Relat Res. 1996, 274-280. |
| 295. |
Lipson, S. J.; Muir, H. Vertebral osteophyte formation in experimental disc degeneration. Morphologic and proteoglycan changes over time. Arthritis Rheum. 1980, 23, 319-324.
doi: 10.1002/(ISSN)1529-0131 URL |
| 296. |
Lipson, S. J.; Muir, H. 1980 Volvo award in basic science. Proteoglycans in experimental intervertebral disc degeneration. Spine (Phila Pa 1976). 1981, 6, 194-210.
doi: 10.1097/00007632-198105000-00002 URL |
| 297. |
Latorre, A.; Albareda, J.; Castiella, T.; Lasierra, J. M.; Seral, F. Experimental model of multidirectional disc hernia in rats. Int Orthop. 1998, 22, 44-48.
doi: 10.1007/s002640050206 URL |
| 298. |
Kim, K. S.; Yoon, S. T.; Li, J.; Park, J. S.; Hutton, W. C. Disc degeneration in the rabbit: a biochemical and radiological comparison between four disc injury models. Spine (Phila Pa 1976). 2005, 30, 33-37.
doi: 10.1097/01.brs.0000149191.02304.9b URL |
| 299. | Urayama, S. Histological and ultrastructural study of degeneration of the lumbar intervertebral disc in the rabbit following nucleotomy, with special reference to the topographical distribution pattern of the degeneration. Nihon Seikeigeka Gakkai Zasshi. 1986, 60, 649-662. |
| 300. |
Takaishi, H.; Nemoto, O.; Shiota, M.; Kikuchi, T.; Yamada, H.; Yamagishi, M.; Yabe, Y. Type-II collagen gene expression is transiently upregulated in experimentally induced degeneration of rabbit intervertebral disc. J Orthop Res. 1997, 15, 528-538.
doi: 10.1002/(ISSN)1554-527X URL |
| 301. |
Melrose, J.; Ghosh, P.; Taylor, T. K.; Hall, A.; Osti, O. L.; Vernon-Roberts, B.; Fraser, R. D. A longitudinal study of the matrix changes induced in the intervertebral disc by surgical damage to the annulus fibrosus. J Orthop Res. 1992, 10, 665-676.
doi: 10.1002/(ISSN)1554-527X URL |
| 302. | Melrose, J.; Ghosh, P.; Taylor, T. K.; Latham, J.; Moore, R. Topographical variation in the catabolism of aggrecan in an ovine annular lesion model of experimental disc degeneration. J Spinal Disord. 1997, 10, 55-67. |
| 303. |
Melrose, J.; Taylor, T. K.; Ghosh, P. The serine proteinase inhibitory proteins of the chondrodystrophoid (beagle) and non-chondrodystrophoid (greyhound) canine intervertebral disc. Electrophoresis. 1997, 18, 1059-1063.
doi: 10.1002/(ISSN)1522-2683 URL |
| 304. |
Fazzalari, N. L.; Costi, J. J.; Hearn, T. C.; Fraser, R. D.; Vernon-Roberts, B.; Hutchinson, J.; Manthey, B. A.; Parkinson, I. H.; Sinclair, C. Mechanical and pathologic consequences of induced concentric anular tears in an ovine model. Spine (Phila Pa 1976). 2001, 26, 2575-2581.
doi: 10.1097/00007632-200112010-00010 URL |
| 305. |
Schollum, M. L.; Appleyard, R. C.; Little, C. B.; Melrose, J. A detailed microscopic examination of alterations in normal anular structure induced by mechanical destabilization in an ovine model of disc degeneration. Spine (Phila Pa 1976). 2010, 35, 1965-1973.
doi: 10.1097/BRS.0b013e3181e0f085 URL |
| 306. |
Kaigle, A. M.; Holm, S. H.; Hansson, T. H. 1997 Volvo Award winner in biomechanical studies. Kinematic behavior of the porcine lumbar spine: a chronic lesion model. Spine (Phila Pa 1976). 1997, 22, 2796-2806.
doi: 10.1097/00007632-199712150-00002 URL |
| 307. |
Zhu, J.; Xia, K.; Yu, W.; Wang, Y.; Hua, J.; Liu, B.; Gong, Z.; Wang, J.; Xu, A.; You, Z.; Chen, Q.; Li, F.; Tao, H.; Liang, C. Sustained release of GDF5 from a designed coacervate attenuates disc degeneration in a rat model. Acta Biomater. 2019, 86, 300-311.
doi: 10.1016/j.actbio.2019.01.028 URL |
| 308. |
Olsewski, J. M.; Schendel, M. J.; Wallace, L. J.; Ogilvie, J. W.; Gundry, C. R. Magnetic resonance imaging and biological changes in injured intervertebral discs under normal and increased mechanical demands. Spine (Phila Pa 1976). 1996, 21, 1945-1951.
doi: 10.1097/00007632-199609010-00001 URL |
| 309. |
Aoki, Y.; Akeda, K.; An, H.; Muehleman, C.; Takahashi, K.; Moriya, H.; Masuda, K. Nerve fiber ingrowth into scar tissue formed following nucleus pulposus extrusion in the rabbit anular-puncture disc degeneration model: effects of depth of puncture. Spine (Phila Pa 1976). 2006, 31, E774-780.
doi: 10.1097/01.brs.0000238681.71537.41 URL |
| 310. |
Masuda, K.; Imai, Y.; Okuma, M.; Muehleman, C.; Nakagawa, K.; Akeda, K.; Thonar, E.; Andersson, G.; An, H. S. Osteogenic protein-1 injection into a degenerated disc induces the restoration of disc height and structural changes in the rabbit anular puncture model. Spine (Phila Pa 1976). 2006, 31, 742-754.
doi: 10.1097/01.brs.0000206358.66412.7b URL |
| 311. |
Mwale, F.; Masuda, K.; Pichika, R.; Epure, L. M.; Yoshikawa, T.; Hemmad, A.; Roughley, P. J.; Antoniou, J. The efficacy of Link N as a mediator of repair in a rabbit model of intervertebral disc degeneration. Arthritis Res Ther. 2011, 13, R120.
doi: 10.1186/ar3423 URL |
| 312. | Liu, Y.; Lin, J.; Wu, X.; Guo, X.; Sun, H.; Yu, B.; Shen, J.; Bai, J.; Chen, Z.; Yang, H.; Geng, D.; Mao, H. Aspirin-mediated attenuation of intervertebral disc degeneration by ameliorating reactive oxygen species in vivo and in vitro. Oxid Med Cell Longev. 2019, 2019, 7189854. |
| 313. |
Han, B.; Zhu, K.; Li, F. C.; Xiao, Y. X.; Feng, J.; Shi, Z. L.; Lin, M.; Wang, J.; Chen, Q. X. A simple disc degeneration model induced by percutaneous needle puncture in the rat tail. Spine (Phila Pa 1976). 2008, 33, 1925-1934.
doi: 10.1097/BRS.0b013e31817c64a9 URL |
| 314. |
Makino, H.; Seki, S.; Yahara, Y.; Shiozawa, S.; Aikawa, Y.; Motomura, H.; Nogami, M.; Watanabe, K.; Sainoh, T.; Ito, H.; Tsumaki, N.; Kawaguchi, Y.; Yamazaki, M.; Kimura, T. A selective inhibition of c-Fos/activator protein-1 as a potential therapeutic target for intervertebral disc degeneration and associated pain. Sci Rep. 2017, 7, 16983.
doi: 10.1038/s41598-017-17289-y URL |
| 315. |
Henriksson, H. B.; Svanvik, T.; Jonsson, M.; Hagman, M.; Horn, M.; Lindahl, A.; Brisby, H. Transplantation of human mesenchymal stems cells into intervertebral discs in a xenogeneic porcine model. Spine (Phila Pa 1976). 2009, 34, 141-148.
doi: 10.1097/BRS.0b013e31818f8c20 URL |
| 316. |
van Heeswijk, V. M.; Thambyah, A.; Robertson, P. A.; Broom, N. D. Does an annular puncture influence the herniation path?: An in vitro mechanical and structural investigation. Spine (Phila Pa 1976). 2018, 43, 467-476.
doi: 10.1097/BRS.0000000000002336 URL |
| 317. |
Piazza, M.; Peck, S. H.; Gullbrand, S. E.; Bendigo, J. R.; Arginteanu, T.; Zhang, Y.; Smith, H. E.; Malhotra, N. R.; Smith, L. J. Quantitative MRI correlates with histological grade in a percutaneous needle injury mouse model of disc degeneration. J Orthop Res. 2018, 36, 2771-2779.
doi: 10.1002/jorr.v36.10 URL |
| 318. |
Liang, H.; Ma, S. Y.; Feng, G.; Shen, F. H.; Joshua, Li, X. Therapeutic effects of adenovirus-mediated growth and differentiation factor-5 in a mice disc degeneration model induced by annulus needle puncture. Spine J. 2010, 10, 32-41.
doi: 10.1016/j.spinee.2009.10.006 URL |
| 319. | Hu, M. H.; Yang, K. C.; Chen, Y. J.; Sun, Y. H.; Lin, F. H.; Yang, S. H. Optimization of puncture injury to rat caudal disc for mimicking early degeneration of intervertebral disc. J Orthop Res. 2018, 36, 202-211. |
| 320. |
Zhang, H.; La Marca, F.; Hollister, S. J.; Goldstein, S. A.; Lin, C. Y. Developing consistently reproducible intervertebral disc degeneration at rat caudal spine by using needle puncture. J Neurosurg Spine. 2009, 10, 522-530.
doi: 10.3171/2009.2.SPINE08925 URL |
| 321. |
Hsieh, A. H.; Hwang, D.; Ryan, D. A.; Freeman, A. K.; Kim, H. Degenerative anular changes induced by puncture are associated with insufficiency of disc biomechanical function. Spine (Phila Pa 1976). 2009, 34, 998-1005.
doi: 10.1097/BRS.0b013e31819c09c4 URL |
| 322. |
Zhang, K. B.; Zheng, Z. M.; Liu, H.; Liu, X. G. The effects of punctured nucleus pulposus on lumbar radicular pain in rats: a behavioral and immunohistochemical study. J Neurosurg Spine. 2009, 11, 492-500.
doi: 10.3171/2009.4.SPINE08744 URL |
| 323. |
Zhang, H.; Wang, L.; Park, J. B.; Park, P.; Yang, V. C.; Hollister, S. J.; La Marca, F.; Lin, C. Y. Intradiscal injection of simvastatin retards progression of intervertebral disc degeneration induced by stab injury. Arthritis Res Ther. 2009, 11, R172.
doi: 10.1186/ar2861 URL |
| 324. |
Sadamasu, A.; Sakuma, Y.; Suzuki, M.; Orita, S.; Yamauchi, K.; Inoue, G.; Aoki, Y.; Ishikawa, T.; Miyagi, M.; Kamoda, H.; Kubota, G.; Oikawa, Y.; Inage, K.; Sainoh, T.; Sato, J.; Nakamura, J.; Toyone, T.; Takahashi, K.; Ohtori, S. Upregulation of NaV1.7 in dorsal root ganglia after intervertebral disc injury in rats. Spine (Phila Pa 1976). 2014, 39, E421-426.
doi: 10.1097/BRS.0000000000000229 URL |
| 325. |
Sato, M.; Inage, K.; Sakuma, Y.; Sato, J.; Orita, S.; Yamauchi, K.; Eguchi, Y.; Ochiai, N.; Kuniyoshi, K.; Aoki, Y.; Nakamura, J.; Miyagi, M.; Suzuki, M.; Kubota, G.; Sainoh, T.; Fujimoto, K.; Shiga, Y.; Abe, K.; Kanamoto, H.; Inoue, G.; Takahashi, K.; Ohtori, S. Anti-RANKL antibodies decrease CGRP expression in dorsal root ganglion neurons innervating injured lumbar intervertebral discs in rats. Eur Spine J. 2015, 24, 2017-2022.
doi: 10.1007/s00586-015-4058-z URL |
| 326. |
Horii, M.; Orita, S.; Nagata, M.; Takaso, M.; Yamauchi, K.; Yamashita, M.; Inoue, G.; Eguchi, Y.; Ochiai, N.; Kishida, S.; Aoki, Y.; Ishikawa, T.; Arai, G.; Miyagi, M.; Kamoda, H.; Kuniyoshi, K.; Suzuki, M.; Nakamura, J.; Toyone, T.; Takahashi, K.; Ohtori, S. Direct application of the tumor necrosis factor-α inhibitor, etanercept, into a punctured intervertebral disc decreases calcitonin gene-related peptide expression in rat dorsal root ganglion neurons. Spine (Phila Pa 1976). 2011, 36, E80-85.
doi: 10.1097/BRS.0b013e3181d4be3c URL |
| 327. |
Li, D.; Yang, H.; Huang, Y.; Wu, Y.; Sun, T.; Li, X. Lumbar intervertebral disc puncture under C-arm fluoroscopy: a new rat model of lumbar intervertebral disc degeneration. Exp Anim. 2014, 63, 227-234.
doi: 10.1538/expanim.63.227 URL |
| 328. |
Liu, W.; Xia, P.; Feng, J.; Kang, L.; Huang, M.; Wang, K.; Song, Y.; Li, S.; Wu, X.; Yang, S.; Yang, C. MicroRNA-132 upregulation promotes matrix degradation in intervertebral disc degeneration. Exp Cell Res. 2017, 359, 39-49.
doi: 10.1016/j.yexcr.2017.08.011 URL |
| 329. |
Liao, J. C. Cell therapy using bone marrow-derived stem cell overexpressing BMP-7 for degenerative discs in a rat tail disc model. Int J Mol Sci. 2016, 17, 147.
doi: 10.3390/ijms17020147 URL |
| 330. |
Tian, Z.; Ma, X.; Yasen, M.; Mauck, R. L.; Qin, L.; Shofer, F. S.; Smith, L. J.; Pacifici, M.; Enomoto-Iwamoto, M.; Zhang, Y. Intervertebral disc degeneration in a percutaneous mouse tail injury model. Am J Phys Med Rehabil. 2018, 97, 170-177.
doi: 10.1097/PHM.0000000000000818 URL |
| 331. |
Zhang, C.; Smith, M. P.; Zhou, G. K.; Lai, A.; Hoy, R. C.; Mroz, V.; Torre, O. M.; Laudier, D. M.; Bradley, E. W.; Westendorf, J. J.; Iatridis, J. C.; Illien-Jünger, S. Phlpp1 is associated with human intervertebral disc degeneration and its deficiency promotes healing after needle puncture injury in mice. Cell Death Dis. 2019, 10, 754.
doi: 10.1038/s41419-019-1985-3 URL |
| 332. |
Xiao, L.; Ding, M.; Zhang, Y.; Chordia, M.; Pan, D.; Shimer, A.; Shen, F.; Glover, D.; Jin, L.; Li, X. A novel modality for functional imaging in acute intervertebral disk herniation via tracking leukocyte infiltration. Mol Imaging Biol. 2017, 19, 703-713.
doi: 10.1007/s11307-016-1038-6 URL |
| 333. |
Tam, V.; Rogers, I.; Chan, D.; Leung, V. Y.; Cheung, K. M. A comparison of intravenous and intradiscal delivery of multipotential stem cells on the healing of injured intervertebral disk. J Orthop Res. 2014, 32, 819-825.
doi: 10.1002/jor.22605 URL |
| 334. |
Yang, F.; Leung, V. Y.; Luk, K. D.; Chan, D.; Cheung, K. M. Injury-induced sequential transformation of notochordal nucleus pulposus to chondrogenic and fibrocartilaginous phenotype in the mouse. J Pathol. 2009, 218, 113-121.
doi: 10.1002/path.v218:1 URL |
| 335. |
Ohta, R.; Tanaka, N.; Nakanishi, K.; Kamei, N.; Nakamae, T.; Izumi, B.; Fujioka, Y.; Ochi, M. Heme oxygenase-1 modulates degeneration of the intervertebral disc after puncture in Bach 1 deficient mice. Eur Spine J. 2012, 21, 1748-1757.
doi: 10.1007/s00586-012-2442-5 URL |
| 336. |
Xin, L.; Xu, W.; Yu, L.; Fan, S.; Wang, W.; Yu, F.; Wang, Z. Effects of annulus defects and implantation of poly(lactic-co-glycolic acid)(PLGA)/fibrin gel scaffolds on nerves ingrowth in a rabbit model of annular injury disc degeneration. J Orthop Surg Res. 2017, 12, 73.
doi: 10.1186/s13018-017-0572-5 URL |
| 337. |
Sobajima, S.; Kompel, J. F.; Kim, J. S.; Wallach, C. J.; Robertson, D. D.; Vogt, M. T.; Kang, J. D.; Gilbertson, L. G. A slowly progressive and reproducible animal model of intervertebral disc degeneration characterized by MRI, X-ray, and histology. Spine (Phila Pa 1976). 2005, 30, 15-24.
doi: 10.1097/01.brs.0000148048.15348.9b URL |
| 338. |
Jacobs, L.; Vo, N.; Coelho, J. P.; Dong, Q.; Bechara, B.; Woods, B.; Hempen, E.; Hartman, R.; Preuss, H.; Balk, J.; Kang, J.; Sowa, G. Glucosamine supplementation demonstrates a negative effect on intervertebral disc matrix in an animal model of disc degeneration. Spine (Phila Pa 1976). 2013, 38, 984-990.
doi: 10.1097/BRS.0b013e318286b31e URL |
| 339. |
Xin, L.; Zhang, C.; Zhong, F.; Fan, S.; Wang, W.; Wang, Z. Minimal invasive annulotomy for induction of disc degeneration and implantation of poly (lactic-co-glycolic acid)(PLGA) plugs for annular repair in a rabbit model. Eur J Med Res. 2016, 21, 7.
doi: 10.1186/s40001-016-0202-4 URL |
| 340. |
Ashinsky, B. G.; Gullbrand, S. E.; Bonnevie, E. D.; Mandalapu, S. A.; Wang, C.; Elliott, D. M.; Han, L.; Mauck, R. L.; Smith, H. E. Multiscale and multimodal structure-function analysis of intervertebral disc degeneration in a rabbit model. Osteoarthritis Cartilage. 2019, 27, 1860-1869.
doi: 10.1016/j.joca.2019.07.016 URL |
| 341. |
Shi, P.; Chee, A.; Liu, W.; Chou, P. H.; Zhu, J.; An, H. S. Therapeutic effects of cell therapy with neonatal human dermal fibroblasts and rabbit dermal fibroblasts on disc degeneration and inflammation. Spine J. 2019, 19, 171-181.
doi: 10.1016/j.spinee.2018.08.005 URL |
| 342. |
Zhou, R. P.; Zhang, Z. M.; Wang, L.; Huang, M. J.; Zheng, X. C.; Cui, Y. N.; Yin, M.; Wang, X. K.; Yao, N. Z.; Chen, T. Y.; Chen, J.; Bai, X. C.; Jin, D. D. Establishing a disc degeneration model using computed tomography-guided percutaneous puncture technique in the rabbit. J Surg Res. 2013, 181, e65-74.
doi: 10.1016/j.jss.2012.07.027 URL |
| 343. | Luo, T. D.;.; Marquez-Lara, A.; Zabarsky, Z. K.; Vines, J. B.; Mowry, K. C.; Jinnah, A. H.; Ma, X.; Berwick, B. W.; Willey, J. S.; Li, Z.; Smith, T. L.; O’Gara, T. J. A percutaneous, minimally invasive annulus fibrosus needle puncture model of intervertebral disc degeneration in rabbits. J Orthop Surg (Hong Kong). 2018, 26, 2309499018792715. |
| 344. |
Luo, T. D.; Vines, J. B.; Zabarsky, Z. K.; Mowry, K. C.; Marquez-Lara, A.; Jinnah, A. H.; Ma, X.; Berwick, B. W.; Willey, J. S.; Smith, T. L.; Li, Z.; O’Gara, T. J. Evaluation of percutaneous intradiscal amniotic suspension allograft in a rabbit model of intervertebral disc degeneration. Spine (Phila Pa 1976). 2019, 44, E329-E337.
doi: 10.1097/BRS.0000000000002851 URL |
| 345. |
Kim, D. W.; Chun, H. J.; Lee, S. K. Percutaneous needle puncture technique to create a rabbit model with traumatic degenerative disk disease. World Neurosurg. 2015, 84, 438-445.
doi: 10.1016/j.wneu.2015.03.066 URL |
| 346. |
Chun, H. J.; Kim, Y. S.; Kim, B. K.; Kim, E. H.; Kim, J. H.; Do, B. R.; Hwang, S. J.; Hwang, J. Y.; Lee, Y. K. Transplantation of human adipose-derived stem cells in a rabbit model of traumatic degeneration of lumbar discs. World Neurosurg. 2012, 78, 364-371.
doi: 10.1016/j.wneu.2011.12.084 URL |
| 347. |
Yang, H.; Wu, J.; Liu, J.; Ebraheim, M.; Castillo, S.; Liu, X.; Tang, T.; Ebraheim, N. A. Transplanted mesenchymal stem cells with pure fibrinous gelatin-transforming growth factor-beta1 decrease rabbit intervertebral disc degeneration. Spine J. 2010, 10, 802-810.
doi: 10.1016/j.spinee.2010.06.019 URL |
| 348. |
Chik, T. K.; Ma, X. Y.; Choy, T. H.; Li, Y. Y.; Diao, H. J.; Teng, W. K.; Han, S. J.; Cheung, K. M.; Chan, B. P. Photochemically crosslinked collagen annulus plug: a potential solution solving the leakage problem of cell-based therapies for disc degeneration. Acta Biomater. 2013, 9, 8128-8139.
doi: 10.1016/j.actbio.2013.05.034 URL |
| 349. |
Li, Z.; Zhang, K.; Li, X.; Pan, H.; Li, S.; Chen, F.; Zhang, J.; Zheng, Z.; Wang, J.; Liu, H. Wnt5a suppresses inflammation-driven intervertebral disc degeneration via a TNF-α/NF-κB-Wnt5a negative-feedback loop. Osteoarthritis Cartilage. 2018, 26, 966-977.
doi: 10.1016/j.joca.2018.04.002 URL |
| 350. |
Elliott, D. M.; Yerramalli, C. S.; Beckstein, J. C.; Boxberger, J. I.; Johannessen, W.; Vresilovic, E. J. The effect of relative needle diameter in puncture and sham injection animal models of degeneration. Spine (Phila Pa 1976). 2008, 33, 588-596.
doi: 10.1097/BRS.0b013e318166e0a2 URL |
| 351. |
Chen, T.; Cheng, X.; Wang, J.; Feng, X.; Zhang, L. Time-Course investigation of intervertebral disc degeneration induced by different sizes of needle punctures in rat tail disc. Med Sci Monit. 2018, 24, 6456-6465.
doi: 10.12659/MSM.910636 URL |
| 352. |
Cunha, C.; Lamas, S.; Gonçalves, R. M.; Barbosa, M. A. Joint analysis of IVD herniation and degeneration by rat caudal needle puncture model. J Orthop Res. 2017, 35, 258-268.
doi: 10.1002/jor.v35.2 URL |
| 353. |
Martin, J. T.; Gorth, D. J.; Beattie, E. E.; Harfe, B. D.; Smith, L. J.; Elliott, D. M. Needle puncture injury causes acute and long-term mechanical deficiency in a mouse model of intervertebral disc degeneration. J Orthop Res. 2013, 31, 1276-1282.
