Biomaterials Translational ›› 2024, Vol. 5 ›› Issue (1): 59-68.doi: 10.12336/biomatertransl.2024.01.006
• RESEARCH ARTICLES • Previous Articles Next Articles
Huaxin Yang1, Mengjia Zheng1, Yuyue Zhang1, Chaochang Li2, Joseph Ho Chi Lai1, Qizheng Zhang1, Kannie WY Chan1, Hao Wang2, Xin Zhao3, Zijiang Yang2,*( ), Chenjie Xu1,*(
), Chenjie Xu1,*( )
)
Received:2023-12-11
Revised:2024-01-20
Accepted:2024-03-08
Online:2024-03-28
Published:2024-03-28
Contact:
Chenjie Xu, 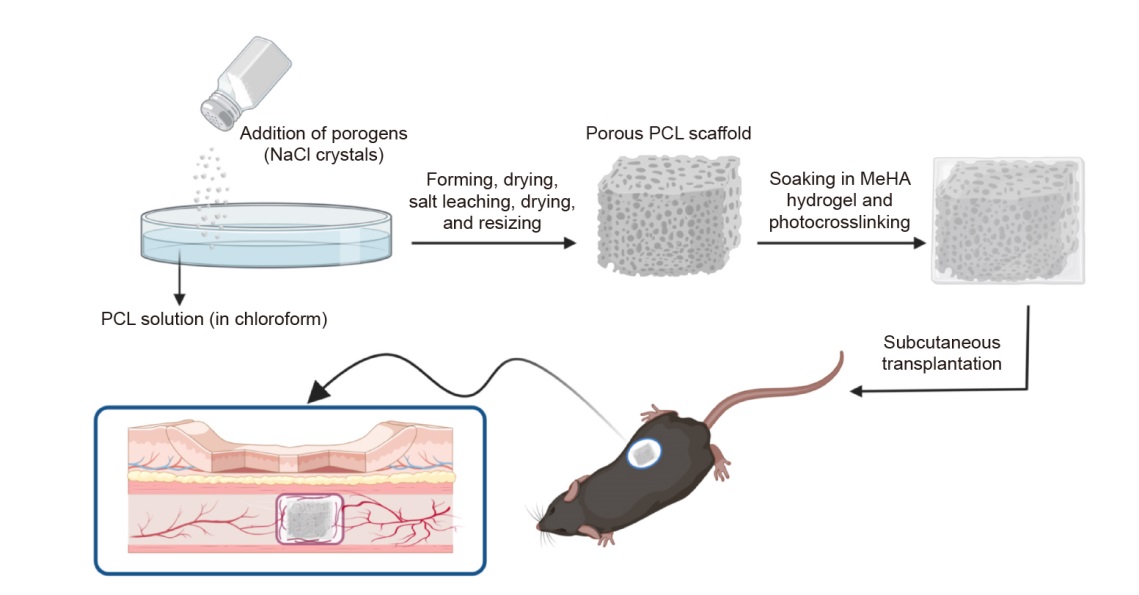
Figure 1. Fabrication and subcutaneous transplantation of PCL/MeHA composite scaffolds. Created with BioRender.com. MeHA: methacrylated hyaluronic acid; NaCl: sodium chloride; PCL: poly(ε-caprolactone).