doi: 10.1002/jor.22355 URL |
| 354. |
Omlor, G. W.; Lorenz, S.; Nerlich, A. G.; Guehring, T.; Richter, W. Disc cell therapy with bone-marrow-derived autologous mesenchymal stromal cells in a large porcine disc degeneration model. Eur Spine J. 2018, 27, 2639-2649.
doi: 10.1007/s00586-018-5728-4 URL |
| 355. |
Omlor, G. W.; Nerlich, A. G.; Lorenz, H.; Bruckner, T.; Richter, W.; Pfeiffer, M.; Gühring, T. Injection of a polymerized hyaluronic acid/collagen hydrogel matrix in an in vivo porcine disc degeneration model. Eur Spine J. 2012, 21, 1700-1708.
doi: 10.1007/s00586-012-2291-2 URL |
| 356. |
Omlor, G. W.; Nerlich, A. G.; Wilke, H. J.; Pfeiffer, M.; Lorenz, H.; Schaaf-Keim, M.; Bertram, H.; Richter, W.; Carstens, C.; Guehring, T. A new porcine in vivo animal model of disc degeneration: response of anulus fibrosus cells, chondrocyte-like nucleus pulposus cells, and notochordal nucleus pulposus cells to partial nucleotomy. Spine (Phila Pa 1976). 2009, 34, 2730-2739.
doi: 10.1097/BRS.0b013e3181b723c9 URL |
| 357. |
Yoon, S. H.; Miyazaki, M.; Hong, S. W.; Tow, B.; Morishita, Y.; Hu, M.; Ahn, S. J.; Wang, J. C. A porcine model of intervertebral disc degeneration induced by annular injury characterized with magnetic resonance imaging and histopathological findings. Laboratory investigation. J Neurosurg Spine. 2008, 8, 450-457.
doi: 10.3171/SPI/2008/8/5/450 URL |
| 358. |
Issy, A. C.; Castania, V.; Silveira, J. W.; Nogueira-Barbosa, M. H.; Salmon, C. E.; Del-Bel, E.; Defino, H. L. Does a small size needle puncture cause intervertebral disc changes? Acta Cir Bras. 2015, 30, 574-579.
doi: 10.1590/S0102-865020150080000009 URL |
| 359. |
Keorochana, G.; Johnson, J. S.; Taghavi, C. E.; Liao, J. C.; Lee, K. B.; Yoo, J. H.; Ngo, S. S.; Wang, J. C. The effect of needle size inducing degeneration in the rat caudal disc: evaluation using radiograph, magnetic resonance imaging, histology, and immunohistochemistry. Spine J. 2010, 10, 1014-1023.
doi: 10.1016/j.spinee.2010.08.013 URL |
| 360. |
Zhang, H.; Yang, S.; Wang, L.; Park, P.; La Marca, F.; Hollister, S. J.; Lin, C. Y. Time course investigation of intervertebral disc degeneration produced by needle-stab injury of the rat caudal spine: laboratory investigation. J Neurosurg Spine. 2011, 15, 404-413.
doi: 10.3171/2011.5.SPINE10811 URL |
| 361. |
Yan, J.; Yang, S.; Sun, H.; Guo, D.; Wu, B.; Ji, F.; Zhou, D. Effects of releasing recombinant human growth and differentiation factor-5 from poly(lactic-co-glycolic acid) microspheres for repair of the rat degenerated intervertebral disc. J Biomater Appl. 2014, 29, 72-80.
doi: 10.1177/0885328213515034 URL |
| 362. |
Than, K. D.; Rahman, S. U.; Wang, L.; Khan, A.; Kyere, K. A.; Than, T. T.; Miyata, Y.; Park, Y. S.; La Marca, F.; Kim, H. M.; Zhang, H.; Park, P.; Lin, C. Y. Intradiscal injection of simvastatin results in radiologic, histologic, and genetic evidence of disc regeneration in a rat model of degenerative disc disease. Spine J. 2014, 14, 1017-1028.
doi: 10.1016/j.spinee.2013.11.034 URL |
| 363. |
Castania, V.; Issy, A. C.; Silveira, J. W.; Ferreira, F. R.; Titze-de-Almeida, S. S.; Resende, F. F.; Ferreira, N. R.; Titze-de-Almeida, R.; Defino, H. L.; Del Bel, E. The presence of the neuronal nitric oxide synthase isoform in the intervertebral disk. Neurotox Res. 2017, 31, 148-161.
doi: 10.1007/s12640-016-9676-7 URL |
| 364. |
Li, L.; Zhou, Z.; Li, J.; Fang, J.; Qing, Y.; Tian, T.; Zhang, S.; Wu, G.; Scotti, A.; Cai, K.; Zhu, W. Diffusion kurtosis imaging provides quantitative assessment of the microstructure changes of disc degeneration: an in vivo experimental study. Eur Spine J. 2019, 28, 1005-1013.
doi: 10.1007/s00586-019-05924-3 URL |
| 365. |
Zhan, S.; Wang, K.; Song, Y.; Li, S.; Yin, H.; Luo, R.; Liao, Z.; Wu, X.; Zhang, Y.; Yang, C. Long non-coding RNA HOTAIR modulates intervertebral disc degenerative changes via Wnt/β-catenin pathway. Arthritis Res Ther. 2019, 21, 201.
doi: 10.1186/s13075-019-1986-8 URL |
| 366. |
Zou, F.; Jiang, J.; Lu, F.; Ma, X.; Xia, X.; Wang, L.; Wang, H. Efficacy of intradiscal hepatocyte growth factor injection for the treatment of intervertebral disc degeneration. Mol Med Rep. 2013, 8, 118-122.
doi: 10.3892/mmr.2013.1450 URL |
| 367. |
Grunert, P.; Hudson, K. D.; Macielak, M. R.; Aronowitz, E.; Borde, B. H.; Alimi, M.; Njoku, I.; Ballon, D.; Tsiouris, A. J.; Bonassar, L. J.; Härtl, R. Assessment of intervertebral disc degeneration based on quantitative magnetic resonance imaging analysis: an in vivo study. Spine (Phila Pa 1976). 2014, 39, E369-378.
doi: 10.1097/BRS.0000000000000194 URL |
| 368. |
Jiang, L. B.; Liu, H. X.; Zhou, Y. L.; Sheng, S. R.; Xu, H. Z.; Xue, E. X. An ultrastructural study of chondroptosis: programmed cell death in degenerative intervertebral discs in vivo. J Anat. 2017, 231, 129-139.
doi: 10.1111/joa.2017.231.issue-1 URL |
| 369. |
Chiang, E. R.; Ma, H. L.; Wang, J. P.; Chang, M. C.; Liu, C. L.; Chen, T. H.; Hung, S. C. Use of allogeneic hypoxic mesenchymal stem cells for treating disc degeneration in rabbits. J Orthop Res. 2019, 37, 1440-1450.
doi: 10.1002/jorr.v37.6 URL |
| 370. |
Li, Z.; Liu, H.; Yang, H.; Wang, J.; Wang, H.; Zhang, K.; Ding, W.; Zheng, Z. Both expression of cytokines and posterior annulus fibrosus rupture are essential for pain behavior changes induced by degenerative intervertebral disc: An experimental study in rats. J Orthop Res. 2014, 32, 262-272.
doi: 10.1002/jor.22494 URL |
| 371. |
Ohnishi, T.; Sudo, H.; Iwasaki, K.; Tsujimoto, T.; Ito, Y. M.; Iwasaki, N. In vivo mouse intervertebral disc degeneration model based on a new histological classification. PLoS One. 2016, 11, e0160486.
doi: 10.1371/journal.pone.0160486 URL |
| 372. |
Arai, I.; Mao, G. P.; Otani, K.; Konno, S.; Kikuchi, S.; Olmarker, K. Indomethacin blocks the nucleus pulposus-induced effects on nerve root function. An experimental study in dogs with assessment of nerve conduction and blood flow following experimental disc herniation. Eur Spine J. 2004, 13, 691-694.
doi: 10.1007/s005860100268 URL |
| 373. |
Kawakami, M.; Matsumoto, T.; Tamaki, T. Roles of thromboxane A2 and leukotriene B4 in radicular pain induced by herniated nucleus pulposus. J Orthop Res. 2001, 19, 472-477.
doi: 10.1016/S0736-0266(00)90032-9 URL |
| 374. |
Kiester, D. P.; Williams, J. M.; Andersson, G. B.; Thonar, E. J.; McNeill, T. W. The dose-related effect of intradiscal chymopapain on rabbit intervertebral discs. Spine (Phila Pa 1976). 1994, 19, 747-751.
doi: 10.1097/00007632-199404000-00003 URL |
| 375. |
Ushirozako, H.; Yoshida, G.; Togawa, D.; Omura, T.; Hasegawa, T.; Yamato, Y.; Banno, T.; Arima, H.; Oe, S.; Mihara, Y.; Yamada, T.; Natsume, T.; Ogawa, S.; Awaga, Y.; Takamatsu, H.; Matsuyama, Y. Brain activation in a cynomolgus macaque model of chymopapain-induced discogenic low back pain: a preliminary study. Spine Surg Relat Res. 2019, 3, 368-376.
doi: 10.22603/ssrr.2018-0110 URL |
| 376. |
Wardlaw, D.; Rithchie, I. K.; Sabboubeh, A. F.; Vavdha, M.; Downing, M.; Eastmond, C. J. Prospective randomized trial of chemonucleolysis compared with surgery for soft disc herniation with 1-year, intermediate, and long-term outcome: part II: the radiological outcome. Spine (Phila Pa 1976). 2013, 38, E1058-1064.
doi: 10.1097/BRS.0b013e3182996301 URL |
| 377. |
Haro, H.; Nishiga, M.; Ishii, D.; Shimomoto, T.; Kato, T.; Takenouchi, O.; Koyanagi, S.; Ohba, T.; Komori, H. Experimental chemonucleolysis with recombinant human matrix metalloproteinase 7 in human herniated discs and dogs. Spine J. 2014, 14, 1280-1290.
doi: 10.1016/j.spinee.2013.11.039 URL |
| 378. |
Ishibashi, K.; Iwai, H.; Koga, H. Chemonucleolysis with chondroitin sulfate ABC endolyase as a novel minimally invasive treatment for patients with lumbar intervertebral disc herniation. J Spine Surg. 2019, 5, S115-S121.
doi: 10.21037/jss URL |
| 379. |
Wu, B.; Meng, C.; Wang, H.; Jia, C.; Zhao, Y. Changes of proteoglycan and collagen II of the adjacent intervertebral disc in the cervical instability models. Biomed Pharmacother. 2016, 84, 754-758.
doi: 10.1016/j.biopha.2016.09.077 URL |
| 380. |
Borem, R.; Walters, J.; Madeline, A.; Madeline, L.; Gill, S.; Easley, J.; Mercuri, J. Characterization of chondroitinase-induced lumbar intervertebral disc degeneration in a sheep model intended for assessing biomaterials. J Biomed Mater Res A. 2021, 109, 1232-1246.
doi: 10.1002/jbm.a.v109.7 URL |
| 381. |
Gullbrand, S. E.; Malhotra, N. R.; Schaer, T. P.; Zawacki, Z.; Martin, J. T.; Bendigo, J. R.; Milby, A. H.; Dodge, G. R.; Vresilovic, E. J.; Elliott, D. M.; Mauck, R. L.; Smith, L. J. A large animal model that recapitulates the spectrum of human intervertebral disc degeneration. Osteoarthritis Cartilage. 2017, 25, 146-156.
doi: 10.1016/j.joca.2016.08.006 URL |
| 382. |
Boxberger, J. I.; Orlansky, A. S.; Sen, S.; Elliott, D. M. Reduced nucleus pulposus glycosaminoglycan content alters intervertebral disc dynamic viscoelastic mechanics. J Biomech. 2009, 42, 1941-1946.
doi: 10.1016/j.jbiomech.2009.05.008 URL |
| 383. |
Masters, K. S.; Shah, D. N.; Leinwand, L. A.; Anseth, K. S. Crosslinked hyaluronan scaffolds as a biologically active carrier for valvular interstitial cells. Biomaterials. 2005, 26, 2517-2525.
doi: 10.1016/j.biomaterials.2004.07.018 URL |
| 384. |
MacBarb, R. F.; Makris, E. A.; Hu, J. C.; Athanasiou, K. A. A chondroitinase-ABC and TGF-β1 treatment regimen for enhancing the mechanical properties of tissue-engineered fibrocartilage. Acta Biomater. 2013, 9, 4626-4634.
doi: 10.1016/j.actbio.2012.09.037 URL |
| 385. |
Huey, D. J.; Athanasiou, K. A. Maturational growth of self-assembled, functional menisci as a result of TGF-β1 and enzymatic chondroitinase-ABC stimulation. Biomaterials. 2011, 32, 2052-2058.
doi: 10.1016/j.biomaterials.2010.11.041 URL |
| 386. |
Jiang, E. Y.; Sloan, S. R., Jr.; Wipplinger, C.; Kirnaz, S.; Härtl, R.; Bonassar, L. J. Proteoglycan removal by chondroitinase ABC improves injectable collagen gel adhesion to annulus fibrosus. Acta Biomater. 2019, 97, 428-436.
doi: 10.1016/j.actbio.2019.08.024 URL |
| 387. |
Mahmoud, M.; Kokozidou, M.; Auffarth, A.; Schulze-Tanzil, G. The relationship between diabetes mellitus type II and intervertebral disc degeneration in diabetic rodent models: a systematic and comprehensive review. Cells. 2020, 9, 2208.
doi: 10.3390/cells9102208 URL |
| 388. | Piccirilli, M.; Tarantino, R.; Anichini, G.; Delfini, R. Multiple disc herniations in a type II diabetic patient: case report and review of the literature. J Neurosurg Sci. 2008, 52, 83-85. |
| 389. |
Illien-Junger, S.; Grosjean, F.; Laudier, D. M.; Vlassara, H.; Striker, G. E.; Iatridis, J. C. Combined anti-inflammatory and anti-AGE drug treatments have a protective effect on intervertebral discs in mice with diabetes. PLoS One. 2013, 8, e64302.
doi: 10.1371/journal.pone.0064302 URL |
| 390. |
Hanssen, N. M. J.; Teraa, M.; Scheijen, J.; Van de Waarenburg, M.; Gremmels, H.; Stehouwer, C. D. A.; Verhaar, M. C.; Schalkwijk, C. G. Plasma methylglyoxal levels are associated with amputations and mortality in severe limb ischemia patients with and without diabetes. Diabetes Care. 2021, 44, 157-163.
doi: 10.2337/dc20-0581 URL |
| 391. | Cannata, F.; Vadalà, G.; Ambrosio, L.; Fallucca, S.; Napoli, N.; Papalia, R.; Pozzilli, P.; Denaro, V. Intervertebral disc degeneration: A focus on obesity and type 2 diabetes. Diabetes Metab Res Rev. 2020, 36, e3224. |
| 392. |
Song, Y.; Wang, Y.; Zhang, Y.; Geng, W.; Liu, W.; Gao, Y.; Li, S.; Wang, K.; Wu, X.; Kang, L.; Yang, C. Advanced glycation end products regulate anabolic and catabolic activities via NLRP3-inflammasome activation in human nucleus pulposus cells. J Cell Mol Med. 2017, 21, 1373-1387.
doi: 10.1111/jcmm.2017.21.issue-7 URL |
| 393. |
Illien-Jünger, S.; Lu, Y.; Qureshi, S. A.; Hecht, A. C.; Cai, W.; Vlassara, H.; Striker, G. E.; Iatridis, J. C. Chronic ingestion of advanced glycation end products induces degenerative spinal changes and hypertrophy in aging pre-diabetic mice. PLoS One. 2015, 10, e0116625.
doi: 10.1371/journal.pone.0116625 URL |
| 394. |
Wang, J.; Hu, J.; Chen, X.; Huang, C.; Lin, J.; Shao, Z.; Gu, M.; Wu, Y.; Tian, N.; Gao, W.; Zhou, Y.; Wang, X.; Zhang, X. BRD4 inhibition regulates MAPK, NF-κB signals, and autophagy to suppress MMP-13 expression in diabetic intervertebral disc degeneration. FASEB J. 2019, 33, 11555-11566.
doi: 10.1096/fsb2.v33.10 URL |
| 395. |
Hu, Y.; Shao, Z.; Cai, X.; Liu, Y.; Shen, M.; Yao, Y.; Yuan, T.; Wang, W.; Ding, F.; Xiong, L. Mitochondrial pathway is involved in advanced glycation end products-induced apoptosis of rabbit annulus fibrosus cells. Spine (Phila Pa 1976). 2019, 44, E585-E595.
doi: 10.1097/BRS.0000000000002930 URL |
| 396. |
Luo, R.; Song, Y.; Liao, Z.; Yin, H.; Zhan, S.; Wang, K.; Li, S.; Li, G.; Ma, L.; Lu, S.; Zhang, Y.; Yang, C. Impaired calcium homeostasis via advanced glycation end products promotes apoptosis through endoplasmic reticulum stress in human nucleus pulposus cells and exacerbates intervertebral disc degeneration in rats. FEBS J. 2019, 286, 4356-4373.
doi: 10.1111/febs.v286.21 URL |
| 397. |
Liao, Z.; Luo, R.; Li, G.; Song, Y.; Zhan, S.; Zhao, K.; Hua, W.; Zhang, Y.; Wu, X.; Yang, C. Exosomes from mesenchymal stem cells modulate endoplasmic reticulum stress to protect against nucleus pulposus cell death and ameliorate intervertebral disc degeneration in vivo. Theranostics. 2019, 9, 4084-4100.
doi: 10.7150/thno.33638 URL |
| 398. |
Lee, M.; Kim, B. J.; Lim, E. J.; Back, S. K.; Lee, J. H.; Yu, S. W.; Hong, S. H.; Kim, J. H.; Lee, S. H.; Jung, W. W.; Sul, D.; Na, H. S. Complete Freund’s adjuvant-induced intervertebral discitis as an animal model for discogenic low back pain. Anesth Analg. 2009, 109, 1287-1296.
doi: 10.1213/ane.0b013e3181b31f39 URL |
| 399. |
Luan, S.; Wan, Q.; Luo, H.; Li, X.; Ke, S.; Lin, C.; Wu, Y.; Wu, S.; Ma, C. Running exercise alleviates pain and promotes cell proliferation in a rat model of intervertebral disc degeneration. Int J Mol Sci. 2015, 16, 2130-2144.
doi: 10.3390/ijms16012130 URL |
| 400. |
Jung, W. W.; Kim, H. S.; Shon, J. R.; Lee, M.; Lee, S. H.; Sul, D.; Na, H. S.; Kim, J. H.; Kim, B. J. Intervertebral disc degeneration-induced expression of pain-related molecules: glial cell-derived neurotropic factor as a key factor. J Neurosurg Anesthesiol. 2011, 23, 329-334.
doi: 10.1097/ANA.0b013e318220f033 URL |
| 401. |
Oegema, T. R., Jr.; Johnson, S. L.; Aguiar, D. J.; Ogilvie, J. W. Fibronectin and its fragments increase with degeneration in the human intervertebral disc. Spine (Phila Pa 1976). 2000, 25, 2742-2747.
doi: 10.1097/00007632-200011010-00005 URL |
| 402. |
Tiaden, A. N.; Klawitter, M.; Lux, V.; Mirsaidi, A.; Bahrenberg, G.; Glanz, S.; Quero, L.; Liebscher, T.; Wuertz, K.; Ehrmann, M.; Richards, P. J. Detrimental role for human high temperature requirement serine protease A1 (HTRA1) in the pathogenesis of intervertebral disc (IVD) degeneration. J Biol Chem. 2012, 287, 21335-21345.
doi: 10.1074/jbc.M112.341032 URL |
| 403. |
Schmidli, M. R.; Sadowska, A.; Cvitas, I.; Gantenbein, B.; Lischer, H. E. L.; Forterre, S.; Hitzl, W.; Forterre, F.; Wuertz-Kozak, K. Fibronectin fragments and inflammation during canine intervertebral disc disease. Front Vet Sci. 2020, 7, 547644.
doi: 10.3389/fvets.2020.547644 URL |
| 404. |
Krock, E.; Rosenzweig, D. H.; Currie, J. B.; Bisson, D. G.; Ouellet, J. A.; Haglund, L. Toll-like Receptor Activation Induces Degeneration of Human Intervertebral Discs. Sci Rep. 2017, 7, 17184.
doi: 10.1038/s41598-017-17472-1 URL |
| 405. |
Liu, H. F.; Zhang, H.; Qiao, G. X.; Ning, B.; Hu, Y. L.; Wang, D. C.; Hu, Y. G. A novel rabbit disc degeneration model induced by fibronectin fragment. Joint Bone Spine. 2013, 80, 301-306.
doi: 10.1016/j.jbspin.2012.07.009 URL |
| 406. |
Greg, Anderson, D.; Li, X.; Tannoury, T.; Beck, G.; Balian, G. A fibronectin fragment stimulates intervertebral disc degeneration in vivo. Spine (Phila Pa 1976). 2003, 28, 2338-2345.
doi: 10.1097/01.BRS.0000096943.27853.BC URL |
| 407. |
Khan, A. N.; Jacobsen, H. E.; Khan, J.; Filippi, C. G.; Levine, M.; Lehman, R. A., Jr.; Riew, K. D.; Lenke, L. G.; Chahine, N. O Inflammatory biomarkers of low back pain and disc degeneration: a review. Ann N Y Acad Sci. 2017, 1410, 68-84.
doi: 10.1111/nyas.2017.1410.issue-1 URL |
| 408. | Cosamalón-Gan, I.; Cosamalón-Gan, T.; Mattos-Piaggio, G.; Villar-Suárez, V.; García-Cosamalón, J.; Vega-Álvarez, J. A. Inflammation in the intervertebral disc herniation. Neurocirugia (Astur: Engl Ed). 2021, 32, 21-35. |
| 409. |
Kim, H.; Hong, J. Y.; Lee, J.; Jeon, W. J.; Ha, I. H. IL-1β promotes disc degeneration and inflammation through direct injection of intervertebral disc in a rat lumbar disc herniation model. Spine J. 2021, 21, 1031-1041.
doi: 10.1016/j.spinee.2021.01.014 URL |
| 410. |
Glant, T. T.; Mikecz, K.; Arzoumanian, A.; Poole, A. R. Proteoglycan-induced arthritis in BALB/c mice. Clinical features and histopathology. Arthritis Rheum. 1987, 30, 201-212.
doi: 10.1002/(ISSN)1529-0131 URL |
| 411. |
Mikecz, K.; Glant, T. T.; Poole, A. R. Immunity to cartilage proteoglycans in BALB/c mice with progressive polyarthritis and ankylosing spondylitis induced by injection of human cartilage proteoglycan. Arthritis Rheum. 1987, 30, 306-318.
doi: 10.1002/(ISSN)1529-0131 URL |
| 412. |
Leroux, J. Y.; Guerassimov, A.; Cartman, A.; Delaunay, N.; Webber, C.; Rosenberg, L. C.; Banerjee, S.; Poole, A. R. Immunity to the G1 globular domain of the cartilage proteoglycan aggrecan can induce inflammatory erosive polyarthritis and spondylitis in BALB/c mice but immunity to G1 is inhibited by covalently bound keratan sulfate in vitro and in vivo. J Clin Invest. 1996, 97, 621-632.
doi: 10.1172/JCI118458 URL |
| 413. |
Shi, S.; Ciurli, C.; Cartman, A.; Pidoux, I.; Poole, A. R.; Zhang, Y. Experimental immunity to the G1 domain of the proteoglycan versican induces spondylitis and sacroiliitis, of a kind seen in human spondylarthropathies. Arthritis Rheum. 2003, 48, 2903-2915.
doi: 10.1002/art.11270 URL |
| 414. |
Tseng, H. W.; Glant, T. T.; Brown, M. A.; Kenna, T. J.; Thomas, G. P.; Pettit, A. R. Early anti-inflammatory intervention ameliorates axial disease in the proteoglycan-induced spondylitis mouse model of ankylosing spondylitis. BMC Musculoskelet Disord. 2017, 18, 228.
doi: 10.1186/s12891-017-1600-7 URL |
| 415. |
Zhou, C.; Cha, T.; Wang, W.; Guo, R.; Li, G. Investigation of alterations in the lumbar disc biomechanics at the adjacent segments after spinal fusion using a combined in vivo and in silico approach. Ann Biomed Eng. 2021, 49, 601-616.
doi: 10.1007/s10439-020-02588-9 URL |
| 416. | Oppenheimer, J. H.; DeCastro, I.; McDonnell, D. E. Minimally invasive spine technology and minimally invasive spine surgery: a historical review. Neurosurg Focus. 2009, 27, E9. |
| 417. |
Loenen, A. C. Y.; Noriega, D. C.; Ruiz Wills, C.; Noailly, J.; Nunley, P. D.; Kirchner, R.; Ito, K.; van Rietbergen, B. Misaligned spinal rods can induce high internal forces consistent with those observed to cause screw pullout and disc degeneration. Spine J. 2021, 21, 528-537.
doi: 10.1016/j.spinee.2020.09.010 URL |
| 418. |
Liu, Y.; Luo, X.; Zhou, J.; Li, N.; Peng, S.; Rong, P.; Wang, W. Prognosis of posterior osteophyte after anterior cervical decompression and fusion in patients with cervical spondylotic myelopathy using three-dimensional computed tomography study. Eur Spine J. 2016, 25, 1861-1868.
doi: 10.1007/s00586-016-4390-y URL |
| 419. |
Phillips, F. M.; Reuben, J.; Wetzel, F. T. Intervertebral disc degeneration adjacent to a lumbar fusion. An experimental rabbit model. J Bone Joint Surg Br. 2002, 84, 289-294.
doi: 10.1302/0301-620X.84B2.0840196 URL |
| 420. |
Foster, M. R.; Allen, M. J.; Schoonmaker, J. E.; Yuan, H. A.; Kanazawa, A.; Park, S. A.; Liu, B. Characterization of a developing lumbar arthrodesis in a sheep model with quantitative instability. Spine J. 2002, 2, 244-250.
doi: 10.1016/S1529-9430(02)00189-4 URL |
| 421. |
Holmes, C.; Elder, B. D.; Ishida, W.; Perdomo-Pantoja, A.; Locke, J.; Cottrill, E.; Lo, S. L.; Witham, T. F. Comparing the efficacy of syngeneic iliac and femoral allografts with iliac crest autograft in a rat model of lumbar spinal fusion. J Orthop Surg Res. 2020, 15, 410.
doi: 10.1186/s13018-020-01936-8 URL |
| 422. |
Kroeber, M.; Unglaub, F.; Guehring, T.; Nerlich, A.; Hadi, T.; Lotz, J.; Carstens, C. Effects of controlled dynamic disc distraction on degenerated intervertebral discs: an in vivo study on the rabbit lumbar spine model. Spine (Phila Pa 1976). 2005, 30, 181-187.
doi: 10.1097/01.brs.0000150487.17562.b1 URL |
| 423. |
Pearce, R. H.; Grimmer, B. J.; Adams, M. E. Degeneration and the chemical composition of the human lumbar intervertebral disc. J Orthop Res. 1987, 5, 198-205.
doi: 10.1002/(ISSN)1554-527X URL |
| 424. |
Huang, Y. C.; Xiao, J.; Leung, V. Y.; Lu, W. W.; Hu, Y.; Luk, K. D. K. Lumbar intervertebral disc allograft transplantation: the revascularisation pattern. Eur Spine J. 2018, 27, 728-736.
doi: 10.1007/s00586-017-5419-6 URL |
| 425. |
Flouzat-Lachaniette, C. H.; Jullien, N.; Bouthors, C.; Beohou, E.; Laurent, B.; Bierling, P.; Dubory, A.; Rouard, H. A novel in vivo porcine model of intervertebral disc degeneration induced by cryoinjury. Int Orthop. 2018, 42, 2263-2272.
doi: 10.1007/s00264-018-3971-2 URL |
| 426. |
Holm, S.; Baranto, A.; Kaigle Holm, A.; Ekström, L.; Swärd, L.; Hansson, T.; Hansson, H. A. Reactive changes in the adolescent porcine spine with disc degeneration due to endplate injury. Vet Comp Orthop Traumatol. 2007, 20, 12-17.
doi: 10.1055/s-0037-1616581 URL |
| 427. |
Kawchuk, G. N.; Kaigle, A. M.; Holm, S. H.; Rod Fauvel, O.; Ekström, L.; Hansson, T. The diagnostic performance of vertebral displacement measurements derived from ultrasonic indentation in an in vivo model of degenerative disc disease. Spine (Phila Pa 1976). 2001, 26, 1348-1355.
doi: 10.1097/00007632-200106150-00018 URL |
| 428. |
Yuan, W.; Che, W.; Jiang, Y. Q.; Yuan, F. L.; Wang, H. R.; Zheng, G. L.; Li, X. L.; Dong, J. Establishment of intervertebral disc degeneration model induced by ischemic sub-endplate in rat tail. Spine J. 2015, 15, 1050-1059.
doi: 10.1016/j.spinee.2015.01.026 URL |
| 429. |
Yin, S.; Du, H.; Zhao, W.; Ma, S.; Zhang, M.; Guan, M.; Liu, M. Inhibition of both endplate nutritional pathways results in intervertebral disc degeneration in a goat model. J Orthop Surg Res. 2019, 14, 138.
doi: 10.1186/s13018-019-1188-8 URL |
| 430. | Peng, B.; Wu, W.; Hou, S.; Li, P.; Zhang, C.; Yang, Y. The pathogenesis of discogenic low back pain. J Bone Joint Surg Br. 2005, 87, 62-67. |
| 431. |
Fagan, A. B.; Sarvestani, G.; Moore, R. J.; Fraser, R. D.; Vernon-Roberts, B.; Blumbergs, P. C. Innervation of anulus tears: an experimental animal study. Spine (Phila Pa 1976). 2010, 35, 1200-1205.
doi: 10.1097/BRS.0b013e3181c02812 URL |
| 432. |
Freemont, A. J.; Watkins, A.; Le Maitre, C.; Baird, P.; Jeziorska, M.; Knight, M. T.; Ross, E. R.; O’Brien, J. P.; Hoyland, J. A. Nerve growth factor expression and innervation of the painful intervertebral disc. J Pathol. 2002, 197, 286-292.
doi: 10.1002/path.1108 URL |
| 433. |
Stefanakis, M.;.; Al-Abbasi, M.; Harding, I.; Pollintine, P.; Dolan, P.; Tarlton, J.; Adams, M. A. Annulus fissures are mechanically and chemically conducive to the ingrowth of nerves and blood vessels. Spine (Phila Pa 1976). 2012, 37, 1883-1891.
doi: 10.1097/BRS.0b013e318263ba59 URL |
| 434. |
Johnson, W. E.; Sivan, S.; Wright, K. T.; Eisenstein, S. M.; Maroudas, A.; Roberts, S. Human intervertebral disc cells promote nerve growth over substrata of human intervertebral disc aggrecan. Spine (Phila Pa 1976). 2006, 31, 1187-1193.
doi: 10.1097/01.brs.0000217669.04903.61 URL |
| 435. |
Melrose, J.; Roberts, S.; Smith, S.; Menage, J.; Ghosh, P. Increased nerve and blood vessel ingrowth associated with proteoglycan depletion in an ovine anular lesion model of experimental disc degeneration. Spine (Phila Pa 1976). 2002, 27, 1278-1285.
doi: 10.1097/00007632-200206150-00007 URL |
| 436. |
Shi, C.; Das, V.; Li, X.; Kc, R.; Qiu, S.; I, O. S.; Ripper, R. L.; Kroin, J. S.; Mwale, F.; Wallace, A. A.; Zhu, B.; Zhao, L.; van Wijnen, A. J.; Ji, M.; Lu, J.; Votta-Velis, G.; Yuan, W.; Im, H. J. Development of an in vivo mouse model of discogenic low back pain. J Cell Physiol. 2018, 233, 6589-6602.
doi: 10.1002/jcp.v233.10 URL |
| 437. |
McMorran, J. G.; Gregory, D. E. The influence of axial compression on the cellular and mechanical function of spinal tissues; emphasis on the nucleus pulposus and annulus fibrosus: a review. J Biomech Eng. 2021, 143, 050802.
doi: 10.1115/1.4049749 URL |
| 438. |
Borem, R.; Madeline, A.; Vela, R., Jr.; Gill, S.; Mercuri, J. Multi-laminate annulus fibrosus repair scaffold with an interlamellar matrix enhances impact resistance, prevents herniation and assists in restoring spinal kinematics. J Mech Behav Biomed Mater. 2019, 95, 41-52.
doi: 10.1016/j.jmbbm.2019.03.030 URL |
| 439. |
Torre, O. M.;.; Evashwick-Rogler, T. W.; Nasser, P.; Iatridis, J. C. Biomechanical test protocols to detect minor injury effects in intervertebral discs. J Mech Behav Biomed Mater. 2019, 95, 13-20.
doi: 10.1016/j.jmbbm.2019.03.024 URL |
| 440. |
Li, J.; Yuan, X.; Li, F.; Wang, F.; Li, Y.; Wang, E.; Yang, X.; Xiang, Y.; Song, E. A novel full endoscopic annular repair technique combined with autologous conditioned plasma intradiscal injection: a new safe serial therapeutic model for the treatment of lumbar disc herniation. Ann Palliat Med. 2021, 10, 292-301.
doi: 10.21037/apm URL |
| 441. | Ren, C.; Qin, R.; Li, Y.; Wang, P. Microendoscopic discectomy combined with annular suture versus percutaneous transforaminal endoscopic discectomy for lumbar disc herniation: a prospective observational study. Pain Physician. 2020, 23, E713-E721. |
| 442. |
Miller, L. E.;.; McGirt, M. J.; Garfin, S. R.; Bono, C. M. Association of annular defect width after lumbar discectomy with risk of symptom recurrence and reoperation: systematic review and meta-analysis of comparative studies. Spine (Phila Pa 1976). 2018, 43, E308-E315.
doi: 10.1097/BRS.0000000000002501 URL |
| 443. |
Peredo, A. P.; Gullbrand, S. E.; Smith, H. E.; Mauck, R. L. Putting the pieces in place: mobilizing cellular players to improve annulus fibrosus repair. Tissue Eng Part B Rev. 2020. doi: 10.1089/ten.TEB.2020.0196.
doi: 10.1089/ten.TEB.2020.0196 URL |
| 444. |
Lequin, M. B.; Barth, M.; Thomė, C.; Bouma, G. J. Primary limited lumbar discectomy with an annulus closure device: one-year clinical and radiographic results from a prospective, multi-center study. Korean J Spine. 2012, 9, 340-347.
doi: 10.14245/kjs.2012.9.4.340 URL |
| 445. |
Strenge, K. B.;.; DiPaola, C. P.; Miller, L. E.; Hill, C. P.; Whitmore, R. G. Multicenter study of lumbar discectomy with Barricaid annular closure device for prevention of lumbar disc reherniation in US patients: A historically controlled post-market study protocol. Medicine (Baltimore). 2019, 98, e16953.
doi: 10.1097/MD.0000000000016953 URL |
| 446. |
Kienzler, J. C.; Fandino, J.; Van de Kelft, E.; Eustacchio, S.; Bouma, G. J. Risk factors for early reherniation after lumbar discectomy with or without annular closure: results of a multicenter randomized controlled study. Acta Neurochir (Wien). 2021, 163, 259-268.
doi: 10.1007/s00701-020-04505-4 URL |
| 447. |
Miller, L. E.; Allen, R. T.; Duhon, B.; Radcliff, K. E. Expert review with meta-analysis of randomized and nonrandomized controlled studies of Barricaid annular closure in patients at high risk for lumbar disc reherniation. Expert Rev Med Devices. 2020, 17, 461-469.
doi: 10.1080/17434440.2020.1745061 URL |
| 448. |
Thomé, C.; Klassen, P. D.; Bouma, G. J.; Kuršumović, A.; Fandino, J.; Barth, M.; Arts, M.; van den Brink, W.; Bostelmann, R.; Hegewald, A.; Heidecke, V.; Vajkoczy, P.; Fröhlich, S.; Wolfs, J.; Assaker, R.; Van de Kelft, E.; Köhler, H. P.; Jadik, S.; Eustacchio, S.; Hes, R.; Martens, F. Annular closure in lumbar microdiscectomy for prevention of reherniation: a randomized clinical trial. Spine J. 2018, 18, 2278-2287.
doi: 10.1016/j.spinee.2018.05.003 URL |
| 449. |
Choy, W. J.; Phan, K.; Diwan, A. D.; Ong, C. S.; Mobbs, R. J. Annular closure device for disc herniation: meta-analysis of clinical outcome and complications. BMC Musculoskelet Disord. 2018, 19, 290.
doi: 10.1186/s12891-018-2213-5 URL |
| 450. |
Bailey, A.; Araghi, A.; Blumenthal, S.; Huffmon, G. V. Prospective, multicenter, randomized, controlled study of anular repair in lumbar discectomy: two-year follow-up. Spine (Phila Pa 1976). 2013, 38, 1161-1169.
doi: 10.1097/BRS.0b013e31828b2e2f URL |
| 451. |
Bouma, G. J.; Ardeshiri, A.; Miller, L. E.; Van de Kelft, E.; Bostelmann, R.; Klassen, P. D.; Flüh, C.; Kuršumović, A. Clinical performance of a bone-anchored annular closure device in older adults. Clin Interv Aging. 2019, 14, 1085-1094.
doi: 10.2147/CIA URL |
| 452. | Likhitpanichkul, M.; Dreischarf, M.; Illien-Junger, S.; Walter, B. A.; Nukaga, T.; Long, R. G.; Sakai, D.; Hecht, A. C.; Iatridis, J. C. Fibrin-genipin adhesive hydrogel for annulus fibrosus repair: performance evaluation with large animal organ culture, in situ biomechanics, and in vivo degradation tests. Eur Cell Mater. 2014, 28, 25-37; discussion 37-38. |
| 453. |
DiStefano, T. J.; Shmukler, J. O.; Danias, G.; Di Pauli von Treuheim, T.; Hom, W. W.; Goldberg, D. A.; Laudier, D. M.; Nasser, P. R.; Hecht, A. C.; Nicoll, S. B.; Iatridis, J. C. Development of a two-part biomaterial adhesive strategy for annulus fibrosus repair and ex vivo evaluation of implant herniation risk. Biomaterials. 2020, 258, 120309.
doi: 10.1016/j.biomaterials.2020.120309 URL |
| 454. |
Ghobril, C.; Grinstaff, M. W. The chemistry and engineering of polymeric hydrogel adhesives for wound closure: a tutorial. Chem Soc Rev. 2015, 44, 1820-1835.
doi: 10.1039/C4CS00332B URL |
| 455. | Chapter 1 - Introduction and Adhesion Theories. In Adhesives Technology Handbook (Second Edition), Ebnesajjad, S., ed. William Andrew Publishing: Norwich, NY, 2009; pp 1-19. |
| 456. |
Li, X.; Wang, Y.; Xu, F.; Zhang, F.; Xu, Y.; Tang, L.; Webster, T. J. Artemisinin loaded mPEG-PCL nanoparticle based photosensitive gelatin methacrylate hydrogels for the treatment of gentamicin induced hearing loss. Int J Nanomedicine. 2020, 15, 4591-4606.
doi: 10.2147/IJN.S245188 URL |
| 457. |
Ügdüler, S.;.; De Somer, T.; Van Geem, K. M.; Roosen, M.; Kulawig, A.; Leineweber, R.; De Meester, S. Towards a better understanding of delamination of multilayer flexible packaging films by carboxylic acids. ChemSusChem. 2021. doi: 10.1002/cssc.202002877.
doi: 10.1002/cssc.202002877 URL |
| 458. |
Benz, K.; Stippich, C.; Fischer, L.; Möhl, K.; Weber, K.; Lang, J.; Steffen, F.; Beintner, B.; Gaissmaier, C.; Mollenhauer, J. A. Intervertebral disc cell- and hydrogel-supported and spontaneous intervertebral disc repair in nucleotomized sheep. Eur Spine J. 2012, 21, 1758-1768.
doi: 10.1007/s00586-012-2443-4 URL |
| 459. |
Tsujimoto, T.; Sudo, H.; Todoh, M.; Yamada, K.; Iwasaki, K.; Ohnishi, T.; Hirohama, N.; Nonoyama, T.; Ukeba, D.; Ura, K.; Ito, Y. M.; Iwasaki, N. An acellular bioresorbable ultra-purified alginate gel promotes intervertebral disc repair: A preclinical proof-of-concept study. EBioMedicine. 2018, 37, 521-534.
doi: 10.1016/j.ebiom.2018.10.055 URL |
| 460. | Doench, I.;.; Ahn Tran, T.; David, L.; Montembault, A.; Viguier, E.; Gorzelanny, C.; Sudre, G.; Cachon, T.; Louback-Mohamed, M.; Horbelt, N.; Peniche-Covas, C.; Osorio-Madrazo, A. Cellulose nanofiber-reinforced chitosan hydrogel composites for intervertebral disc tissue repair. Biomimetics (Basel). 2019, 4, 19. |
| 461. |
Long, R. G.; Zderic, I.; Gueorguiev, B.; Ferguson, S. J.; Alini, M.; Grad, S.; Iatridis, J. C. Effects of level, loading rate, injury and repair on biomechanical response of ovine cervical intervertebral discs. Ann Biomed Eng. 2018, 46, 1911-1920.
doi: 10.1007/s10439-018-2077-8 URL |
| 462. |
Fujii, K.; Lai, A.; Korda, N.; Hom, W. W.; Evashwick-Rogler, T. W.; Nasser, P.; Hecht, A. C.; Iatridis, J. C. Ex-vivo biomechanics of repaired rat intervertebral discs using genipin crosslinked fibrin adhesive hydrogel. J Biomech. 2020, 113, 110100.
doi: 10.1016/j.jbiomech.2020.110100 URL |
| 463. |
Peng, Y.; Huang, D.; Li, J.; Liu, S.; Qing, X.; Shao, Z. Genipin-crosslinked decellularized annulus fibrosus hydrogels induces tissue-specific differentiation of bone mesenchymal stem cells and intervertebral disc regeneration. J Tissue Eng Regen Med. 2020, 14, 497-509.
doi: 10.1002/term.v14.3 URL |
| 464. | Cruz, M. A.;.; McAnany, S.; Gupta, N.; Long, R. G.; Nasser, P.; Eglin, D.; Hecht, A. C.; Illien-Junger, S.; Iatridis, J. C. Structural and chemical modification to improve adhesive and material properties of fibrin-genipin for repair of annulus fibrosus defects in intervertebral disks. J Biomech Eng. 2017, 139, 0845011-0845017. |
| 465. |
Sharabi, M.; Wertheimer, S.; Wade, K. R.; Galbusera, F.; Benayahu, D.; Wilke, H. J.; Haj-Ali, R. Towards intervertebral disc engineering: Bio-mimetics of form and function of the annulus fibrosus lamellae. J Mech Behav Biomed Mater. 2019, 94, 298-307.
doi: 10.1016/j.jmbbm.2019.03.023 URL |
| 466. |
Shamsah, A. H.; Cartmell, S. H.; Richardson, S. M.; Bosworth, L. A. Tissue engineering the annulus fibrosus using 3D rings of electrospun PCL:PLLA angle-ply nanofiber sheets. Front Bioeng Biotechnol. 2019, 7, 437.
doi: 10.3389/fbioe.2019.00437 URL |
| 467. |
Gluais, M.; Clouet, J.; Fusellier, M.; Decante, C.; Moraru, C.; Dutilleul, M.; Veziers, J.; Lesoeur, J.; Dumas, D.; Abadie, J.; Hamel, A.; Bord, E.; Chew, S. Y.; Guicheux, J.; Le Visage, C. In vitro and in vivo evaluation of an electrospun-aligned microfibrous implant for annulus fibrosus repair. Biomaterials. 2019, 205, 81-93.
doi: 10.1016/j.biomaterials.2019.03.010 URL |
| 468. |
Tavakoli, J.; Elliott, D. M.; Costi, J. J. The ultra-structural organization of the elastic network in the intra- and inter-lamellar matrix of the intervertebral disc. Acta Biomater. 2017, 58, 269-277.
doi: 10.1016/j.actbio.2017.05.036 URL |
| 469. |
Park, S. H.; Gil, E. S.; Mandal, B. B.; Cho, H.; Kluge, J. A.; Min, B. H.; Kaplan, D. L. Annulus fibrosus tissue engineering using lamellar silk scaffolds. J Tissue Eng Regen Med. 2012, 6 Suppl 3, s24-33.
doi: 10.1002/term.v6.S3 URL |
| 470. |
Bhattacharjee, M.; Miot, S.; Gorecka, A.; Singha, K.; Loparic, M.; Dickinson, S.; Das, A.; Bhavesh, N. S.; Ray, A. R.; Martin, I.; Ghosh, S. Oriented lamellar silk fibrous scaffolds to drive cartilage matrix orientation: towards annulus fibrosus tissue engineering. Acta Biomater. 2012, 8, 3313-3325.
doi: 10.1016/j.actbio.2012.05.023 URL |
| 471. |
Bhunia, B. K.; Kaplan, D. L.; Mandal, B. B. Silk-based multilayered angle-ply annulus fibrosus construct to recapitulate form and function of the intervertebral disc. Proc Natl Acad Sci U S A. 2018, 115, 477-482.
doi: 10.1073/pnas.1715912115 URL |
| 472. |
Bowles, R. D.; Gebhard, H. H.; Härtl, R.; Bonassar, L. J. Tissue-engineered intervertebral discs produce new matrix, maintain disc height, and restore biomechanical function to the rodent spine. Proc Natl Acad Sci U S A. 2011, 108, 13106-13111.
doi: 10.1073/pnas.1107094108 URL |
| 473. | De Pieri, A.; Byerley, A. M.; Musumeci, C. R.; Salemizadehparizi, F.; Vanderhorst, M. A.; Wuertz-Kozak, K. Electrospinning and 3D bioprinting for intervertebral disc tissue engineering. JOR Spine. 2020, 3, e1117. |
| 474. |
Beckett, L. E.; Lewis, J. T.; Tonge, T. K.; Korley, L. T. J. Enhancement of the mechanical properties of hydrogels with continuous fibrous reinforcement. ACS Biomater Sci Eng. 2020, 6, 5453-5473.