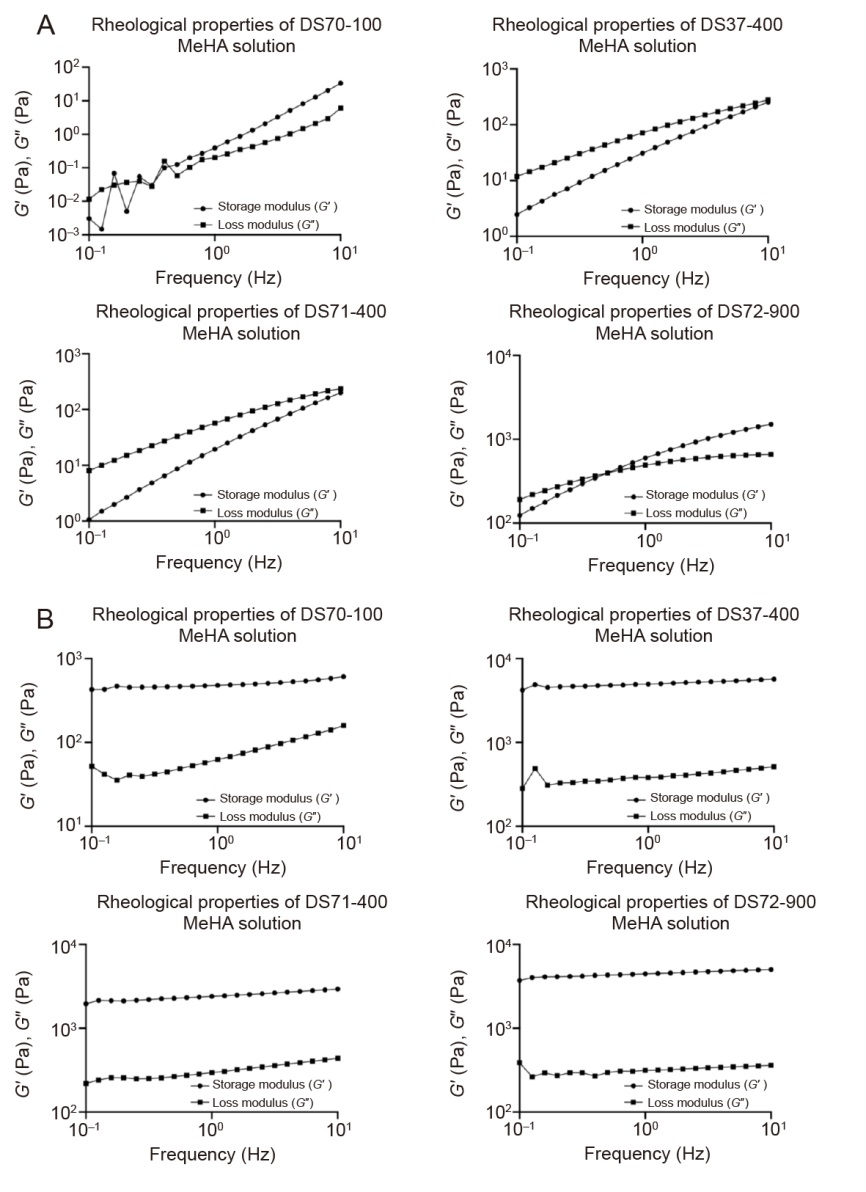
Figure 2. Characterisation of rheological properties. (A, B) Storage modulus (G′) and loss modulus (G″) of MeHA solutions (5% (w/w); A) and derived MeHA hydrogels (B). Data corresponded to one experiment. DS: degree of substitution; MeHA: methacrylated hyaluronic acid.