doi: 10.1021/acsbiomaterials.0c00911 URL |
| 475. | Alexeev, D.; Cui, S.; Grad, S.; Li, Z.; Ferguson, S. J. Mechanical and biological characterization of a composite annulus fibrosus repair strategy in an endplate delamination model. JOR Spine. 2020, 3, e1107. |
| 476. |
Xiao, L.; Wu, M.; Yan, F.; Xie, Y.; Liu, Z.; Huang, H.; Yang, Z.; Yao, S.; Cai, L. A radial 3D polycaprolactone nanofiber scaffold modified by biomineralization and silk fibroin coating promote bone regeneration in vivo. Int J Biol Macromol. 2021, 172, 19-29.
doi: 10.1016/j.ijbiomac.2021.01.036 URL |
| 477. |
Liu, W.; Thomopoulos, S.; Xia, Y. Electrospun nanofibers for regenerative medicine. Adv Healthc Mater. 2012, 1, 10-25.
doi: 10.1002/adhm.201100021 URL |
| 478. |
Parham, S.; Kharazi, A. Z.; Bakhsheshi-Rad, H. R.; Ghayour, H.; Ismail, A. F.; Nur, H.; Berto, F. Electrospun nano-fibers for biomedical and tissue engineering applications: a comprehensive review. Materials (Basel). 2020, 13, 2153.
doi: 10.3390/ma13092153 URL |
| 479. |
Zhou, P.; Chu, G.; Yuan, Z.; Wang, H.; Zhang, W.; Mao, Y.; Zhu, X.; Chen, W.; Yang, H.; Li, B. Regulation of differentiation of annulus fibrosus-derived stem cells using heterogeneous electrospun fibrous scaffolds. J Orthop Translat. 2021, 26, 171-180.
doi: 10.1016/j.jot.2020.02.003 URL |
| 480. |
Ma, J.; He, Y.; Liu, X.; Chen, W.; Wang, A.; Lin, C. Y.; Mo, X.; Ye, X. A novel electrospun-aligned nanoyarn/three-dimensional porous nanofibrous hybrid scaffold for annulus fibrosus tissue engineering. Int J Nanomedicine. 2018, 13, 1553-1567.
doi: 10.2147/IJN URL |
| 481. |
Xu, H.; Xu, B.; Yang, Q.; Li, X.; Ma, X.; Xia, Q.; Zhang, Y.; Zhang, C.; Wu, Y.; Zhang, Y. Comparison of decellularization protocols for preparing a decellularized porcine annulus fibrosus scaffold. PLoS One. 2014, 9, e86723.
doi: 10.1371/journal.pone.0086723 URL |
| 482. |
Saldin, L. T.; Cramer, M. C.; Velankar, S. S.; White, L. J.; Badylak, S. F. Extracellular matrix hydrogels from decellularized tissues: Structure and function. Acta Biomater. 2017, 49, 1-15.
doi: 10.1016/j.actbio.2016.11.068 URL |
| 483. |
D’Este, M.; Eglin, D.; Alini, M. Lessons to be learned and future directions for intervertebral disc biomaterials. Acta Biomater. 2018, 78, 13-22.
doi: 10.1016/j.actbio.2018.08.004 URL |
| 484. |
McGuire, R.; Borem, R.; Mercuri, J. The fabrication and characterization of a multi-laminate, angle-ply collagen patch for annulus fibrosus repair. J Tissue Eng Regen Med. 2017, 11, 3488-3493.
doi: 10.1002/term.2250 URL |
| 485. |
Liu, C.; Xiao, L.; Zhang, Y.; Zhao, Q.; Xu, H. Regeneration of annulus fibrosus tissue using a DAFM/PECUU-blended electrospun scaffold. J Biomater Sci Polym Ed. 2020, 31, 2347-2361.
doi: 10.1080/09205063.2020.1812038 URL |
| 486. |
Chen, H.; Han, Q.; Wang, C.; Liu, Y.; Chen, B.; Wang, J. Porous scaffold design for additive manufacturing in orthopedics: a review. Front Bioeng Biotechnol. 2020, 8, 609.
doi: 10.3389/fbioe.2020.00609 URL |
| 487. |
Lutzweiler, G.; Ndreu Halili, A.; Engin Vrana, N. The overview of porous, bioactive scaffolds as instructive biomaterials for tissue regeneration and their clinical translation. Pharmaceutics. 2020, 12, 602.
doi: 10.3390/pharmaceutics12070602 URL |
| 488. |
Chen, X.; Fan, H.; Deng, X.; Wu, L.; Yi, T.; Gu, L.; Zhou, C.; Fan, Y.; Zhang, X. Scaffold structural microenvironmental cues to guide tissue regeneration in bone tissue applications. Nanomaterials (Basel). 2018, 8, 960.
doi: 10.3390/nano8110960 URL |
| 489. |
Griffon, D. J.; Sedighi, M. R.; Schaeffer, D. V.; Eurell, J. A.; Johnson, A. L. Chitosan scaffolds: interconnective pore size and cartilage engineering. Acta Biomater. 2006, 2, 313-320.
doi: 10.1016/j.actbio.2005.12.007 URL |
| 490. | Nava, M. M.; Draghi, L.; Giordano, C.; Pietrabissa, R. The effect of scaffold pore size in cartilage tissue engineering. J Appl Biomater Funct Mater. 2016, 14, e223-229. |
| 491. | Xu, H.; Xu, B.; Yang, Q.; Li, X.; Ma, X.; Xia, Q.; Zhang, C.; Wu, Y. Fabrication and analysis of a novel tissue engineered composite biphasic scaffold for annulus fibrosus and nucleus pulposus. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2013, 27, 475-480. |
| 492. |
Xu, B.; Xu, H.; Wu, Y.; Li, X.; Zhang, Y.; Ma, X.; Yang, Q. Intervertebral disc tissue engineering with natural extracellular matrix-derived biphasic composite scaffolds. PLoS One. 2015, 10, e0124774.
doi: 10.1371/journal.pone.0124774 URL |
| 493. |
Oh, S. H.; Kim, T. H.; Im, G. I.; Lee, J. H. Investigation of pore size effect on chondrogenic differentiation of adipose stem cells using a pore size gradient scaffold. Biomacromolecules. 2010, 11, 1948-1955.
doi: 10.1021/bm100199m URL |
| 494. |
Nehrer, S.; Breinan, H. A.; Ramappa, A.; Young, G.; Shortkroff, S.; Louie, L. K.; Sledge, C. B.; Yannas, I. V.; Spector, M. Matrix collagen type and pore size influence behaviour of seeded canine chondrocytes. Biomaterials. 1997, 18, 769-776.
doi: 10.1016/S0142-9612(97)00001-X URL |
| 495. |
Im, G. I.; Ko, J. Y.; Lee, J. H. Chondrogenesis of adipose stem cells in a porous polymer scaffold: influence of the pore size. Cell Transplant. 2012, 21, 2397-2405.
doi: 10.3727/096368912X638865 URL |
| 496. |
Kim, H. Y.; Kim, H. N.; Lee, S. J.; Song, J. E.; Kwon, S. Y.; Chung, J. W.; Lee, D.; Khang, G. Effect of pore sizes of PLGA scaffolds on mechanical properties and cell behaviour for nucleus pulposus regeneration in vivo. J Tissue Eng Regen Med. 2017, 11, 44-57.
doi: 10.1002/term.v11.1 URL |
| 497. |
Chou, P. H.; Chee, A.; Shi, P.; Lin, C. L.; Zhao, Y.; Zhang, L.; An, H. S. Small molecule antagonist of C-C chemokine receptor 1 (CCR1) reduces disc inflammation in the rabbit model. Spine J. 2020, 20, 2025-2036.
doi: 10.1016/j.spinee.2020.07.001 URL |
| 498. | Zhang, W.; Nie, L.; Guo, Y. J.; Han, L. X.; Wang, X.; Zhao, H.; Han, Y. G.; Zhang, Y. Q.; Cheng, L. Th17 cell frequency and IL-17 concentration correlate with pre- and postoperative pain sensation in patients with intervertebral disk degeneration. Orthopedics. 2014, 37, e685-691. |
| 499. |
Kawakubo, A.; Uchida, K.; Miyagi, M.; Nakawaki, M.; Satoh, M.; Sekiguchi, H.; Yokozeki, Y.; Inoue, G.; Takaso, M. Investigation of resident and recruited macrophages following disc injury in mice. J Orthop Res. 2020, 38, 1703-1709.
doi: 10.1002/jor.v38.8 URL |
| 500. |
Sussman, E. M.; Halpin, M. C.; Muster, J.; Moon, R. T.; Ratner, B. D. Porous implants modulate healing and induce shifts in local macrophage polarization in the foreign body reaction. Ann Biomed Eng. 2014, 42, 1508-1516.
doi: 10.1007/s10439-013-0933-0 URL |
| 501. |
Zhu, Y.; Hideyoshi, S.; Jiang, H.; Matsumura, Y.; Dziki, J. L.; LoPresti, S. T.; Huleihel, L.; Faria, G. N. F.; Fuhrman, L. C.; Lodono, R.; Badylak, S. F.; Wagner, W. R. Injectable, porous, biohybrid hydrogels incorporating decellularized tissue components for soft tissue applications. Acta Biomater. 2018, 73, 112-126.
doi: 10.1016/j.actbio.2018.04.003 URL |
| 502. |
Veiseh, O.; Doloff, J. C.; Ma, M.; Vegas, A. J.; Tam, H. H.; Bader, A. R.; Li, J.; Langan, E.; Wyckoff, J.; Loo, W. S.; Jhunjhunwala, S.; Chiu, A.; Siebert, S.; Tang, K.; Hollister-Lock, J.; Aresta-Dasilva, S.; Bochenek, M.; Mendoza-Elias, J.; Wang, Y.; Qi, M.; Lavin, D. M.; Chen, M.; Dholakia, N.; Thakrar, R.; Lacík, I.; Weir, G. C.; Oberholzer, J.; Greiner, D. L.; Langer, R.; Anderson, D. G. . Size- and shape-dependent foreign body immune response to materials implanted in rodents and non-human primates. Nat Mater. 2015, 14, 643-651.
doi: 10.1038/nmat4290 URL |
| 503. |
Jia, Y.; Yang, W.; Zhang, K.; Qiu, S.; Xu, J.; Wang, C.; Chai, Y. Nanofiber arrangement regulates peripheral nerve regeneration through differential modulation of macrophage phenotypes. Acta Biomater. 2019, 83, 291-301.
doi: 10.1016/j.actbio.2018.10.040 URL |
| 504. |
McWhorter, F. Y.; Wang, T.; Nguyen, P.; Chung, T.; Liu, W. F. Modulation of macrophage phenotype by cell shape. Proc Natl Acad Sci U S A. 2013, 110, 17253-17258.
doi: 10.1073/pnas.1308887110 URL |
| 505. |
Tylek, T.; Blum, C.; Hrynevich, A.; Schlegelmilch, K.; Schilling, T.; Dalton, P. D.; Groll, J. Precisely defined fiber scaffolds with 40 μm porosity induce elongation driven M2-like polarization of human macrophages. Biofabrication. 2020, 12, 025007.
doi: 10.1088/1758-5090/ab5f4e URL |
| 506. | Roberts, S.; Evans, H.; Trivedi, J.; Menage, J. Histology and pathology of the human intervertebral disc. J Bone Joint Surg Am. 2006, 88 Suppl 2, 10-14. |
| 507. |
Shen, J.; Xu, S.; Zhou, H.; Liu, H.; Jiang, W.; Hao, J.; Hu, Z. IL-1β induces apoptosis and autophagy via mitochondria pathway in human degenerative nucleus pulposus cells. Sci Rep. 2017, 7, 41067.
doi: 10.1038/srep41067 URL |
| 508. | Sivan, S. S.; Wachtel, E.; Roughley, P. Structure, function, aging and turnover of aggrecan in the intervertebral disc. Biochim Biophys Acta. 2014, 1840, 3181-3189. |
| 509. |
Tengblad, A.; Pearce, R. H.; Grimmer, B. J. Demonstration of link protein in proteoglycan aggregates from human intervertebral disc. Biochem J. 1984, 222, 85-92.
doi: 10.1042/bj2220085 URL |
| 510. | Roughley, P.; Martens, D.; Rantakokko, J.; Alini, M.; Mwale, F.; Antoniou, J. The involvement of aggrecan polymorphism in degeneration of human intervertebral disc and articular cartilage. Eur Cell Mater. 2006, 11, 1-7; discussion 7. |
| 511. |
Ray, C. D. The PDN prosthetic disc-nucleus device. Eur Spine J. 2002, 11 Suppl 2, S137-142.
doi: 10.1007/s00586-002-0425-7 URL |
| 512. | Ceylan, A.; Aşik, I.; Özgencil, G. E.; Erken, B. Clinical results of intradiscal hydrogel administration (GelStix) in lumbar degenerative disc disease. Turk J Med Sci. 2019, 49, 1634-1639. |
| 513. | Durdag, E.; Ayden, O.; Albayrak, S.; Atci, I. B.; Armagan, E. Fragmentation to epidural space: first documented complication of Gelstix(TM.). Turk Neurosurg. 2014, 24, 602-605. |
| 514. |
Coric, D.; Mummaneni, P. V. Nucleus replacement technologies. J Neurosurg Spine. 2008, 8, 115-120.
doi: 10.3171/SPI/2008/8/2/115 URL |
| 515. |
Berlemann, U.; Schwarzenbach, O. An injectable nucleus replacement as an adjunct to microdiscectomy: 2 year follow-up in a pilot clinical study. Eur Spine J. 2009, 18, 1706-1712.
doi: 10.1007/s00586-009-1136-0 URL |
| 516. |
Eysel, P.; Rompe, J.; Schoenmayr, R.; Zoellner, J. Biomechanical behaviour of a prosthetic lumbar nucleus. Acta Neurochir (Wien). 1999, 141, 1083-1087.
doi: 10.1007/s007010050486 URL |
| 517. | Scholz, B.; Kinzelmann, C.; Benz, K.; Mollenhauer, J.; Wurst, H.; Schlosshauer, B. Suppression of adverse angiogenesis in an albumin-based hydrogel for articular cartilage and intervertebral disc regeneration. Eur Cell Mater. 2010, 20, 24-36; discussion 36-37. |
| 518. |
Tschugg, A.; Michnacs, F.; Strowitzki, M.; Meisel, H. J.; Thomé, C. A prospective multicenter phase I/II clinical trial to evaluate safety and efficacy of NOVOCART disc plus autologous disc chondrocyte transplantation in the treatment of nucleotomized and degenerative lumbar disc to avoid secondary disease: study protocol for a randomized controlled trial. Trials. 2016, 17, 108.
doi: 10.1186/s13063-016-1239-y URL |
| 519. |
Choi, U. Y.; Joshi, H. P.; Payne, S.; Kim, K. T.; Kyung, J. W.; Choi, H.; Cooke, M. J.; Kwon, S. Y.; Roh, E. J.; Sohn, S.; Shoichet, M. S.; Han, I. An injectable hyaluronan-methylcellulose (HAMC) hydrogel combined with Wharton’s jelly-derived mesenchymal stromal cells (WJ-MSCs) promotes degenerative disc repair. Int J Mol Sci. 2020, 21, 7391.
doi: 10.3390/ijms21197391 URL |
| 520. |
Häckel, S.; Zolfaghar, M.; Du, J.; Hoppe, S.; Benneker, L. M.; Garstka, N.; Peroglio, M.; Alini, M.; Grad, S.; Yayon, A.; Li, Z. Fibrin-hyaluronic acid hydrogel (RegenoGel) with fibroblast growth factor-18 for in vitro 3D culture of human and bovine nucleus pulposus cells. Int J Mol Sci. 2019, 20, 5036.
doi: 10.3390/ijms20205036 URL |
| 521. |
Ukeba, D.; Sudo, H.; Tsujimoto, T.; Ura, K.; Yamada, K.; Iwasaki, N. Bone marrow mesenchymal stem cells combined with ultra-purified alginate gel as a regenerative therapeutic strategy after discectomy for degenerated intervertebral discs. EBioMedicine. 2020, 53, 102698.
doi: 10.1016/j.ebiom.2020.102698 URL |
| 522. |
Alinejad, Y.; Adoungotchodo, A.; Grant, M. P.; Epure, L. M.; Antoniou, J.; Mwale, F.; Lerouge, S. Injectable chitosan hydrogels with enhanced mechanical properties for nucleus pulposus regeneration. Tissue Eng Part A. 2019, 25, 303-313.
doi: 10.1089/ten.tea.2018.0170 URL |
| 523. | Gan, Y.; Li, S.; Li, P.; Xu, Y.; Wang, L.; Zhao, C.; Ouyang, B.; Tu, B.; Zhang, C.; Luo, L.; Luo, X.; Mo, X.; Zhou, Q. A controlled release codelivery system of MSCs encapsulated in dextran/gelatin hydrogel with tgf-β3-loaded nanoparticles for nucleus pulposus regeneration. Stem Cells Int. 2016, 2016, 9042019. |
| 524. |
Nguyen, N. T.; Milani, A. H.; Jennings, J.; Adlam, D. J.; Freemont, A. J.; Hoyland, J. A.; Saunders, B. R. Highly compressive and stretchable poly(ethylene glycol) based hydrogels synthesised using pH-responsive nanogels without free-radical chemistry. Nanoscale. 2019, 11, 7921-7930.
doi: 10.1039/C9NR01535C URL |
| 525. |
Leone, G.; Consumi, M.; Lamponi, S.; Bonechi, C.; Tamasi, G.; Donati, A.; Rossi, C.; Magnani, A. Thixotropic PVA hydrogel enclosing a hydrophilic PVP core as nucleus pulposus substitute. Mater Sci Eng C Mater Biol Appl. 2019, 98, 696-704.
doi: 10.1016/j.msec.2019.01.039 URL |
| 526. |
Schmocker, A.; Khoushabi, A.; Frauchiger, D. A.; Gantenbein, B.; Schizas, C.; Moser, C.; Bourban, P. E.; Pioletti, D. P. A photopolymerized composite hydrogel and surgical implanting tool for a nucleus pulposus replacement. Biomaterials. 2016, 88, 110-119.
doi: 10.1016/j.biomaterials.2016.02.015 URL |
| 527. |
Peng, Y.; Huang, D.; Liu, S.; Li, J.; Qing, X.; Shao, Z. Biomaterials-induced stem cells specific differentiation into intervertebral disc lineage cells. Front Bioeng Biotechnol. 2020, 8, 56.
doi: 10.3389/fbioe.2020.00056 URL |
| 528. |
Chen, P.; Ning, L.; Qiu, P.; Mo, J.; Mei, S.; Xia, C.; Zhang, J.; Lin, X.; Fan, S. Photo-crosslinked gelatin-hyaluronic acid methacrylate hydrogel-committed nucleus pulposus-like differentiation of adipose stromal cells for intervertebral disc repair. J Tissue Eng Regen Med. 2019, 13, 682-693.
doi: 10.1002/term.v13.4 URL |
| 529. |
Binch, A. L. A.; Ratcliffe, L. P. D.; Milani, A. H.; Saunders, B. R.; Armes, S. P.; Hoyland, J. A. Site-directed differentiation of human adipose-derived mesenchymal stem cells to nucleus pulposus cells using an injectable hydroxyl-functional diblock copolymer worm gel. Biomacromolecules. 2021, 22, 837-845.
doi: 10.1021/acs.biomac.0c01556 URL |
| 530. | Wang, Z.; Liu, H.; Luo, W.; Cai, T.; Li, Z.; Liu, Y.; Gao, W.; Wan, Q.; Wang, X.; Wang, J.; Wang, Y.; Yang, X. Regeneration of skeletal system with genipin crosslinked biomaterials. J Tissue Eng. 2020, 11, 2041731420974861. |
| 531. | Panebianco, C. J.; DiStefano, T. J.; Mui, B.; Hom, W. W.; Iatridis, J. C. Crosslinker concentration controls TGFβ-3 release and annulus fibrosus cell apoptosis in genipin-crosslinked fibrin hydrogels. Eur Cell Mater. 2020, 39, 211-226. |
| 532. |
Zhou, X.; Wang, J.; Fang, W.; Tao, Y.; Zhao, T.; Xia, K.; Liang, C.; Hua, J.; Li, F.; Chen, Q. Genipin cross-linked type II collagen/chondroitin sulfate composite hydrogel-like cell delivery system induces differentiation of adipose-derived stem cells and regenerates degenerated nucleus pulposus. Acta Biomater. 2018, 71, 496-509.
doi: 10.1016/j.actbio.2018.03.019 URL |
| 533. |
Wu, X.; Liu, Y.; Guo, X.; Zhou, W.; Wang, L.; Shi, J.; Tao, Y.; Zhu, M.; Geng, D.; Yang, H.; Mao, H. Prolactin inhibits the progression of intervertebral disc degeneration through inactivation of the NF-κB pathway in rats. Cell Death Dis. 2018, 9, 98.
doi: 10.1038/s41419-017-0151-z URL |
| 534. | Issy, A. C.; Castania, V.; Castania, M.; Salmon, C. E.; Nogueira-Barbosa, M. H.; Bel, E. D.; Defino, H. L. Experimental model of intervertebral disc degeneration by needle puncture in Wistar rats. Braz JMed Biol Res. 2013, 46, 235-244. |
| 535. |
Alfredo Uquillas, J.; Kishore, V.; Akkus, O. Genipin crosslinking elevates the strength of electrochemically aligned collagen to the level of tendons. J Mech Behav Biomed Mater. 2012, 15, 176-189.
doi: 10.1016/j.jmbbm.2012.06.012 URL |
| 536. |
Censi, R.; Di Martino, P.; Vermonden, T.; Hennink, W. E. Hydrogels for protein delivery in tissue engineering. J Control Release. 2012, 161, 680-692.
doi: 10.1016/j.jconrel.2012.03.002 URL |
| 537. |
Francisco, A. T.; Mancino, R. J.; Bowles, R. D.; Brunger, J. M.; Tainter, D. M.; Chen, Y. T.; Richardson, W. J.; Guilak, F.; Setton, L. A. Injectable laminin-functionalized hydrogel for nucleus pulposus regeneration. Biomaterials. 2013, 34, 7381-7388.
doi: 10.1016/j.biomaterials.2013.06.038 URL |
| 538. |
Zhu, Y.; Tan, J.; Zhu, H.; Lin, G.; Yin, F.; Wang, L.; Song, K.; Wang, Y.; Zhou, G.; Yi, W. Development of kartogenin-conjugated chitosan-hyaluronic acid hydrogel for nucleus pulposus regeneration. Biomater Sci. 2017, 5, 784-791.
doi: 10.1039/C7BM00001D URL |
| 539. |
Sulaiman, S. B.; Idrus, R. B. H.; Hwei, N. M. Gelatin microsphere for cartilage tissue engineering: current and future strategies. Polymers (Basel). 2020, 12, 2404.
doi: 10.3390/polym12102404 URL |
| 540. |
Leong, W.; Wang, D. A. Cell-laden polymeric microspheres for biomedical applications. Trends Biotechnol. 2015, 33, 653-666.
doi: 10.1016/j.tibtech.2015.09.003 URL |
| 541. |
Patel, K. S.; Patel, M. B. Preparation and evaluation of chitosan microspheres containing nicorandil. Int J Pharm Investig. 2014, 4, 32-37.
doi: 10.4103/2230-973X.127738 URL |
| 542. |
Wang, X.; Wenk, E.; Matsumoto, A.; Meinel, L.; Li, C.; Kaplan, D. L. Silk microspheres for encapsulation and controlled release. J Control Release. 2007, 117, 360-370.
doi: 10.1016/j.jconrel.2006.11.021 URL |
| 543. |
Saravanan, M.; Bhaskar, K.; Maharajan, G.; Pillai, K. S. Development of gelatin microspheres loaded with diclofenac sodium for intra-articular administration. J Drug Target. 2011, 19, 96-103.
doi: 10.3109/10611861003733979 URL |
| 544. |
Dhamecha, D.; Movsas, R.; Sano, U.; Menon, J. U. Applications of alginate microspheres in therapeutics delivery and cell culture: Past, present and future. Int J Pharm. 2019, 569, 118627.
doi: 10.1016/j.ijpharm.2019.118627 URL |
| 545. |
Xu, H.; Sun, M.; Wang, C.; Xia, K.; Xiao, S.; Wang, Y.; Ying, L.; Yu, C.; Yang, Q.; He, Y.; Liu, A.; Chen, L. Growth differentiation factor-5-gelatin methacryloyl injectable microspheres laden with adipose-derived stem cells for repair of disc degeneration. Biofabrication. 2020, 13, 015010.
doi: 10.1088/1758-5090/abc4d3 URL |
| 546. |
Xu, H.; Sun, M.; Wang, C.; Xia, K.; Xiao, S.; Wang, Y.; Ying, L.; Yu, C.; Yang, Q.; He, Y.; Liu, A.; Chen, L. GDF5-GelMA injectable microspheres laden with adipose-derived stem cells for disc degeneration repair. Biofabrication. 2020. doi: 10.1088/1758-5090/abc4d3.
doi: 10.1088/1758-5090/abc4d3 URL |
| 547. |
Feng, G.; Zhang, Z.; Dang, M.; Rambhia, K. J.; Ma, P. X. Nanofibrous spongy microspheres to deliver rabbit mesenchymal stem cells and anti-miR-199a to regenerate nucleus pulposus and prevent calcification. Biomaterials. 2020, 256, 120213.
doi: 10.1016/j.biomaterials.2020.120213 URL |
| 548. | Xia, K.; Zhu, J.; Hua, J.; Gong, Z.; Yu, C.; Zhou, X.; Wang, J.; Huang, X.; Yu, W.; Li, L.; Gao, J.; Chen, Q.; Li, F.; Liang, C. Intradiscal injection of induced pluripotent stem cell-derived nucleus pulposus-like cell-seeded polymeric microspheres promotes rat disc regeneration. Stem Cells Int. 2019, 2019, 6806540. |
| 549. |
Shan, Z.; Lin, X.; Wang, S.; Zhang, X.; Pang, Y.; Li, S.; Yu, T.; Fan, S.; Zhao, F. An injectable nucleus pulposus cell-modified decellularized scaffold: biocompatible material for prevention of disc degeneration. Oncotarget. 2017, 8, 40276-40288.
doi: 10.18632/oncotarget.v8i25 URL |
| 550. |
Liang, C. Z.; Li, H.; Tao, Y. Q.; Peng, L. H.; Gao, J. Q.; Wu, J. J.; Li, F. C.; Hua, J. M.; Chen, Q. X. Dual release of dexamethasone and TGF-β3 from polymeric microspheres for stem cell matrix accumulation in a rat disc degeneration model. Acta Biomater. 2013, 9, 9423-9433.
doi: 10.1016/j.actbio.2013.08.019 URL |
| 551. |
Wu, D.; Yu, Y.; Zhao, C.; Shou, X.; Piao, Y.; Zhao, X.; Zhao, Y.; Wang, S. NK-cell-encapsulated porous microspheres via microfluidic electrospray for tumor immunotherapy. ACS Appl Mater Interfaces. 2019, 11, 33716-33724.
doi: 10.1021/acsami.9b12816 URL |
| 552. | Jo, Y. K.; Lee, D. Biopolymer microparticles prepared by microfluidics for biomedical applications. Small. 2020, 16, e1903736. |
| 553. |
Sangtani, A.; Nag, O. K.; McMasters, J.; Panitch, A. Collagen-binding nanoparticles for extracellular anti-inflammatory peptide delivery decrease platelet activation, promote endothelial migration, and suppress inflammation. Acta Biomater. 2017, 49, 78-88.
doi: 10.1016/j.actbio.2016.11.023 URL |
| 554. | Sangtani, A.; Nag, O. K.; Field, L. D.; Breger, J. C.; Delehanty, J. B. Multifunctional nanoparticle composites: progress in the use of soft and hard nanoparticles for drug delivery and imaging. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2017, 9. |
| 555. |
Ji, M. L.; Jiang, H.; Zhang, X. J.; Shi, P. L.; Li, C.; Wu, H.; Wu, X. T.; Wang, Y. T.; Wang, C.; Lu, J. Preclinical development of a microRNA-based therapy for intervertebral disc degeneration. Nat Commun. 2018, 9, 5051.
doi: 10.1038/s41467-018-07360-1 URL |
| 556. |
Liang, C.; Li, H.; Li, C.; Yang, Z.; Zhou, X.; Tao, Y.; Xiao, Y.; Li, F.; Chen, Q. Fabrication of a layered microstructured polymeric microspheres as a cell carrier for nucleus pulposus regeneration. J Biomater Sci Polym Ed. 2012, 23, 2287-2302.
doi: 10.1163/156856211X614789 URL |
| 557. |
van der Meel, R.; Vehmeijer, L. J.; Kok, R. J.; Storm, G.; van Gaal, E. V. Ligand-targeted particulate nanomedicines undergoing clinical evaluation: current status. Adv Drug Deliv Rev. 2013, 65, 1284-1298.
doi: 10.1016/j.addr.2013.08.012 URL |
| 558. |
Lammers, T.; Kiessling, F.; Hennink, W. E.; Storm, G. Drug targeting to tumors: principles, pitfalls and (pre-) clinical progress. J Control Release. 2012, 161, 175-187.
doi: 10.1016/j.jconrel.2011.09.063 URL |
| 559. |
Xiao, S.; Tang, Y.; Lv, Z.; Lin, Y.; Chen, L. Nanomedicine - advantages for their use in rheumatoid arthritis theranostics. J Control Release. 2019, 316, 302-316.
doi: 10.1016/j.jconrel.2019.11.008 URL |
| 560. |
Nadra, I.; Boccaccini, A. R.; Philippidis, P.; Whelan, L. C.; McCarthy, G. M.; Haskard, D. O.; Landis, R. C. Effect of particle size on hydroxyapatite crystal-induced tumor necrosis factor alpha secretion by macrophages. Atherosclerosis. 2008, 196, 98-105.
doi: 10.1016/j.atherosclerosis.2007.02.005 URL |
| 561. |
Pradal, J.; Maudens, P.; Gabay, C.; Seemayer, C. A.; Jordan, O.; Allémann, E. Effect of particle size on the biodistribution of nano- and microparticles following intra-articular injection in mice. Int J Pharm. 2016, 498, 119-129.
doi: 10.1016/j.ijpharm.2015.12.015 URL |
| 562. |
Patel, S.; Kim, J.; Herrera, M.; Mukherjee, A.; Kabanov, A. V.; Sahay, G. Brief update on endocytosis of nanomedicines. Adv Drug Deliv Rev. 2019, 144, 90-111.
doi: 10.1016/j.addr.2019.08.004 URL |
| 563. |
Yameen, B.; Choi, W. I.; Vilos, C.; Swami, A.; Shi, J.; Farokhzad, O. C. Insight into nanoparticle cellular uptake and intracellular targeting. J Control Release. 2014, 190, 485-499.
doi: 10.1016/j.jconrel.2014.06.038 URL |
| 564. |
Kang, M. L.; Ko, J. Y.; Kim, J. E.; Im, G. I. Intra-articular delivery of kartogenin-conjugated chitosan nano/microparticles for cartilage regeneration. Biomaterials. 2014, 35, 9984-9994.
doi: 10.1016/j.biomaterials.2014.08.042 URL |
| 565. |
Chen, Z.; Liu, D.; Wang, J.; Wu, L.; Li, W.; Chen, J.; Cai, B. C.; Cheng, H. Development of nanoparticles-in-microparticles system for improved local retention after intra-articular injection. Drug Deliv. 2014, 21, 342-350.
doi: 10.3109/10717544.2013.848495 URL |
| 566. |
Abrego, G.; Alvarado, H.; Souto, E. B.; Guevara, B.; Bellowa, L. H.; Garduño, M. L.; Garcia, M. L.; Calpena, A. C. Biopharmaceutical profile of hydrogels containing pranoprofen-loaded PLGA nanoparticles for skin administration: In vitro, ex vivo and in vivo characterization. Int J Pharm. 2016, 501, 350-361.
doi: 10.1016/j.ijpharm.2016.01.071 URL |
| 567. |
Shirazi-Adl, A.; Taheri, M.; Urban, J. P. Analysis of cell viability in intervertebral disc: Effect of endplate permeability on cell population. J Biomech. 2010, 43, 1330-1336.
doi: 10.1016/j.jbiomech.2010.01.023 URL |
| 568. |
Bowles, R. D.; Williams, R. M.; Zipfel, W. R.; Bonassar, L. J. Self-assembly of aligned tissue-engineered annulus fibrosus and intervertebral disc composite via collagen gel contraction. Tissue Eng Part A. 2010, 16, 1339-1348.
doi: 10.1089/ten.tea.2009.0442 URL |
| 569. |
Mizuno, H.; Roy, A. K.; Zaporojan, V.; Vacanti, C. A.; Ueda, M.; Bonassar, L. J. Biomechanical and biochemical characterization of composite tissue-engineered intervertebral discs. Biomaterials. 2006, 27, 362-370.
doi: 10.1016/j.biomaterials.2005.06.042 URL |
| 570. |
Iatridis, J. C. Tissue engineering: Function follows form. Nat Mater. 2009, 8, 923-924.
doi: 10.1038/nmat2577 URL |
| 571. |
Nerurkar, N. L.; Elliott, D. M.; Mauck, R. L. Mechanical design criteria for intervertebral disc tissue engineering. J Biomech. 2010, 43, 1017-1030.
doi: 10.1016/j.jbiomech.2009.12.001 URL |
| 572. | Luk, K. D.; Ruan, D. K.; Chow, D. H.; Leong, J. C. Intervertebral disc autografting in a bipedal animal model. Clin Orthop Relat Res. 1997, 13-26. |
| 573. | Frick, S. L.; Hanley, E. N., Jr.; Meyer, R. A.; Jr.; Ramp, W. K.; Chapman, T. M. Lumbar intervertebral disc transfer. A canine study. Spine (Phila Pa 1976). 1994, 19, 1826-1834; discussion 1834-1835. |
| 574. | Chan, S. C.; Lam, S.; Leung, V. Y.; Chan, D.; Luk, K. D.; Cheung, K. M. Minimizing cryopreservation-induced loss of disc cell activity for storage of whole intervertebral discs. Eur Cell Mater. 2010, 19, 273-283. |
| 575. |
Ruan, D.; He, Q.; Ding, Y.; Hou, L.; Li, J.; Luk, K. D. Intervertebral disc transplantation in the treatment of degenerative spine disease: a preliminary study. Lancet. 2007, 369, 993-999.
doi: 10.1016/S0140-6736(07)60496-6 URL |
| 576. |
Ding, Y.; Ruan, D. K.; He, Q.; Hou, L. S.; Lin, J. N.; Cui, H. P. Imaging evaluation and relative significance in cases of cervical disk allografting: radiographic character after total disk transplantation. Clinical spine surgery. 2016, 29, E488-E495.
doi: 10.1097/BSD.0b013e318290fc41 URL |
| 577. |
Tumialán, L. M.; Ponton, R. P.; Garvin, A.; Gluf, W. M. Arthroplasty in the military: a preliminary experience with ProDisc-C and ProDisc-L. Neurosurg Focus. 2010, 28, E18.
doi: 10.3171/2010.1.FOCUS102 URL |
| 578. |
Yue, J. J.; Mo, F. F. Clinical study to evaluate the safety and effectiveness of the Aesculap Activ-L artificial disc in the treatment of degenerative disc disease. BMC Surg. 2010, 10, 14.
doi: 10.1186/1471-2482-10-14 URL |
| 579. |
Plais, N.; Thevenot, X.; Cogniet, A.; Rigal, J.; Le Huec, v. Maverick total disc arthroplasty performs well at 10 years follow-up: a prospective study with HRQL and balance analysis. Eur Spine J. 2018, 27, 720-727.
doi: 10.1007/s00586-017-5065-z URL |
| 580. |
Gornet, M. F.; Burkus, J. K.; Dryer, R. F.; Peloza, J. H.; Schranck, F. W.; Copay, A. G. Lumbar disc arthroplasty versus anterior lumbar interbody fusion: 5-year outcomes for patients in the Maverick disc investigational device exemption study. J Neurosurg Spine. 2019, 31, 347-356.
doi: 10.3171/2019.2.SPINE181037 URL |
| 581. |
Sandhu, F. A.; Dowlati, E.; Garica, R. Lumbar Arthroplasty: Past, Present, and Future. Neurosurgery. 2020, 86, 155-169.
doi: 10.1093/neuros/nyz439 URL |
| 582. | Austen, S.; Punt, I. M.; Cleutjens, J. P.; Willems, P. C.; Kurtz, S. M.; MacDonald, D. W.; van Rhijn, L. W.; van Ooij,, A. Clinical, radiological, histological and retrieval findings of Activ-L and Mobidisc total disc replacements: a study of two patients. Eur Spine J. 2012, 21 Suppl 4, S513-520. |
| 583. |
Yue, J. J.; Garcia, R.; Blumenthal, S.; Coric, D.; Patel, V. V.; Dinh, D. H.; Buttermann, G. R.; Deutsch, H.; Miller, L. E.; Persaud, E. J.; Ferko, N. C. Five-year results of a randomized controlled trial for lumbar artificial discs in single-level degenerative disc disease. Spine (Phila Pa 1976). 2019, 44, 1685-1696.
doi: 10.1097/BRS.0000000000003171 URL |
| 584. |
Aunoble, S.; Donkersloot, P.; Le Huec, J. C. Dislocations with intervertebral disc prosthesis: two case reports. Eur Spine J. 2004, 13, 464-467.
doi: 10.1007/s00586-004-0687-3 URL |
| 585. |
Shim, C. S.; Lee, S.; Maeng, D. H.; Lee, S. H. Vertical split fracture of the vertebral body following total disc replacement using ProDisc: report of two cases. J Spinal Disord Tech. 2005, 18, 465-469.
doi: 10.1097/01.bsd.0000159035.35365.df URL |
| 586. | François, J.; Coessens, R.; Lauweryns, P. Early removal of a Maverick disc prosthesis: surgical findings and morphological changes. Acta Orthop Belg. 2007, 73, 122-127. |
| 587. |
Zeh, A.; Becker, C.; Planert, M.; Lattke, P.; Wohlrab, D. Time-dependent release of cobalt and chromium ions into the serum following implantation of the metal-on-metal Maverick type artificial lumbar disc (Medtronic Sofamor Danek). Arch Orthop Trauma Surg. 2009, 129, 741-746.
doi: 10.1007/s00402-008-0677-8 URL |
| 588. |
Gornet, M. F.; Burkus, J. K.; Harper, M. L.; Chan, F. W.; Skipor, A. K.; Jacobs, J. J. Prospective study on serum metal levels in patients with metal-on-metal lumbar disc arthroplasty. Eur Spine J. 2013, 22, 741-746.
doi: 10.1007/s00586-012-2581-8 URL |
| 589. |
Natu, S.; Sidaginamale, R. P.; Gandhi, J.; Langton, D. J.; Nargol, A. V. Adverse reactions to metal debris: histopathological features of periprosthetic soft tissue reactions seen in association with failed metal on metal hip arthroplasties. J Clin Pathol. 2012, 65, 409-418.
doi: 10.1136/jclinpath-2011-200398 URL |
| 590. | Sasso, R. C.; Kenneth Burkus, J.; LeHuec, J. C. Retrograde ejaculation after anterior lumbar interbody fusion: transperitoneal versus retroperitoneal exposure. Spine (Phila Pa 1976). 2003, 28, 1023-1026. |
| 591. | Tropiano, P.; Huang, R. C.; Girardi, F. P.; Cammisa, F. P. Jr.; Marnay, T. Lumbar total disc replacement. Surgical technique. J Bone Joint Surg Am. 2006, 88 Suppl 1 Pt 1, 50-64. |
| 592. |
Härtl, R.; Joeris, A.; McGuire, R. A. Comparison of the safety outcomes between two surgical approaches for anterior lumbar fusion surgery: anterior lumbar interbody fusion (ALIF) and extreme lateral interbody fusion (ELIF). Eur Spine J. 2016, 25, 1484-1521.
doi: 10.1007/s00586-016-4407-6 URL |
| 593. |
Brau, S. A.; Spoonamore, M. J.; Snyder, L.; Gilbert, C.; Rhonda, G.; Williams, L. A.; Watkins, R. G. Nerve monitoring changes related to iliac artery compression during anterior lumbar spine surgery. Spine J. 2003, 3, 351-355.
doi: 10.1016/S1529-9430(03)00067-6 URL |
| 594. |
Cui, X. D.; Li, H. T.; Zhang, W.; Zhang, L. L.; Luo, Z. P.; Yang, H. L. Mid- to long-term results of total disc replacement for lumbar degenerative disc disease: a systematic review. J Orthop Surg Res. 2018, 13, 326.
doi: 10.1186/s13018-018-1032-6 URL |
| 595. |
Salzmann, S. N.; Shue, J.; Hughes, A. P. Lateral lumbar interbody fusion-outcomes and complications. Curr Rev Musculoskelet Med. 2017, 10, 539-546.
doi: 10.1007/s12178-017-9444-1 URL |
| 596. |
Martin, J. T.; Milby, A. H.; Chiaro, J. A.; Kim, D. H.; Hebela, N. M.; Smith, L. J.; Elliott, D. M.; Mauck, R. L. Translation of an engineered nanofibrous disc-like angle-ply structure for intervertebral disc replacement in a small animal model. Acta Biomater. 2014, 10, 2473-2481.
doi: 10.1016/j.actbio.2014.02.024 URL |
| 597. |
Norbertczak, H. T.; Ingham, E.; Fermor, H. L.; Wilcox, R. K. Decellularized intervertebral discs: a potential replacement for degenerate human discs. Tissue Eng Part C Methods. 2020, 26, 565-576.
doi: 10.1089/ten.tec.2020.0104 URL |
| 598. |
Hensley, A.; Rames, J.; Casler, V.; Rood, C.; Walters, J.; Fernandez, C.; Gill, S.; Mercuri, J. J. Decellularization and characterization of a whole intervertebral disk xenograft scaffold. J Biomed Mater Res A. 2018, 106, 2412-2423.
doi: 10.1002/jbm.a.v106.9 URL |
| 599. |
Moriguchi, Y.; Mojica-Santiago, J.; Grunert, P.; Pennicooke, B.; Berlin, C.; Khair, T.; Navarro-Ramirez, R.; Ricart Arbona, R. J.; Nguyen, J.; Härtl, R.; Bonassar, L. J. Total disc replacement using tissue-engineered intervertebral discs in the canine cervical spine. PLoS One. 2017, 12, e0185716.
doi: 10.1371/journal.pone.0185716 URL |
| 600. |
Grunert, P.; Borde, B. H.; Towne, S. B.; Moriguchi, Y.; Hudson, K. D.; Bonassar, L. J.; Härtl, R. Riboflavin crosslinked high-density collagen gel for the repair of annular defects in intervertebral discs: An in vivo study. Acta Biomater. 2015, 26, 215-224.
doi: 10.1016/j.actbio.2015.06.006 URL |
| 601. | Sloan, S. R., Jr.; Wipplinger, C.; Kirnaz, S.; Navarro-Ramirez, R.; Schmidt, F.; McCloskey, D.; Pannellini, T.; Schiavinato, A.; Härtl, R.; Bonassar, L. J. Combined nucleus pulposus augmentation and annulus fibrosus repair prevents acute intervertebral disc degeneration after discectomy. Sci Transl Med. 2020, 12, eaay2380. |
| 602. |
Pennicooke, B.; Hussain, I.; Berlin, C.; Sloan, S. R.; Borde, B.; Moriguchi, Y.; Lang, G.; Navarro-Ramirez, R.; Cheetham, J.; Bonassar, L. J.; Härtl, R. Annulus fibrosus repair using high-density collagen gel: an in vivo ovine model. Spine (Phila Pa 1976). 2018, 43, E208-E215.
doi: 10.1097/BRS.0000000000002334 URL |
| 603. |
Zhang, Y.; Drapeau, S.; An, H. S.; Markova, D.; Lenart, B. A.; Anderson, D. G. Histological features of the degenerating intervertebral disc in a goat disc-injury model. Spine (Phila Pa 1976). 2011, 36, 1519-1527.
doi: 10.1097/BRS.0b013e3181f60b39 URL |
| 604. |
Matta, A.; Karim, M. Z.; Gerami, H.; Jun, P.; Funabashi, M.; Kawchuk, G.; Goldstein, A.; Foltz, W.; Sussman, M.; Eek, B. C.; Erwin, W. M. NTG-101: a novel molecular therapy that halts the progression of degenerative disc disease. Sci Rep. 2018, 8, 16809.
doi: 10.1038/s41598-018-35011-4 URL |
| 605. | Long, R. G.; Ferguson, S. J.; Benneker, L. M.; Sakai, D.; Li, Z.; Pandit, A.; Grijpma, D. W.; Eglin, D.; Zeiter, S.; Schmid, T.; Eberli, U.; Nehrbass, D.; Di Pauli von Treuheim, T.; Alini, M.; Iatridis, J. C.; Grad, S. Morphological and biomechanical effects of annulus fibrosus injury and repair in an ovine cervical model. JOR Spine. 2020, 3, e1074. |
| 606. |
Liu, W.; Liu, D.; Zheng, J.; Shi, P.; Chou, P. H.; Oh, C.; Chen, D.; An, H. S.; Chee, A. Annulus fibrosus cells express and utilize C-C chemokine receptor 5 (CCR5) for migration. Spine J. 2017, 17, 720-726.
doi: 10.1016/j.spinee.2017.01.010 URL |
| 607. |
Huang, B. R.; Bau, D. T.; Chen, T. S.; Chuang, I. C.; Tsai, C. F.; Chang, P. C.; Hsu, H. C.; Lu, D. Y. Pro-inflammatory stimuli influence expression of intercellular adhesion molecule 1 in human anulus fibrosus cells through FAK/ERK/GSK3 and PKCδ signaling pathways. Int J Mol Sci. 2018, 20, 77.