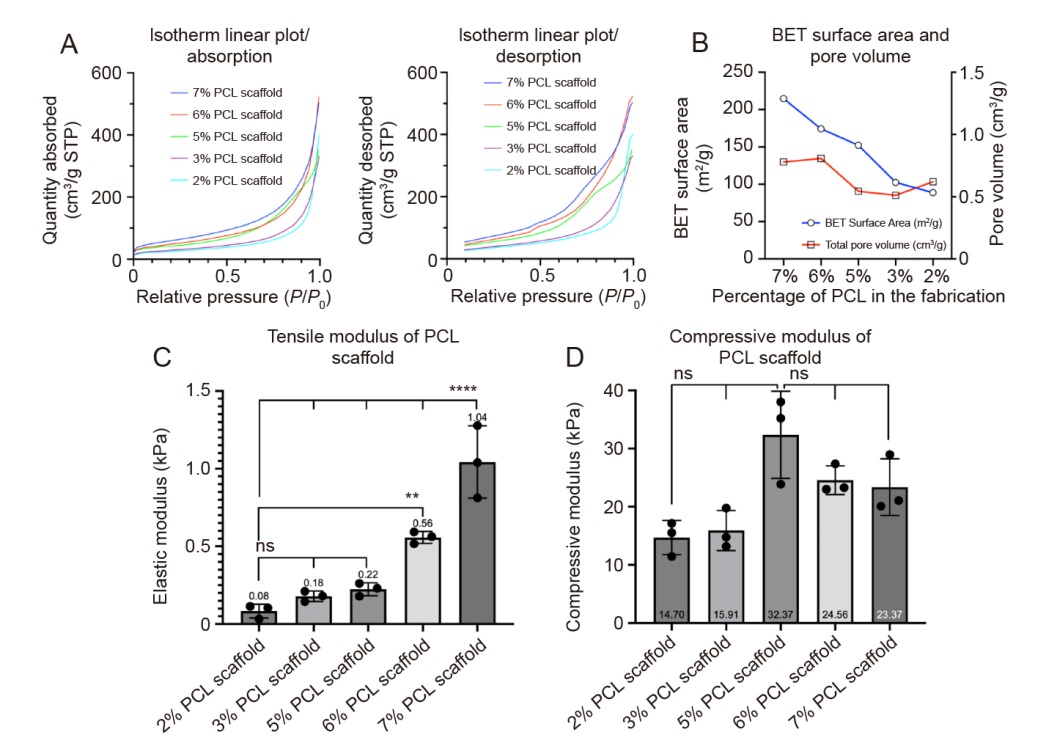
Figure 3. Characterisation of PCL scaffolds. (A) Isotherm linear plot of absorption and desorption of PCL scaffolds with different ratios of PCL and NaCl (number of samples per scaffold, n = 5). (B) BET surface area and pore volume of PCL scaffolds with different ratios of PCL and NaCl (n = 5). (C) Tensile moduli of PCL scaffolds with different ratios of PCL and NaCl (n = 3; three measurements were obtained per scaffold). (D) Compressive moduli of PCL scaffolds with different ratios of PCL and NaCl (n = 3; three measurements were obtained per scaffold). Data are expressed as mean ± SD. **P < 0.01, ****P < 0.0001 (one-way analysis of variance followed by Dunnett’s multiple comparison test). BET: Brunauer-Emmett-Teller; NaCl: sodium chloride; ns: not significant; P: the equilibrium adsorption pressure of the gas; P0: the saturated vapour pressure of the gas at the adsorption temperature; PCL: poly(ε-caprolactone).
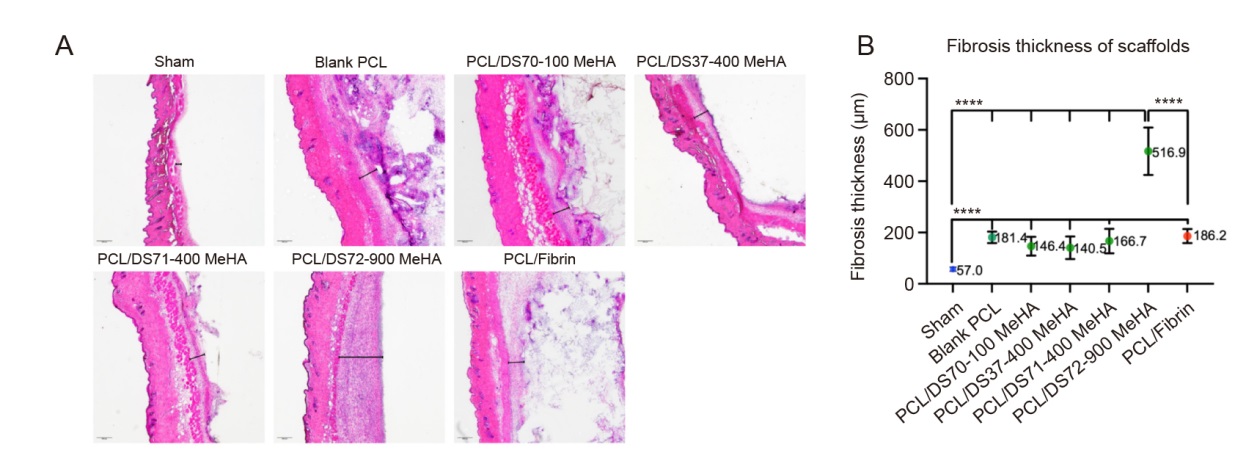
Figure 4. Histological evaluation of scaffold sections after four-week implantation in mice. (A) H&E staining results: interaction of scaffolds with surrounding tissues (microscopy images at 4× magnification; the black line segment indicates the fibrotic capsule). (B) Fibrosis thickness of different scaffolds