doi: 10.3390/ijms20010077 URL |
| 608. |
Stich, S.; Möller, A.; Cabraja, M.; Krüger, J. P.; Hondke, S.; Endres, M.; Ringe, J.; Sittinger, M. Chemokine CCL25 induces migration and extracellular matrix production of anulus fibrosus-derived cells. Int J Mol Sci. 2018, 19, 2207.
doi: 10.3390/ijms19082207 URL |
| 609. |
Zhou, Z.; Zeiter, S.; Schmid, T.; Sakai, D.; Iatridis, J. C.; Zhou, G.; Richards, R. G.; Alini, M.; Grad, S.; Li, Z. Effect of the CCL5-releasing fibrin gel for intervertebral disc regeneration. Cartilage. 2020, 11, 169-180.
doi: 10.1177/1947603518764263 URL |
| 610. |
Magge, S. N.; Malik, S. Z.; Royo, N. C.; Chen, H. I.; Yu, L.; Snyder, E. Y.; O’Rourke, D. M.; Watson, D. J. Role of monocyte chemoattractant protein-1 (MCP-1/CCL2) in migration of neural progenitor cells toward glial tumors. J Neurosci Res. 2009, 87, 1547-1555.
doi: 10.1002/jnr.v87:7 URL |
| 611. |
Wang, L.; Li, H.; Lin, J.; He, R.; Chen, M.; Zhang, Y.; Liao, Z.; Zhang, C. CCR2 improves homing and engraftment of adipose-derived stem cells in dystrophic mice. Stem Cell Res Ther. 2021, 12, 12.
doi: 10.1186/s13287-020-02065-z URL |
| 612. |
Frapin, L.; Clouet, J.; Chédeville, C.; Moraru, C.; Samarut, E.; Henry, N.; André, M.; Bord, E.; Halgand, B.; Lesoeur, J.; Fusellier, M.; Guicheux, J.; Le Visage, C. Controlled release of biological factors for endogenous progenitor cell migration and intervertebral disc extracellular matrix remodelling. Biomaterials. 2020, 253, 120107.
doi: 10.1016/j.biomaterials.2020.120107 URL |
| 613. |
Watanabe, Y.; Tsuchiya, A.; Seino, S.; Kawata, Y.; Kojima, Y.; Ikarashi, S.; Starkey Lewis, P. J.; Lu, W. Y.; Kikuta, J.; Kawai, H.; Yamagiwa, S.; Forbes, S. J.; Ishii, M.; Terai, S. Mesenchymal stem cells and induced bone marrow-derived macrophages synergistically improve liver fibrosis in mice. Stem Cells Transl Med. 2019, 8, 271-284.
doi: 10.1002/sct3.2019.8.issue-3 URL |
| 614. |
Wang, Q.; Zhang, F.; Lei, Y.; Liu, P.; Liu, C.; Tao, Y. microRNA-322/424 promotes liver fibrosis by regulating angiogenesis through targeting CUL2/HIF-1α pathway. Life Sci. 2021, 266, 118819.
doi: 10.1016/j.lfs.2020.118819 URL |
| 615. |
Liu, W.; Wang, L.; Zhang, J.; Qiao, L.; Liu, Y.; Yang, X.; Zhang, J.; Zheng, W.; Ma, Z. Purification of recombinant human chemokine CCL2 in E. coli and its function in ovarian cancer. 3 Biotech. 2021, 11, 8.
doi: 10.1007/s13205-020-02571-0 URL |
| 616. |
Liu, W.; Wang, W.; Zhang, N.; Di, W. The role of CCL20-CCR6 axis in ovarian cancer metastasis. Onco Targets Ther. 2020, 13, 12739-12750.
doi: 10.2147/OTT.S280309 URL |
| 617. |
Korbecki, J.; Kojder, K.; Simińska, D.; Bohatyrewicz, R.; Gutowska, I.; Chlubek, D.; Baranowska-Bosiacka, I. CC chemokines in a tumor: a review of pro-cancer and anti-cancer properties of the ligands of receptors CCR1, CCR2, CCR3, and CCR4. Int J Mol Sci. 2020, 21, 8412.
doi: 10.3390/ijms21218412 URL |
| 618. |
Raghu, H.; Lepus, C. M.; Wang, Q.; Wong, H. H.; Lingampalli, N.; Oliviero, F.; Punzi, L.; Giori, N. J.; Goodman, S. B.; Chu, C. R.; Sokolove, J. B.; Robinson, W. H. CCL2/CCR2, but not CCL5/CCR5, mediates monocyte recruitment, inflammation and cartilage destruction in osteoarthritis. Ann Rheum Dis. 2017, 76, 914-922.
doi: 10.1136/annrheumdis-2016-210426 URL |
| 619. |
Schwager, J.; Richard, N.; Fowler, A.; Seifert, N.; Raederstorff, D. Carnosol and related substances modulate chemokine and cytokine production in macrophages and chondrocytes. Molecules. 2016, 21, 465.
doi: 10.3390/molecules21040465 URL |
| 620. |
Hingert, D.; Barreto Henriksson, H.; Baranto, A.; Brisby, H. BMP-3 promotes matrix production in co-cultured stem cells and disc cells from low back pain patients. Tissue Eng Part A. 2020, 26, 47-56.
doi: 10.1089/ten.tea.2019.0125 URL |
| 621. |
Elmasry, S.; Asfour, S.; de Rivero Vaccari, J. P.; Travascio, F. A computational model for investigating the effects of changes in bioavailability of insulin-like growth factor-1 on the homeostasis of the intervertebral disc. Comput Biol Med. 2016, 78, 126-137.
doi: 10.1016/j.compbiomed.2016.09.016 URL |
| 622. |
Takegami, K.; An, H. S.; Kumano, F.; Chiba, K.; Thonar, E. J.; Singh, K.; Masuda, K. Osteogenic protein-1 is most effective in stimulating nucleus pulposus and annulus fibrosus cells to repair their matrix after chondroitinase ABC-induced in vitro chemonucleolysis. Spine J. 2005, 5, 231-238.
doi: 10.1016/j.spinee.2004.11.001 URL |
| 623. |
Wallach, C. J.; Kim, J. S.; Sobajima, S.; Lattermann, C.; Oxner, W. M.; McFadden, K.; Robbins, P. D.; Gilbertson, L. G.; Kang, J. D. Safety assessment of intradiscal gene transfer: a pilot study. Spine J. 2006, 6, 107-112.
doi: 10.1016/j.spinee.2005.05.002 URL |
| 624. |
Cho, H.; Lee, S.; Park, S. H.; Huang, J.; Hasty, K. A.; Kim, S. J. Synergistic effect of combined growth factors in porcine intervertebral disc degeneration. Connect Tissue Res. 2013, 54, 181-186.
doi: 10.3109/03008207.2013.775258 URL |
| 625. | Vadalà, G.; Mozetic, P.; Rainer, A.; Centola, M.; Loppini, M.; Trombetta, M.; Denaro, V. Bioactive electrospun scaffold for annulus fibrosus repair and regeneration. Eur Spine J. 2012, 21 Suppl 1, S20-26. |
| 626. |
Murata, K.; Kokubun, T.; Onitsuka, K.; Oka, Y.; Kano, T.; Morishita, Y.; Ozone, K.; Kuwabara, N.; Nishimoto, J.; Isho, T.; Takayanagi, K.; Kanemura, N. Controlling joint instability after anterior cruciate ligament transection inhibits transforming growth factor-beta-mediated osteophyte formation. Osteoarthritis Cartilage. 2019, 27, 1185-1196.
doi: 10.1016/j.joca.2019.03.008 URL |
| 627. |
Kaneko, H.; Ishijima, M.; Futami, I.; Tomikawa-Ichikawa, N.; Kosaki, K.; Sadatsuki, R.; Yamada, Y.; Kurosawa, H.; Kaneko, K.; Arikawa-Hirasawa, E. Synovial perlecan is required for osteophyte formation in knee osteoarthritis. Matrix Biol. 2013, 32, 178-187.
doi: 10.1016/j.matbio.2013.01.004 URL |
| 628. |
Fuller, E. S.; Shu, C.; Smith, M. M.; Little, C. B.; Melrose, J. Hyaluronan oligosaccharides stimulate matrix metalloproteinase and anabolic gene expression in vitro by intervertebral disc cells and annular repair in vivo. J Tissue Eng Regen Med. 2018, 12, e216-e226.
doi: 10.1002/term.2319 URL |
| 629. |
Binch, A. L. A.; Fitzgerald, J. C.; Growney, E. A.; Barry, F. Cell-based strategies for IVD repair: clinical progress and translational obstacles. Nat Rev Rheumatol. 2021, 17, 158-175.
doi: 10.1038/s41584-020-00568-w URL |
| 630. | Chen, S.; Deng, X.; Ma, K.; Zhao, L.; Huang, D.; Li, Z.; Shao, Z. Icariin improves the viability and function of cryopreserved human nucleus pulposus-derived mesenchymal stem cells. Oxid Med Cell Longev. 2018, 2018, 3459612. |
| 631. |
Li, Z.; Chen, S.; Ma, K.; Lv, X.; Lin, H.; Hu, B.; He, R.; Shao, Z. CsA attenuates compression-induced nucleus pulposus mesenchymal stem cells apoptosis via alleviating mitochondrial dysfunction and oxidative stress. Life Sci. 2018, 205, 26-37.
doi: 10.1016/j.lfs.2018.05.014 URL |
| 632. |
Pas, H. I.; Winters, M.; Haisma, H. J.; Koenis, M. J.; Tol, J. L.; Moen, M. H. Stem cell injections in knee osteoarthritis: a systematic review of the literature. Br J Sports Med. 2017, 51, 1125-1133.
doi: 10.1136/bjsports-2016-096793 URL |
| 633. |
Song, H. G.; Rumma, R. T.; Ozaki, C. K.; Edelman, E. R.; Chen, C. S. Vascular tissue engineering: progress, challenges, and clinical promise. Cell Stem Cell. 2018, 22, 340-354.
doi: 10.1016/j.stem.2018.02.009 URL |
| 634. |
Jin, J. Stem Cell Treatments. JAMA. 2017, 317, 330.
doi: 10.1001/jama.2016.17822 URL |
| 635. |
Sackett, S. D.; Brown, M. E.; Tremmel, D. M.; Ellis, T.; Burlingham, W. J.; Odorico, J. S. Modulation of human allogeneic and syngeneic pluripotent stem cells and immunological implications for transplantation. Transplant Rev (Orlando). 2016, 30, 61-70.
doi: 10.1016/j.trre.2016.02.001 URL |
| 636. |
Xiao, L.; Huang, R.; Zhang, Y.; Li, T.; Dai, J.; Nannapuneni, N.; Chastanet, T. R.; Chen, M.; Shen, F. H.; Jin, L.; Dorn, H. C.; Li, X. A new formyl peptide receptor-1 antagonist conjugated fullerene nanoparticle for targeted treatment of degenerative disc diseases. ACS Appl Mater Interfaces. 2019, 11, 38405-38416.
doi: 10.1021/acsami.9b11783 URL |
| 637. |
Qu, Y.; Wang, Z.; Zhou, H.; Kang, M.; Dong, R.; Zhao, J. Oligosaccharide nanomedicine of alginate sodium improves therapeutic results of posterior lumbar interbody fusion with cages for degenerative lumbar disease in osteoporosis patients by downregulating serum miR-155. Int J Nanomedicine. 2017, 12, 8459-8469.
doi: 10.2147/IJN URL |
| 638. |
Robinson, K.; Platt, S.; Bibi, K.; Banovic, F.; Barber, R.; Howerth, E. W.; Madsen, G. A pilot study on the safety of a novel antioxidant nanoparticle delivery system and its indirect effects on cytokine levels in four dogs. Front Vet Sci. 2020, 7, 447.
doi: 10.3389/fvets.2020.00447 URL |
| 639. |
Tryfonidou, M. A.; de Vries, G.; Hennink, W. E.; Creemers, L. B. “Old Drugs, New Tricks” - local controlled drug release systems for treatment of degenerative joint disease. Adv Drug Deliv Rev. 2020, 160, 170-185.
doi: 10.1016/j.addr.2020.10.012 URL |
| 640. |
van Tulder, M.; Koes, B.; Bombardier, C. Low back pain. Best Pract Res Clin Rheumatol. 2002, 16, 761-775.
doi: 10.1053/berh.2002.0267 URL |
| 641. | van Tulder, M.; Koes, B. Low back pain (chronic). Clin Evid. 2006, 1634-1653. |
| 642. |
van Tulder, M.; Becker, A.; Bekkering, T.; Breen, A.; del Real, M. T.; Hutchinson, A.; Koes, B.; Laerum, E.; Malmivaara, A.; COST B13 Working Group on Guidelines for the Management of Acute Low Back Pain in Primary Care. Chapter 3. European guidelines for the management of acute nonspecific low back pain in primary care. Eur Spine J. 2006, 15 Suppl 2, S169-191.
doi: 10.1007/s00586-006-1071-2 URL |
| 643. |
Lyu, F. J.; Cui, H.; Pan, H.; Mc Cheung, K.; Cao, X.; Iatridis, J. C.; Zheng, Z. Painful intervertebral disc degeneration and inflammation: from laboratory evidence to clinical interventions. Bone Res. 2021, 9, 7.
doi: 10.1038/s41413-020-00125-x URL |
| 644. |
Mosley, G. E.; Evashwick-Rogler, T. W.; Lai, A.; Iatridis, J. C. Looking beyond the intervertebral disc: the need for behavioral assays in models of discogenic pain. Ann N Y Acad Sci. 2017, 1409, 51-66.
doi: 10.1111/nyas.2017.1409.issue-1 URL |
| 645. |
Kambiz, S.; Baas, M.; Duraku, L. S.; Kerver, A. L.; Koning, A. H.; Walbeehm, E. T.; Ruigrok, T. J. Innervation mapping of the hind paw of the rat using Evans Blue extravasation, Optical Surface Mapping and CASAM. J Neurosci Methods. 2014, 229, 15-27.
doi: 10.1016/j.jneumeth.2014.03.015 URL |
| 646. |
Símaro, G. V.; Lemos, M.; Mangabeira da Silva, J. J.; Ribeiro, V. P.; Arruda, C.; Schneider, A. H.; Wagner de Souza Wanderley, C.; Carneiro, L. J.; Mariano, R. L.; Ambrósio, S. R.; Faloni de Andrade, S.; Banderó-Filho, V. C.; Sasse, A.; Sheridan, H.; Andrade, E. S. M. L.; Bastos, J. K. Antinociceptive and anti-infammatory activities of Copaifera pubiflora Benth oleoresin and its major metabolite ent-hardwickiic acid. J Ethnopharmacol. 2021, 271, 113883.
doi: 10.1016/j.jep.2021.113883 URL |
| 647. | Molstad, D. H. H.; Bradley, E. W. Pain and Activity Measurements. Methods Mol Biol. 2021, 2221, 291-299. |
| 648. |
Heinsinger, N. M.; Spagnuolo, G.; Allahyari, R. V.; Galer, S.; Fox, T.; Jaffe, D. A.; Thomas, S. J.; Iacovitti, L.; Lepore, A. C. Facial grimace testing as an assay of neuropathic pain-related behavior in a mouse model of cervical spinal cord injury. Exp Neurol. 2020, 334, 113468.
doi: 10.1016/j.expneurol.2020.113468 URL |
| 649. |
Deuis, J. R.; Dvorakova, L. S.; Vetter, I. Methods used to evaluate pain behaviors in rodents. Front Mol Neurosci. 2017, 10, 284.
doi: 10.3389/fnmol.2017.00284 URL |
| 650. |
Chaplan, S. R.; Bach, F. W.; Pogrel, J. W.; Chung, J. M.; Yaksh, T. L. Quantitative assessment of tactile allodynia in the rat paw. J Neurosci Methods. 1994, 53, 55-63.
doi: 10.1016/0165-0270(94)90144-9 URL |
| 651. |
Evans, D. W.; De Nunzio, A. M. Controlled manual loading of body tissues: towards the next generation of pressure algometer. Chiropr Man Therap. 2020, 28, 51.
doi: 10.1186/s12998-020-00340-7 URL |
| 652. |
Plinsinga, M. L.; Meeus, M.; Brink, M. S.; Heugen, N.; van Wilgen, P. Evidence of widespread mechanical hyperalgesia but not exercise induced analgesia in athletes with mild patellar tendinopathy compared to pain-free matched controls: a blinded exploratory study. Am J Phys Med Rehabil. 2020. doi: 10.1097/PHM.0000000000001673.
doi: 10.1097/PHM.0000000000001673 URL |
| 653. |
Ilnytska, O.; Lyzogubov, V. V.; Stevens, M. J.; Drel, V. R.; Mashtalir, N.; Pacher, P.; Yorek, M. A.; Obrosova, I. G. Poly(ADP-ribose) polymerase inhibition alleviates experimental diabetic sensory neuropathy. Diabetes. 2006, 55, 1686-1694.
doi: 10.2337/db06-0067 URL |
| 654. |
Yalcin, I.; Charlet, A.; Freund-Mercier, M. J.; Barrot, M.; Poisbeau, P. Differentiating thermal allodynia and hyperalgesia using dynamic hot and cold plate in rodents. J Pain. 2009, 10, 767-773.
doi: 10.1016/j.jpain.2009.01.325 URL |
| 655. | Bannon, A. W.; Malmberg, A. B. Models of nociception: hot-plate, tail-flick, and formalin tests in rodents. Curr Protoc Neurosci. 2007, Chapter 8, Unit 8.9. |
| 656. |
Driscoll, C.; Chanalaris, A.; Knights, C.; Ismail, H.; Sacitharan, P. K.; Gentry, C.; Bevan, S.; Vincent, T. L. Nociceptive sensitizers are regulated in damaged joint tissues, including articular cartilage, when osteoarthritic mice display pain behavior. Arthritis Rheumatol. 2016, 68, 857-867.
doi: 10.1002/art.v68.4 URL |
| 657. |
Millecamps, M.; Stone, L. S. Delayed onset of persistent discogenic axial and radiating pain after a single-level lumbar intervertebral disc injury in mice. Pain. 2018, 159, 1843-1855.
doi: 10.1097/j.pain.0000000000001284 URL |
| 658. |
Wang, T.; Hurwitz, O.; Shimada, S. G.; Tian, D.; Dai, F.; Zhou, J.; Ma, C.; LaMotte, R. H. Anti-nociceptive effects of bupivacaine-encapsulated PLGA nanoparticles applied to the compressed dorsal root ganglion in mice. Neurosci Lett. 2018, 668, 154-158.
doi: 10.1016/j.neulet.2018.01.031 URL |
| 659. |
Kim, J. S.; Kroin, J. S.; Li, X.; An, H. S.; Buvanendran, A.; Yan, D.; Tuman, K. J.; van Wijnen, A. J.; Chen, D.; Im, H. J. The rat intervertebral disk degeneration pain model: relationships between biological and structural alterations and pain. Arthritis Res Ther. 2011, 13, R165.
doi: 10.1186/ar3485 URL |
| 660. |
Kinser, A. M.; Sands, W. A.; Stone, M. H. Reliability and validity of a pressure algometer. J Strength Cond Res. 2009, 23, 312-314.
doi: 10.1519/JSC.0b013e31818f051c URL |
| 661. | O’Callaghan, J. P.; Holtzman, S. G. Quantification of the analgesic activity of narcotic antagonists by a modified hot-plate procedure. J Pharmacol Exp Ther. 1975, 192, 497-505. |
| 662. |
Yoon, C.; Wook, Y. Y.; Sik, N. H.; Ho, K. S.; Mo, C. J. Behavioral signs of ongoing pain and cold allodynia in a rat model of neuropathic pain. Pain. 1994, 59, 369-376.
doi: 10.1016/0304-3959(94)90023-X URL |
| 663. | Tajerian, M.; Millecamps, M.; Stone, L. S. Morphine and clonidine synergize to ameliorate low back pain in mice. Pain research and treatment. 2012, 2012, 150842. |
| 664. |
Aono, Y.; Hasegawa, H.; Yamazaki, Y.; Shimada, T.; Fujita, T.; Yamashita, T.; Fukumoto, S. Anti-FGF-23 neutralizing antibodies ameliorate muscle weakness and decreased spontaneous movement of Hyp mice. J Bone Miner Res. 2011, 26, 803-810.
doi: 10.1002/jbmr.275 URL |
| 665. |
Sluka, K. A.; Rasmussen, L. A. Fatiguing exercise enhances hyperalgesia to muscle inflammation. Pain. 2010, 148, 188-197.
doi: 10.1016/j.pain.2009.07.001 URL |
| 666. |
Call, J. A.; McKeehen, J. N.; Novotny, S. A.; Lowe, D. A. Progressive resistance voluntary wheel running in the mdx mouse. Muscle Nerve. 2010, 42, 871-880.
doi: 10.1002/mus.21764 URL |
| 667. |
Krock, E.; Millecamps, M.; Currie, J. B.; Stone, L. S.; Haglund, L. Low back pain and disc degeneration are decreased following chronic toll-like receptor 4 inhibition in a mouse model. Osteoarthritis Cartilage. 2018, 26, 1236-1246.
doi: 10.1016/j.joca.2018.06.002 URL |
| 668. |
Keshavarzi, S.; Kermanshahi, S.; Karami, L.; Motaghinejad, M.; Motevalian, M.; Sadr, S. Protective role of metformin against methamphetamine induced anxiety, depression, cognition impairment and neurodegeneration in rat: The role of CREB/BDNF and Akt/GSK3 signaling pathways. Neurotoxicology. 2019, 72, 74-84.
doi: 10.1016/j.neuro.2019.02.004 URL |
| 669. |
Tappe-Theodor, A.; Kuner, R. Studying ongoing and spontaneous pain in rodents--challenges and opportunities. Eur J Neurosci. 2014, 39, 1881-1890.
doi: 10.1111/ejn.12643 URL |
| 670. |
Denenberg, V. H. Open-field bheavior in the rat: what does it mean? Ann N Y Acad Sci. 1969, 159, 852-859.
doi: 10.1111/nyas.1969.159.issue-3 URL |
| 671. |
Parent, A. J.; Beaudet, N.; Beaudry, H.; Bergeron, J.; Bérubé, P.; Drolet, G.; Sarret, P.; Gendron, L. Increased anxiety-like behaviors in rats experiencing chronic inflammatory pain. Behav Brain Res. 2012, 229, 160-167.
doi: 10.1016/j.bbr.2012.01.001 URL |
| 672. | Portfors, C. V. Types and functions of ultrasonic vocalizations in laboratory rats and mice. J Am Assoc Lab Anim Sci. 2007, 46, 28-34. |
| 673. | Williams, W. O.; Riskin, D. K.; Mott, A. K. Ultrasonic sound as an indicator of acute pain in laboratory mice. J Am Assoc Lab Anim Sci. 2008, 47, 8-10. |
| 674. |
Langford, D. J.; Bailey, A. L.; Chanda, M. L.; Clarke, S. E.; Drummond, T. E.; Echols, S.; Glick, S.; Ingrao, J.; Klassen-Ross, T.; Lacroix-Fralish, M. L.; Matsumiya, L.; Sorge, R. E.; Sotocinal, S. G.; Tabaka, J. M.; Wong, D.; van den Maagdenberg, A. M.; Ferrari, M. D.; Craig, K. D.; Mogil, J. S. Coding of facial expressions of pain in the laboratory mouse. Nat Methods. 2010, 7, 447-449.
doi: 10.1038/nmeth.1455 URL |
| 675. | Sotocinal, S. G.; Sorge, R. E.; Zaloum, A.; Tuttle, A. H.; Martin, L. J.; Wieskopf, J. S.; Mapplebeck, J. C.; Wei, P.; Zhan, S.; Zhang, S.; McDougall, J. J.; King, O. D.; Mogil, J. S. The Rat Grimace Scale: a partially automated method for quantifying pain in the laboratory rat via facial expressions. Mol Pain. 2011, 7, 55. |
| 676. | Hohlbaum, K.; Corte, G. M.; Humpenöder, M.; Merle, R.; Thöne-Reineke, C. Reliability of the Mouse Grimace Scale in C57BL/6JRj Mice. Animals (Basel). 2020, 10, 1648. |
| 677. |
Miller, A. L.; Leach, M. C. The Mouse Grimace Scale: a clinically useful tool? PLoS One. 2015, 10, e0136000.
doi: 10.1371/journal.pone.0136000 URL |
| 678. | Daneshmandi, L.; Barajaa, M.; Tahmasbi Rad, A.; Sydlik, S. A.; Laurencin, C. T. Graphene-based biomaterials for bone regenerative engineering: a comprehensive review of the field and considerations regarding biocompatibility and biodegradation. Adv Healthc Mater. 2021, 10, e2001414. |
| 679. |
Rahmati, M.; Mozafari, M. Biocompatibility of alumina-based biomaterials-A review. J Cell Physiol. 2019, 234, 3321-3335.
doi: 10.1002/jcp.27292 URL |
| 680. |
Patel, H. Blood biocompatibility enhancement of biomaterials by heparin immobilization: a review. Blood Coagul Fibrinolysis. 2021, 32, 237-247.
doi: 10.1097/MBC.0000000000001011 URL |
| 681. |
Huang, H.; Evankovich, J.; Yan, W.; Nace, G.; Zhang, L.; Ross, M.; Liao, X.; Billiar, T.; Xu, J.; Esmon, C. T.; Tsung, A. Endogenous histones function as alarmins in sterile inflammatory liver injury through Toll-like receptor 9 in mice. Hepatology. 2011, 54, 999-1008.
doi: 10.1002/hep.v54.3 URL |
| 682. |
Daly, K. A.; Liu, S.; Agrawal, V.; Brown, B. N.; Johnson, S. A.; Medberry, C. J.; Badylak, S. F. Damage associated molecular patterns within xenogeneic biologic scaffolds and their effects on host remodeling. Biomaterials. 2012, 33, 91-101.
doi: 10.1016/j.biomaterials.2011.09.040 URL |
| 683. |
Alsousi, A. A.; Igwe, O. J. Redox-active trace metal-induced release of high mobility group box 1(HMGB1) and inflammatory cytokines in fibroblast-like synovial cells is Toll-like receptor 4 (TLR4) dependent. Biochim Biophys Acta Mol Basis Dis. 2018, 1864, 3847-3858.
doi: 10.1016/j.bbadis.2018.08.029 URL |
| 684. |
Ranzato, E.; Patrone, M.; Pedrazzi, M.; Burlando, B. HMGb1 promotes scratch wound closure of HaCaT keratinocytes via ERK1/2 activation. Mol Cell Biochem. 2009, 332, 199-205.
doi: 10.1007/s11010-009-0192-4 URL |
| 685. |
Fahmy-Garcia, S.; van Driel, M.; Witte-Buoma, J.; Walles, H.; van Leeuwen, J.; van Osch, G.; Farrell, E. NELL-1, HMGB1, and CCN2 enhance migration and vasculogenesis, but not osteogenic differentiation compared to BMP2. Tissue Eng Part A. 2018, 24, 207-218.
doi: 10.1089/ten.tea.2016.0537 URL |
| 686. |
Steinke, J. W.; Platts-Mills, T. A.; Commins, S. P. The alpha-gal story: lessons learned from connecting the dots. J Allergy Clin Immunol. 2015, 135, 589-596; quiz 597.
doi: 10.1016/j.jaci.2014.12.1947 URL |
| 687. |
Gabrielli, A.; Candela, M.; Ricciatti, A. M.; Caniglia, M. L.; Wieslander, J. Antibodies to mouse laminin in patients with systemic sclerosis (scleroderma) recognize galactosyl (alpha 1-3)-galactose epitopes. Clin Exp Immunol. 1991, 86, 367-373.
doi: 10.1111/j.1365-2249.1991.tb02939.x URL |
| 688. | Perea-Gil, I.; Uriarte, J. J.; Prat-Vidal, C.; Gálvez-Montón, C.; Roura, S.; Llucià-Valldeperas, A.; Soler-Botija, C.; Farré, R.; Navajas, D.; Bayes-Genis, A. In vitro comparative study of two decellularization protocols in search of an optimal myocardial scaffold for recellularization. Am J Transl Res. 2015, 7, 558-573. |
| 689. |
Ning, L. J.; Jiang, Y. L.; Zhang, C. H.; Zhang, Y.; Yang, J. L.; Cui, J.; Zhang, Y. J.; Yao, X.; Luo, J. C.; Qin, T. W. Fabrication and characterization of a decellularized bovine tendon sheet for tendon reconstruction. J Biomed Mater Res A. 2017, 105, 2299-2311.
doi: 10.1002/jbm.v105.8 URL |
| 690. |
Bourgine, P. E.; Pippenger, B. E.; Todorov, A., Jr.; Tchang, L.; Martin, I. Tissue decellularization by activation of programmed cell death. Biomaterials. 2013, 34, 6099-6108.
doi: 10.1016/j.biomaterials.2013.04.058 URL |
| 691. | Limana, F.; Germani, A.; Zacheo, A.; Kajstura, J.; Di Carlo, A.; Borsellino, G.; Leoni, O.; Palumbo, R.; Battistini, L.; Rastaldo, R.; Müller, S.; Pompilio, G.; Anversa, P.; Bianchi, M. E.; Capogrossi, M. C. Exogenous high-mobility group box 1 protein induces myocardial regeneration after infarction via enhanced cardiac C-kit+ cell proliferation and differentiation. Circ Res. 2005, 97, e73-83. |
| 692. |
van Steenberghe, M.; Schubert, T.; Xhema, D.; Bouzin, C.; Guiot, Y.; Duisit, J.; Abdelhamid, K.; Gianello, P. Enhanced vascular regeneration with chemically/physically treated bovine/human pericardium in rodents. J Surg Res. 2018, 222, 167-179.
doi: 10.1016/j.jss.2017.09.043 URL |
| 693. |
Duisit, J.; Orlando, G.; Debluts, D.; Maistriaux, L.; Xhema, D.; de Bisthoven, Y. J.; Galli, C.; Peloso, A.; Behets, C.; Lengelé, B.; Gianello, P. Decellularization of the porcine ear generates a biocompatible, nonimmunogenic extracellular matrix platform for face subunit bioengineering. Ann Surg. 2018, 267, 1191-1201.
doi: 10.1097/SLA.0000000000002181 URL |
| 694. |
Wang, Z.; Sun, F.; Lu, Y.; Pan, S.; Yang, W.; Zhang, G.; Ma, J.; Shi, H. Rapid preparation of decellularized trachea as a 3D scaffold for organ engineering. Int J Artif Organs. 2021, 44, 55-64.
doi: 10.1177/0391398820924041 URL |
| 695. |
Naso, F.; Gandaglia, A.; Iop, L.; Spina, M.; Gerosa, G. Alpha-Gal detectors in xenotransplantation research: a word of caution. Xenotransplantation. 2012, 19, 215-220.
doi: 10.1111/xen.2012.19.issue-4 URL |
| 696. |
Gao, H. W.; Li, S. B.; Sun, W. Q.; Yun, Z. M.; Zhang, X.; Song, J. W.; Zhang, S. K.; Leng, L.; Ji, S. P.; Tan, Y. X.; Gong, F. Quantification of α-Gal antigen removal in the porcine dermal tissue by α-galactosidase. Tissue Eng Part C Methods. 2015, 21, 1197-1204.
doi: 10.1089/ten.tec.2015.0129 URL |
| 697. |
Kim, M. S.; Lim, H. G.; Kim, Y. J. Calcification of decellularized and alpha-galactosidase-treated bovine pericardial tissue in an alpha-Gal knock-out mouse implantation model: comparison with primate pericardial tissue. Eur J Cardiothorac Surg. 2016, 49, 894-900.
doi: 10.1093/ejcts/ezv189 URL |
| 698. |
Katti, D. S.; Lakshmi, S.; Langer, R.; Laurencin, C. T. Toxicity, biodegradation and elimination of polyanhydrides. Adv Drug Deliv Rev. 2002, 54, 933-961.
doi: 10.1016/S0169-409X(02)00052-2 URL |
| 699. |
Cuesta, A.; Del Valle, M. E.; García-Suárez, O.; Viña, E.; Cabo, R.; Vázquez, G.; Cobo, J. L.; Murcia, A.; Alvarez-Vega, M.; García-Cosamalón, J.; Vega, J. A. Acid-sensing ion channels in healthy and degenerated human intervertebral disc. Connect Tissue Res. 2014, 55, 197-204.
doi: 10.3109/03008207.2014.884083 URL |
| 700. |
Zhou, R. P.; Wu, X. S.; Wang, Z. S.; Xie, Y. Y.; Ge, J. F.; Chen, F. H. Novel insights into acid-sensing ion channels: implications for degenerative diseases. Aging Dis. 2016, 7, 491-501.
doi: 10.14336/AD.2015.1213 URL |
| 701. |
Alizadeh, M.; Rezakhani, L.; Khodaei, M.; Soleimannejad, M.; Alizadeh, A. Evaluating the effects of vacuum on the microstructure and biocompatibility of bovine decellularized pericardium. J Tissue Eng Regen Med. 2021, 15, 116-128.
doi: 10.1002/term.v15.2 URL |
| 702. |
Cheng, J.; Li, J.; Cai, Z.; Xing, Y.; Wang, C.; Guo, L.; Gu, Y. Decellularization of porcine carotid arteries using low-concentration sodium dodecyl sulfate. Int J Artif Organs. 2020. doi: 10.1177/0391398820975420.
doi: 10.1177/0391398820975420 URL |
| 703. |
Wang, C.; Wang, M.; Xu, T.; Zhang, X.; Lin, C.; Gao, W.; Xu, H.; Lei, B.; Mao, C. Engineering bioactive self-healing antibacterial exosomes hydrogel for promoting chronic diabetic wound healing and complete skin regeneration. Theranostics. 2019, 9, 65-76.
doi: 10.7150/thno.29766 URL |
| 704. |
Liu, S.; Zhou, C.; Mou, S.; Li, J.; Zhou, M.; Zeng, Y.; Luo, C.; Sun, J.; Wang, Z.; Xu, W. Biocompatible graphene oxide-collagen composite aerogel for enhanced stiffness and in situ bone regeneration. Mater Sci Eng C Mater Biol Appl. 2019, 105, 110137.
doi: 10.1016/j.msec.2019.110137 URL |
| 705. |
Liu, Y.; Segura, T. Biomaterials-mediated regulation of macrophage cell fate. Front Bioeng Biotechnol. 2020, 8, 609297.
doi: 10.3389/fbioe.2020.609297 URL |
| 706. | Lee, J.; Byun, H.; Madhurakkat Perikamana, S. K.; Lee, S.; Shin, H. Current advances in immunomodulatory biomaterials for bone regeneration. Adv Healthc Mater. 2019, 8, e1801106. |
| 707. | Christo, S. N.; Diener, K. R.; Bachhuka, A.; Vasilev, K.; Hayball, J. D. Innate immunity and biomaterials at the nexus: friends or foes. Biomed Res Int. 2015, 2015, 342304. |
| 708. | Varma, D. M.; Lin, H. A.; Long, R. G.; Gold, G. T.; Hecht, A. C.; Iatridis, J. C.; Nicoll, S. B. Thermoresponsive, redox-polymerized cellulosic hydrogels undergo in situ gelation and restore intervertebral disc biomechanics post discectomy. Eur Cell Mater. 2018, 35, 300-317. |
| 709. |
Shaha, R. K.; Merkel, D. R.; Anderson, M. P.; Devereaux, E. J.; Patel, R. R.; Torbati, A. H.; Willett, N.; Yakacki, C. M.; Frick, C. P. Biocompatible liquid-crystal elastomers mimic the intervertebral disc. Mech Behav Biomed Mater. 2020, 107, 103757.
doi: 10.1016/j.jmbbm.2020.103757 URL |
| 710. |
Frith, J. E.; Cameron, A. R.; Menzies, D. J.; Ghosh, P.; Whitehead, D. L.; Gronthos, S.; Zannettino, A. C.; Cooper-White, J. J. An injectable hydrogel incorporating mesenchymal precursor cells and pentosan polysulphate for intervertebral disc regeneration. Biomaterials. 2013, 34, 9430-9440.
doi: 10.1016/j.biomaterials.2013.08.072 URL |
| 711. |
Zhu, D.; Cockerill, I.; Su, Y.; Zhang, Z.; Fu, J.; Lee, K. W.; Ma, J.; Okpokwasili, C.; Tang, L.; Zheng, Y.; Qin, Y. X.; Wang, Y. Mechanical strength, biodegradation, and in vitro and in vivo biocompatibility of Zn biomaterials. ACS Appl Mater Interfaces. 2019, 11, 6809-6819.
doi: 10.1021/acsami.8b20634 URL |
| 712. |
Huang, Y. C.; Urban, J. P.; Luk, K. D. Intervertebral disc regeneration: do nutrients lead the way? Nat Rev Rheumatol. 2014, 10, 561-566.
doi: 10.1038/nrrheum.2014.91 URL |
| 713. |
Sawamura, K.; Ikeda, T.; Nagae, M.; Okamoto, S.; Mikami, Y.; Hase, H.; Ikoma, K.; Yamada, T.; Sakamoto, H.; Matsuda, K.; Tabata, Y.; Kawata, M.; Kubo, T. Characterization of in vivo effects of platelet-rich plasma and biodegradable gelatin hydrogel microspheres on degenerated intervertebral discs. Tissue Eng Part A. 2009, 15, 3719-3727.
doi: 10.1089/ten.tea.2008.0697 URL |
| 714. |
Santos, J. M.; Pereira, S.; Sequeira, D. B.; Messias, A. L.; Martins, J. B.; Cunha, H.; Palma, P. J.; Santos, A. C. Biocompatibility of a bioceramic silicone-based sealer in subcutaneous tissue. J Oral Sci. 2019, 61, 171-177.
doi: 10.2334/josnusd.18-0145 URL |
| 715. |
Benetti, F.; de Azevedo Queiroz Í, O.; Oliveira, P. H. C.; Conti, L. C.; Azuma, M. M.; Oliveira, S. H. P.; Cintra, L. T. A. Cytotoxicity and biocompatibility of a new bioceramic endodontic sealer containing calcium hydroxide. Braz Oral Res. 2019, 33, e042.
doi: 10.1590/1807-3107bor-2019.vol33.0042 URL |
| 716. |
Tukmachev, D.; Forostyak, S.; Koci, Z.; Zaviskova, K.; Vackova, I.; Vyborny, K.; Sandvig, I.; Sandvig, A.; Medberry, C. J.; Badylak, S. F.; Sykova, E.; Kubinova, S. Injectable extracellular matrix hydrogels as scaffolds for spinal cord injury repair. Tissue Eng Part A. 2016, 22, 306-317.
doi: 10.1089/ten.tea.2015.0422 URL |
| 717. |
Wolf, M. T.; Daly, K. A.; Brennan-Pierce, E. P.; Johnson, S. A.; Carruthers, C. A.; D’Amore, A.; Nagarkar, S. P.; Velankar, S. S.; Badylak, S. F. A hydrogel derived from decellularized dermal extracellular matrix. Biomaterials. 2012, 33, 7028-7038.
doi: 10.1016/j.biomaterials.2012.06.051 URL |
| 718. |
Zhang, C.; Shang, Y.; Chen, X.; Midgley, A. C.; Wang, Z.; Zhu, D.; Wu, J.; Chen, P.; Wu, L.; Wang, X.; Zhang, K.; Wang, H.; Kong, D.; Yang, Z.; Li, Z.; Chen, X. Supramolecular nanofibers containing arginine-glycine-aspartate (RGD) peptides boost therapeutic efficacy of extracellular vesicles in kidney repair. ACS Nano. 2020, 14, 12133-12147.
doi: 10.1021/acsnano.0c05681 URL |
| 719. |
Ong, S. R.; Trabbic-Carlson, K. A.; Nettles, D. L.; Lim, D. W.; Chilkoti, A.; Setton, L. A. Epitope tagging for tracking elastin-like polypeptides. Biomaterials. 2006, 27, 1930-1935.
doi: 10.1016/j.biomaterials.2005.10.018 URL |
| 720. | Sharma, P.; Brown, S.; Walter, G.; Santra, S.; Moudgil, B. Nanoparticles for bioimaging. Adv Colloid Interface Sci. 2006, 123-126, 471-485. |
| 721. |
Frangioni, J. V. In vivo near-infrared fluorescence imaging. Curr Opin Chem Biol. 2003, 7, 626-634.
doi: 10.1016/j.cbpa.2003.08.007 URL |
| 722. |
Jalani, G.; Naccache, R.; Rosenzweig, D. H.; Lerouge, S.; Haglund, L.; Vetrone, F.; Cerruti, M. Real-time, non-invasive monitoring of hydrogel degradation using LiYF4:Yb(3+)/Tm(3+) NIR-to-NIR upconverting nanoparticles. Nanoscale. 2015, 7, 11255-11262.
doi: 10.1039/C5NR02482J URL |
| 723. | Sheehan, N. J. Magnetic resonance imaging for low back pain: indications and limitations. Postgrad Med J. 2010, 86, 374-378. |
| 724. |
Lai, A.; Chow, D. H.; Siu, W. S.; Holmes, A. D.; Tang, F. H. Reliability of radiographic intervertebral disc height measurement for in vivo rat-tail model. Med Eng Phys. 2007, 29, 814-819.
doi: 10.1016/j.medengphy.2006.08.013 URL |
| 725. |
Masuda, K.; Aota, Y.; Muehleman, C.; Imai, Y.; Okuma, M.; Thonar, E. J.; Andersson, G. B.; An, H. S. A novel rabbit model of mild, reproducible disc degeneration by an anulus needle puncture: correlation between the degree of disc injury and radiological and histological appearances of disc degeneration. Spine (Phila Pa 1976). 2005, 30, 5-14.
doi: 10.1097/01.brs.0000148152.04401.20 URL |
| 726. |
Deyo, R. A.; Loeser, J. D.; Bigos, S. J. Herniated lumbar intervertebral disk. Ann Intern Med. 1990, 112, 598-603.
doi: 10.7326/0003-4819-112-8-598 URL |
| 727. |
Ng, S. K.; Urquhart, D. M.; Fitzgerald, P. B.; Cicuttini, F. M.; Hussain, S. M.; Fitzgibbon, B. M. The relationship between structural and functional brain changes and altered emotion and cognition in chronic low back pain brain changes: a systematic review of MRI and fMRI studies. Clin J Pain. 2018, 34, 237-261.
doi: 10.1097/AJP.0000000000000534 URL |
| 728. |
Roughley, P. J.; Alini, M.; Antoniou, J. The role of proteoglycans in aging, degeneration and repair of the intervertebral disc. Biochem Soc Trans. 2002, 30, 869-874.
doi: 10.1042/bst0300869 URL |
| 729. |
Pfirrmann, C. W.; Metzdorf, A.; Zanetti, M.; Hodler, J.; Boos, N. Magnetic resonance classification of lumbar intervertebral disc degeneration. Spine (Phila Pa 1976). 2001, 26, 1873-1878.
doi: 10.1097/00007632-200109010-00011 URL |
| 730. |
Luoma, K.; Vehmas, T.; Riihimäki, H.; Raininko, R. Disc height and signal intensity of the nucleus pulposus on magnetic resonance imaging as indicators of lumbar disc degeneration. Spine (Phila Pa 1976). 2001, 26, 680-686.
doi: 10.1097/00007632-200103150-00026 URL |
| 731. |
Benneker, L. M.; Heini, P. F.; Anderson, S. E.; Alini, M.; Ito, K. Correlation of radiographic and MRI parameters to morphological and biochemical assessment of intervertebral disc degeneration. Eur Spine J. 2005, 14, 27-35.
doi: 10.1007/s00586-004-0759-4 URL |
| 732. |
Wang, A. M.; Cao, P.; Yee, A.; Chan, D.; Wu, E. X. Detection of extracellular matrix degradation in intervertebral disc degeneration by diffusion magnetic resonance spectroscopy. Magn Reson Med. 2015, 73, 1703-1712.
doi: 10.1002/mrm.v73.5 URL |
| 733. |
Yu, L. P.; Qian, W. W.; Yin, G. Y.; Ren, Y. X.; Hu, Z. Y. MRI assessment of lumbar intervertebral disc degeneration with lumbar degenerative disease using the Pfirrmann grading systems. PLoS One. 2012, 7, e48074.
doi: 10.1371/journal.pone.0048074 URL |
| 734. |
Waldenberg, C.; Hebelka, H.; Brisby, H.; Lagerstrand, K. M. MRI histogram analysis enables objective and continuous classification of intervertebral disc degeneration. Eur Spine J. 2018, 27, 1042-1048.
doi: 10.1007/s00586-017-5264-7 URL |
| 735. | Hutton, W. C.; Malko, J. A.; Fajman, W. A. Lumbar disc volume measured by MRI: effects of bed rest, horizontal exercise, and vertical loading. Aviat Space Environ Med. 2003, 74, 73-78. |
| 736. | Malko, J. A.; Hutton, W. C.; Giacometti, A. R.; Kater, G.; Greenfield, P.; Boden, S. D.; Silcox, D. H. Do diurnal changes in loading affect the interpretation of MRI scans of the lumbar spine? J Spinal Disord. 1996, 9, 129-135. |
| 737. |
Roberts, N.; Hogg, D.; Whitehouse, G. H.; Dangerfield, P. Quantitative analysis of diurnal variation in volume and water content of lumbar intervertebral discs. Clin Anat. 1998, 11, 1-8.
doi: 10.1002/(ISSN)1098-2353 URL |
| 738. |
Che, Y. J.; Guo, J. B.; Liang, T.; Chen, X.; Zhang, W.; Yang, H. L.; Luo, Z. P. Assessment of changes in the micro-nano environment of intervertebral disc degeneration based on Pfirrmann grade. Spine J. 2019, 19, 1242-1253.
doi: 10.1016/j.spinee.2019.01.008 URL |
| 739. |
Jensen, T. S.; Albert, H. B.; Soerensen, J. S.; Manniche, C.; Leboeuf-Yde, C. Natural course of disc morphology in patients with sciatica: an MRI study using a standardized qualitative classification system. Spine (Phila Pa 1976). 2006, 31, 1605-1612; discussion 1613.
doi: 10.1097/01.brs.0000221992.77779.37 URL |
| 740. |
Boden, S. D.; Davis, D. O.; Dina, T. S.; Patronas, N. J.; Wiesel, S. W. Abnormal magnetic-resonance scans of the lumbar spine in asymptomatic subjects. A prospective investigation. J Bone Joint Surg Am. 1990, 72, 403-408.