based on images obtained at 4× magnification (samples per implant, n = 3, with five measurements obtained per implant). Data are expressed as mean ± SD. *P < 0.05, ****P < 0.0001 (one-way analysis of variance followed by Dunnett’s multiple comparison test). DS: degree of substitution; H&E: haematoxylin & eosin; MeHA: methacrylated hyaluronic acid; ns: not significant; PCL: poly(ε-caprolactone).
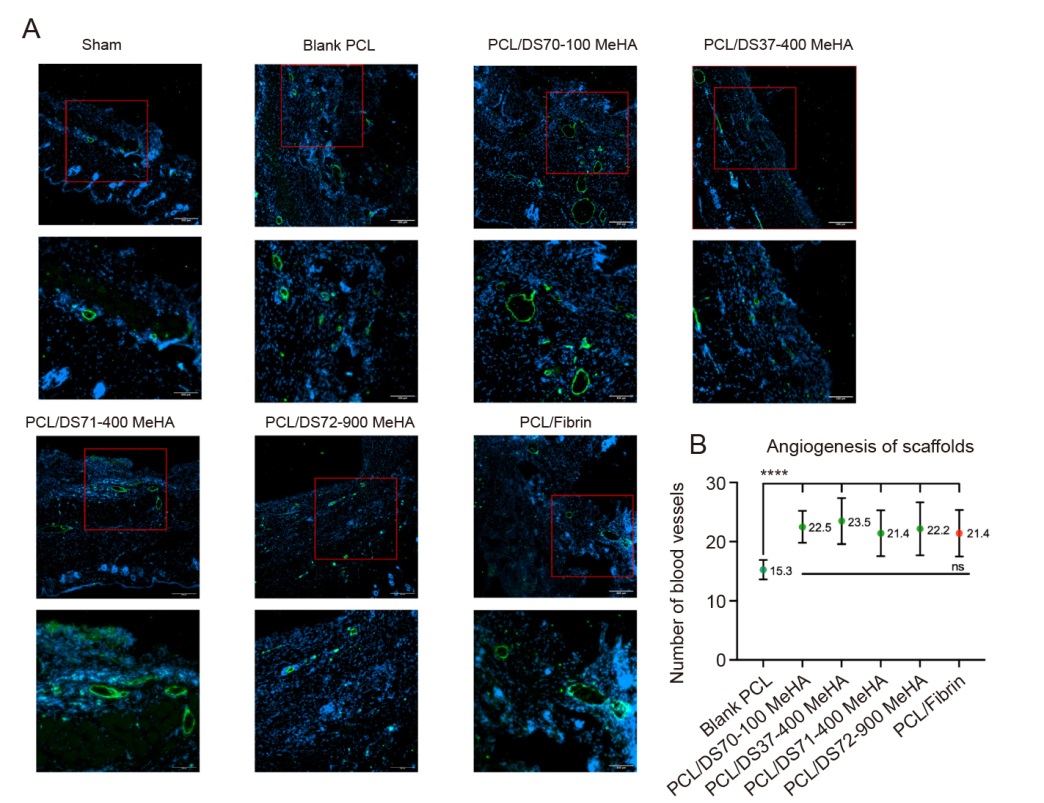
Figure 5. vWF staining results. (A) New blood vessels (microscopy images obtained at magnification of 10× (upper) and 20× (lower)). The square indicates the area chosen for amplification. All of the PCL/MeHA composite scaffolds (regardless of molecular weight and DS) successfully induced angiogenesis similar to the PCL/fibrin scaffolds. The PCL scaffold effectively induced the vascular regeneration. (B) Quantitative analysis of angiogenesis: number of blood vessels associated with different scaffolds based on 10× magnified images (samples per implant, n = 3, with five measurements obtained per implant). Data are expressed as mean ± SD. ****P < 0.0001 (one-way analysis of variance followed by Dunnett’s multiple comparison test). DS: degree of substitution; MeHA: methacrylated hyaluronic acid; ns: not significant; PCL: poly(ε-caprolactone); vWF: von Willebrand factor.