doi: 10.2106/00004623-199072030-00013 URL |
| 741. |
Jensen, M. C.; Brant-Zawadzki, M. N.; Obuchowski, N.; Modic, M. T.; Malkasian, D.; Ross, J. S. Magnetic resonance imaging of the lumbar spine in people without back pain. N Engl J Med. 1994, 331, 69-73.
doi: 10.1056/NEJM199407143310201 URL |
| 742. |
Chavhan, G. B.; Babyn, P. S.; Thomas, B.; Shroff, M. M.; Haacke, E. M. Principles, techniques, and applications of T2*-based MR imaging and its special applications. Radiographics. 2009, 29, 1433-1449.
doi: 10.1148/rg.295095034 URL |
| 743. | Foltz, M. H.; Kage, C. C.; Johnson, C. P.; Ellingson, A. M. Noninvasive assessment of biochemical and mechanical properties of lumbar discs through quantitative magnetic resonance imaging in asymptomatic volunteers. J Biomech Eng. 2017, 139, 1110021-1110027. |
| 744. |
Samartzis, D.; Borthakur, A.; Belfer, I.; Bow, C.; Lotz, J. C.; Wang, H. Q.; Cheung, K. M.; Carragee, E.; Karppinen, J. Novel diagnostic and prognostic methods for disc degeneration and low back pain. Spine J. 2015, 15, 1919-1932.
doi: 10.1016/j.spinee.2014.09.010 URL |
| 745. |
Farshad-Amacker, N. A.; Farshad, M.; Winklehner, A.; Andreisek, G. MR imaging of degenerative disc disease. Eur J Radiol. 2015, 84, 1768-1776.
doi: 10.1016/j.ejrad.2015.04.002 URL |
| 746. |
Nguyen, A. M.; Johannessen, W.; Yoder, J. H.; Wheaton, A. J.; Vresilovic, E. J.; Borthakur, A.; Elliott, D. M. Noninvasive quantification of human nucleus pulposus pressure with use of T1rho-weighted magnetic resonance imaging. J Bone Joint Surg Am. 2008, 90, 796-802.
doi: 10.2106/JBJS.G.00667 URL |
| 747. |
Rong, R.; Zhang, C. Y.; Wang, X. Y. Normal appearance of large field diffusion weighted imaging on 3.0T MRI Chin Med Sci J. 2008, 23, 158-161.
doi: 10.1016/S1001-9294(09)60031-6 URL |
| 748. |
Ludescher, B.; Effelsberg, J.; Martirosian, P.; Steidle, G.; Markert, B.; Claussen, C.; Schick, F. T2- and diffusion-maps reveal diurnal changes of intervertebral disc composition: an in vivo MRI study at 1.5 Tesla. J Magn Reson Imaging. 2008, 28, 252-257.
doi: 10.1002/jmri.v28:1 URL |
| 749. |
Arpinar, V. E.; Rand, S. D.; Klein, A. P.; Maiman, D. J.; Muftuler, L. T. Changes in perfusion and diffusion in the endplate regions of degenerating intervertebral discs: a DCE-MRI study. Eur Spine J. 2015, 24, 2458-2467.
doi: 10.1007/s00586-015-4172-y URL |
| 750. |
Kealey, S. M.; Aho, T.; Delong, D.; Barboriak, D. P.; Provenzale, J. M.; Eastwood, J. D. Assessment of apparent diffusion coefficient in normal and degenerated intervertebral lumbar disks: initial experience. Radiology. 2005, 235, 569-574.
doi: 10.1148/radiol.2352040437 URL |
| 751. | Li, L. Y.; Wu, X. L.; Roman, R. J.; Fan, F.; Qiu, C. S.; Chen, B. H. Diffusion-weighted 7.0T magnetic resonance imaging in assessment of intervertebral disc degeneration in rats. Chin Med J (Engl). 2018, 131, 63-68. |
| 752. |
Paul, C. P. L.; Smit, T. H.; de Graaf, M.; Holewijn, R. M.; Bisschop, A.; van de Ven, P. M.; Mullender, M. G.; Helder, M. N.; Strijkers, G. J. Quantitative MRI in early intervertebral disc degeneration: T1rho correlates better than T2 and ADC with biomechanics, histology and matrix content. PLoS One. 2018, 13, e0191442.
doi: 10.1371/journal.pone.0191442 URL |
| 753. |
Ward, K. M.; Balaban, R. S. Determination of pH using water protons and chemical exchange dependent saturation transfer (CEST). Magn Reson Med. 2000, 44, 799-802.
doi: 10.1002/(ISSN)1522-2594 URL |
| 754. |
Pulickal, T.; Boos, J.; Konieczny, M.; Sawicki, L. M.; Müller-Lutz, A.; Bittersohl, B.; Gerß, J.; Eichner, M.; Wittsack, H. J.; Antoch, G.; Schleich, C. MRI identifies biochemical alterations of intervertebral discs in patients with low back pain and radiculopathy. Eur Radiol. 2019, 29, 6443-6446.
doi: 10.1007/s00330-019-06305-6 URL |
| 755. |
Hang, D.; Li, F.; Che, W.; Wu, X.; Wan, Y.; Wang, J.; Zheng, Y. One-stage positron emission tomography and magnetic resonance imaging to assess mesenchymal stem cell survival in a canine model of intervertebral disc degeneration. Stem Cells Dev. 2017, 26, 1334-1343.
doi: 10.1089/scd.2017.0103 URL |
| 756. | Saldanha, K. J.; Piper, S. L.; Ainslie, K. M.; Kim, H. T.; Majumdar, S. Magnetic resonance imaging of iron oxide labelled stem cells: applications to tissue engineering based regeneration of the intervertebral disc. Eur Cell Mater. 2008, 16, 17-25. |
| 757. | Lalande, C.; Miraux, S.; Derkaoui, S. M.; Mornet, S.; Bareille, R.; Fricain, J. C.; Franconi, J. M.; Le Visage, C.; Letourneur, D.; Amédée, J.; Bouzier-Sore, A. K. Magnetic resonance imaging tracking of human adipose derived stromal cells within three-dimensional scaffolds for bone tissue engineering. Eur Cell Mater. 2011, 21, 341-354. |
| 758. |
Martin, J. T.; Milby, A. H.; Ikuta, K.; Poudel, S.; Pfeifer, C. G.; Elliott, D. M.; Smith, H. E.; Mauck, R. L. A radiopaque electrospun scaffold for engineering fibrous musculoskeletal tissues: Scaffold characterization and in vivo applications. Acta Biomater. 2015, 26, 97-104.
doi: 10.1016/j.actbio.2015.08.001 URL |
| 759. |
Gullbrand, S. E.; Schaer, T. P.; Agarwal, P.; Bendigo, J. R.; Dodge, G. R.; Chen, W.; Elliott, D. M.; Mauck, R. L.; Malhotra, N. R.; Smith, L. J. Translation of an injectable triple-interpenetrating-network hydrogel for intervertebral disc regeneration in a goat model. Acta Biomater. 2017, 60, 201-209.
doi: 10.1016/j.actbio.2017.07.025 URL |
| 760. | Nomura, T.; Mochida, J.; Okuma, M.; Nishimura, K.; Sakabe, K. Nucleus pulposus allograft retards intervertebral disc degeneration. Clin Orthop Relat Res. 2001, 94-101. |
| 761. |
Nishimura, K.; Mochida, J. Percutaneous reinsertion of the nucleus pulposus. An experimental study. Spine (Phila Pa 1976). 1998, 23, 1531-1538; discussion 1539.
doi: 10.1097/00007632-199807150-00006 URL |
| 762. |
Miyazaki, S.; Diwan, A. D.; Kato, K.; Cheng, K.; Bae, W. C.; Sun, Y.; Yamada, J.; Muehleman, C.; Lenz, M. E.; Inoue, N.; Sah, R. L.; Kawakami, M.; Masuda, K. ISSLS PRIZE IN BASIC SCIENCE 2018: Growth differentiation factor-6 attenuated pro-inflammatory molecular changes in the rabbit anular-puncture model and degenerated disc-induced pain generation in the rat xenograft radiculopathy model. Eur Spine J. 2018, 27, 739-751.
doi: 10.1007/s00586-018-5488-1 URL |
| 763. | Qian, J.; Ge, J.; Yan, Q.; Wu, C.; Yang, H.; Zou, J. Selection of the Optimal Puncture Needle for Induction of a Rat Intervertebral Disc Degeneration Model. Pain Physician. 2019, 22, 353-360. |
| 764. | Chen, Z.; Zhang, W.; Deng, M.; Li, Y.; Zhou, Y. CircGLCE alleviates intervertebral disc degeneration by regulating apoptosis and matrix degradation through the targeting of miR-587/STAP1. Aging (Albany NY). 2020, 12, 21971-21991. |
| 765. |
Wang, D.; He, X.; Wang, D.; Peng, P.; Xu, X.; Gao, B.; Zheng, C.; Wang, H.; Jia, H.; Shang, Q.; Sun, Z.; Luo, Z.; Yang, L. Quercetin suppresses apoptosis and attenuates intervertebral disc degeneration via the SIRT1-autophagy pathway. Front Cell Dev Biol. 2020, 8, 613006.
doi: 10.3389/fcell.2020.613006 URL |
| 766. |
Huang, J. F.; Zheng, X. Q.; Lin, J. L.; Zhang, K.; Tian, H. J.; Zhou, W. X.; Wang, H.; Gao, Z.; Jin, H. M.; Wu, A. M. Sinapic acid inhibits IL-1β-induced apoptosis and catabolism in nucleus pulposus cells and ameliorates intervertebral disk degeneration. J Inflamm Res. 2020, 13, 883-895.
doi: 10.2147/JIR.S278556 URL |
| 767. |
Thompson, J. P.; Pearce, R. H.; Schechter, M. T.; Adams, M. E.; Tsang, I. K.; Bishop, P. B. Preliminary evaluation of a scheme for grading the gross morphology of the human intervertebral disc. Spine (Phila Pa 1976). 1990, 15, 411-415.
doi: 10.1097/00007632-199005000-00012 URL |
| 768. |
Boos, N.; Weissbach, S.; Rohrbach, H.; Weiler, C.; Spratt, K. F.; Nerlich, A. G. Classification of age-related changes in lumbar intervertebral discs: 2002 Volvo Award in basic science. Spine (Phila Pa 1976). 2002, 27, 2631-2644.
doi: 10.1097/00007632-200212010-00002 URL |
| 769. | Fusellier, M.; Colombier, P.; Lesoeur, J.; Youl, S.; Madec, S.; Gauthier, O.; Hamel, O.; Guicheux, J.; Clouet, J. Longitudinal comparison of enzyme- and laser-treated intervertebral disc by MRI, X-ray, and histological analyses reveals discrepancies in the progression of disc degeneration: a rabbit study. Biomed Res Int. 2016, 2016, 5498271. |
| 770. |
Phillips, K. L.; Jordan-Mahy, N.; Nicklin, M. J.; Le Maitre, C. L. Interleukin-1 receptor antagonist deficient mice provide insights into pathogenesis of human intervertebral disc degeneration. Ann Rheum Dis. 2013, 72, 1860-1867.
doi: 10.1136/annrheumdis-2012-202266 URL |
| 771. |
Groh, A. M. R.; Fournier, D. E.; Battié, M. C.; Séguin, C. A. Innervation of the human intervertebral disc: a scoping review. Pain Med. 2021, 22, 1281-1304.
doi: 10.1093/pm/pnab070 URL |
| 772. |
Liu, C.; Jin, Z.; Ge, X.; Zhang, Y.; Xu, H. Decellularized annulus fibrosus matrix/chitosan hybrid hydrogels with basic fibroblast growth factor for annulus fibrosus tissue engineering. Tissue Eng Part A. 2019, 25, 1605-1613.
doi: 10.1089/ten.tea.2018.0297 URL |
| 773. | ASTM F2258-05(2015). Standard Test Method for Strength Properties of Tissue Adhesives in Tension. ASTM International: West Conshohocken, PA, 2015. |
| 774. | ASTM F2255-05(2015). Standard Test Method for Strength Properties of Tissue Adhesives in Lap-Shear by Tension Loading. ASTM International: West Conshohocken, PA, 2015. |
| 775. | ASTM F2458-05(2015). Standard Test Method for Wound Closure Strength of Tissue Adhesives and Sealants. ASTM International: West Conshohocken, PA, 2015. |
| 776. | ASTM F2392-04(2015). Standard Test Method for Burst Strength of Surgical Sealants. ASTM International: Conshohocken, PA, 2015. |
| 777. |
Schmitz, T. C.; Salzer, E.; Crispim, J. F.; Fabra, G. T.; LeVisage, C.; Pandit, A.; Tryfonidou, M.; Maitre, C. L.; Ito, K. Characterization of biomaterials intended for use in the nucleus pulposus of degenerated intervertebral discs. Acta Biomater. 2020, 114, 1-15.
doi: 10.1016/j.actbio.2020.08.001 URL |
| 778. |
Thoreson, O.; Ekström, L.; Hansson, H. A.; Todd, C.; Witwit, W.; Swärd Aminoff, A.; Jonasson, P.; Baranto, A. The effect of repetitive flexion and extension fatigue loading on the young porcine lumbar spine, a feasibility study of MRI and histological analyses. J Exp Orthop. 2017, 4, 16.
doi: 10.1186/s40634-017-0091-7 URL |
| 779. |
Noguchi, M.; Gooyers, C. E.; Karakolis, T.; Noguchi, K.; Callaghan, J. P. Is intervertebral disc pressure linked to herniation?: An in-vitro study using a porcine model. J Biomech. 2016, 49, 1824-1830.
doi: 10.1016/j.jbiomech.2016.04.018 URL |
| 780. |
Brown, M. D.; Holmes, D. C.; Heiner, A. D. Measurement of cadaver lumbar spine motion segment stiffness. Spine (Phila Pa 1976). 2002, 27, 918-922.
doi: 10.1097/00007632-200205010-00006 URL |
| 781. | Newell, N.; Rivera Tapia, D.; Rahman, T.; Lim, S.; O’Connell, G. D.; Holsgrove, T. P. Influence of testing environment and loading rate on intervertebral disc compressive mechanics: An assessment of repeatability at three different laboratories. JOR Spine. 2020, 3, e21110. |
| 782. |
Shanks, H. R.; Wu, S.; Nguyen, N. T.; Lu, D.; Saunders, B. R. Including fluorescent nanoparticle probes within injectable gels for remote strain measurements and discrimination between compression and tension. Soft Matter. 2021, 17, 1048-1055.
doi: 10.1039/D0SM01635G URL |
| 783. |
Iatridis, J. C.; Mente, P. L.; Stokes, I. A.; Aronsson, D. D.; Alini, M. Compression-induced changes in intervertebral disc properties in a rat tail model. Spine (Phila Pa 1976). 1999, 24, 996-1002.
doi: 10.1097/00007632-199905150-00013 URL |
| 784. |
Adams, M. A.; Roughley, P. J. What is intervertebral disc degeneration, and what causes it? Spine (Phila Pa 1976). 2006, 31, 2151-2161.
doi: 10.1097/01.brs.0000231761.73859.2c URL |
| 785. |
Shao, Z.; Wang, B.; Shi, Y.; Xie, C.; Huang, C.; Chen, B.; Zhang, H.; Zeng, G.; Liang, H.; Wu, Y.; Zhou, Y.; Tian, N.; Wu, A.; Gao, W.; Wang, X.; Zhang, X. Senolytic agent Quercetin ameliorates intervertebral disc degeneration via the Nrf2/NF-κB axis. Osteoarthritis Cartilage. 2021, 29, 413-422.
doi: 10.1016/j.joca.2020.11.006 URL |
| 786. |
Patil, P.; Falabella, M.; Saeed, A.; Lee, D.; Kaufman, B.; Shiva, S.; Croix, C. S.; Van Houten, B.; Niedernhofer, L. J.; Robbins, P. D.; Lee, J.; Gwendolyn, S.; Vo, N. V. Oxidative stress-induced senescence markedly increases disc cell bioenergetics. Mech Ageing Dev. 2019, 180, 97-106.
doi: 10.1016/j.mad.2019.04.006 URL |
| 787. |
Rajasekaran, S.; Tangavel, C.; Aiyer, S. N.; Nayagam, S. M.; Raveendran, M.; Demonte, N. L.; Subbaiah, P.; Kanna, R.; Shetty, A. P.; Dharmalingam, K. ISSLS PRIZE IN CLINICAL SCIENCE 2017: Is infection the possible initiator of disc disease? An insight from proteomic analysis. Eur Spine J. 2017, 26, 1384-1400.
doi: 10.1007/s00586-017-4972-3 URL |
| 788. |
Benzakour, T.; Igoumenou, V.; Mavrogenis, A. F.; Benzakour, A. Current concepts for lumbar disc herniation. Int Orthop. 2019, 43, 841-851.
doi: 10.1007/s00264-018-4247-6 URL |
| 789. |
Martin, J. T.; Kim, D. H.; Milby, A. H.; Pfeifer, C. G.; Smith, L. J.; Elliott, D. M.; Smith, H. E.; Mauck, R. L. In vivo performance of an acellular disc-like angle ply structure (DAPS) for total disc replacement in a small animal model. J Orthop Res. 2017, 35, 23-31.
doi: 10.1002/jor.v35.1 URL |
| 790. | Lim, K. Z.; Daly, C. D.; Ghosh, P.; Jenkin, G.; Oehme, D.; Cooper-White, J.; Naidoo, T.; Goldschlager, T. Ovine lumbar intervertebral disc degeneration model utilizing a lateral retroperitoneal drill bit injury. J Vis Exp. 2017, 55753. |
| 791. | Schek, R. M.; Michalek, A. J.; Iatridis, J. C. Genipin-crosslinked fibrin hydrogels as a potential adhesive to augment intervertebral disc annulus repair. Eur Cell Mater. 2011, 21, 373-383. |
| 792. | Fakhoury, J.; Dowling, T. J. Cervical degenerative disc disease. In StatPearls, StatPearls Publishing: Treasure Island (FL), 2021. |
| 793. |
Ho-Pham, L. T.; Lai, T. Q.; Mai, L. D.; Doan, M. C.; Pham, H. N.; Nguyen, T. V. Prevalence and pattern of radiographic intervertebral disc degeneration in Vietnamese: a population-based study. Calcif Tissue Int. 2015, 96, 510-517.
doi: 10.1007/s00223-015-9986-4 URL |
| 794. |
Zou, F.; Yang, S.; Jiang, J.; Lu, F.; Xia, X.; Ma, X. Adjacent intervertebral disk height decrease phenomenon after single-level transforaminal lumbar interbody fusion of the lumbar spine. World Neurosurg. 2019, 128, e308-e314.
doi: 10.1016/j.wneu.2019.04.141 URL |
| 795. |
Hei, L.; Ge, Z.; Yuan, W.; Suo, L.; Suo, Z.; Lin, L.; Ding, H.; Qiu, Y. Evaluation of a rabbit model of adjacent intervertebral disc degeneration after fixation and fusion and maintenance in an upright feeding cage. Neurol Res. 2021, 43, 447-457.
doi: 10.1080/01616412.2020.1866804 URL |
| 796. |
Liu, Z.; Zheng, Z.; Qi, J.; Wang, J.; Zhou, Q.; Hu, F.; Liang, J.; Li, C.; Zhang, W.; Zhang, X. CD24 identifies nucleus pulposus progenitors/notochordal cells for disc regeneration. J Biol Eng. 2018, 12, 35.
doi: 10.1186/s13036-018-0129-0 URL |
| 797. |
Yang, S. H.; Hu, M. H.; Wu, C. C.; Chen, C. W.; Sun, Y. H.; Yang, K. C. CD24 expression indicates healthier phenotype and less tendency of cellular senescence in human nucleus pulposus cells. Artif Cells Nanomed Biotechnol. 2019, 47, 3021-3028.
doi: 10.1080/21691401.2019.1642205 URL |
| 798. | Xunlu, Y.; Minshan, F.; Liguo, Z.; Jiawen, Z.; Xu, W.; Jie, Y.; Shangquan, W.; He, Y.; Long, L.; Tao, H.; Xuepeng, L. Integrative bioinformatics analysis reveals potential gene biomarkers and analysis of function in human degenerative disc annulus fibrosus cells. Biomed Res Int. 2019, 2019, 9890279. |
| 799. |
Rutges, J.; Creemers, L. B.; Dhert, W.; Milz, S.; Sakai, D.; Mochida, J.; Alini, M.; Grad, S. Variations in gene and protein expression in human nucleus pulposus in comparison with annulus fibrosus and cartilage cells: potential associations with aging and degeneration. Osteoarthritis Cartilage. 2010, 18, 416-423.
doi: 10.1016/j.joca.2009.09.009 URL |
| [1] | Shuqin Cao, Quan Yuan. An update of nanotopographical surfaces in modulating stem cell fate: a narrative review [J]. Biomaterials Translational, 2022, 3(1): 55-64. |
| [2] | Emma Steijvers, Armaan Ghei, Zhidao Xia. Manufacturing artificial bone allografts: a perspective [J]. Biomaterials Translational, 2022, 3(1): 65-80. |
| [3] | Ke Hu, Yuxuan Li, Zunxiang Ke, Hongjun Yang, Chanjun Lu, Yiqing Li, Yi Guo, Weici Wang. History, progress and future challenges of artificial blood vessels: a narrative review [J]. Biomaterials Translational, 2022, 3(1): 81-98. |
| [4] | Yizhong Peng, Jinye Li, Hui Lin, Shuo Tian, Sheng Liu, Feifei Pu, Lei Zhao, Kaige Ma, Xiangcheng Qing, Zengwu Shao. Endogenous repair theory enriches construction strategies for orthopaedic biomaterials: a narrative review [J]. Biomaterials Translational, 2021, 2(4): 343-360. |
| [5] | Ge Yan, Yuqi Liu, Minghui Xie, Jiawei Shi, Weihua Qiao, Nianguo Dong. Experimental and computational models for tissue-engineered heart valves: a narrative review [J]. Biomaterials Translational, 2021, 2(4): 361-375. |
| [6] | Xirui Jing, Qiuyue Ding, Qinxue Wu, Weijie Su, Keda Yu, Yanlin Su, Bing Ye, Qing Gao, Tingfang Sun, Xiaodong Guo. Magnesium-based materials in orthopaedics: material properties and animal models [J]. Biomaterials Translational, 2021, 2(3): 197-213. |
| [7] | Kamolrat Metavarayuth, Esteban Villarreal, Hui Wang, Qian Wang. Surface topography and free energy regulate osteogenesis of stem cells: effects of shape-controlled gold nanoparticles [J]. Biomaterials Translational, 2021, 2(2): 165-173. |
| [8] | Pingli Wu, Yangyang Liang, Guoming Sun. Engineering immune-responsive biomaterials for skin regeneration [J]. Biomaterials Translational, 2021, 2(1): 61-71. |
| [9] | Isak Jatoi, Jingyu Fan. A biomaterials viewpoint for the 2020 SARS-CoV-2 vaccine development [J]. Biomaterials Translational, 2021, 2(1): 30-42. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||