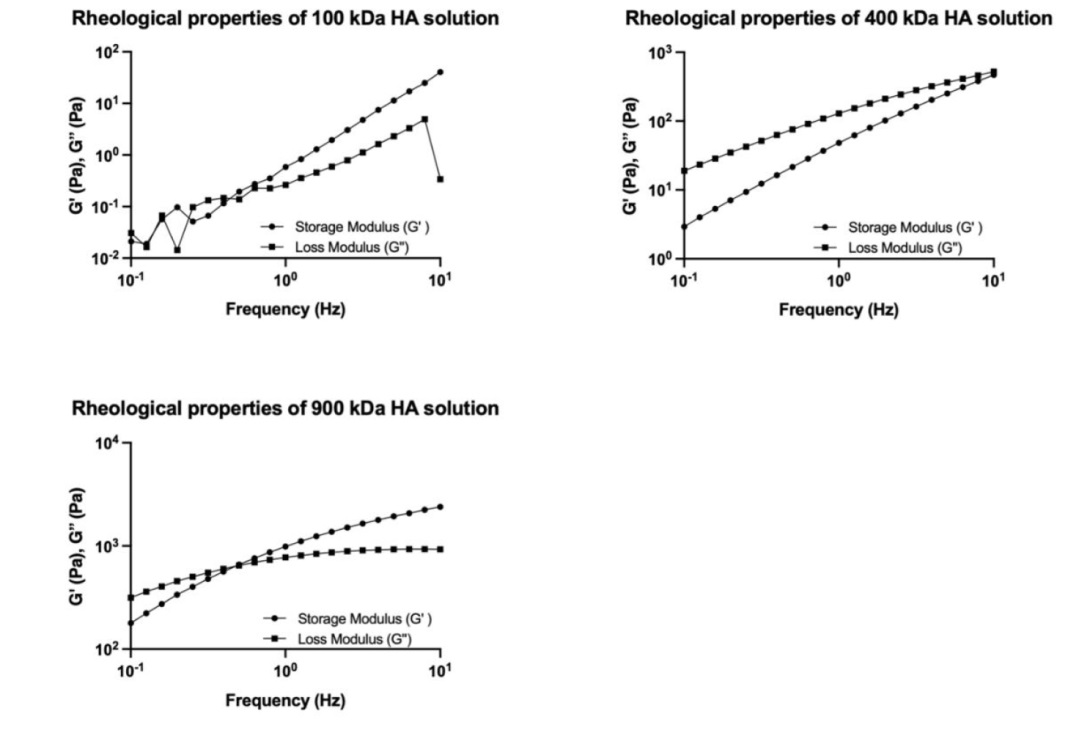
Additional Figure 2. Storage modulus (G′) and loss modulus (G″) of unmodified HA solutions (5% (w/w)). Data corresponded to one experiment. HA: hyaluronic acid.
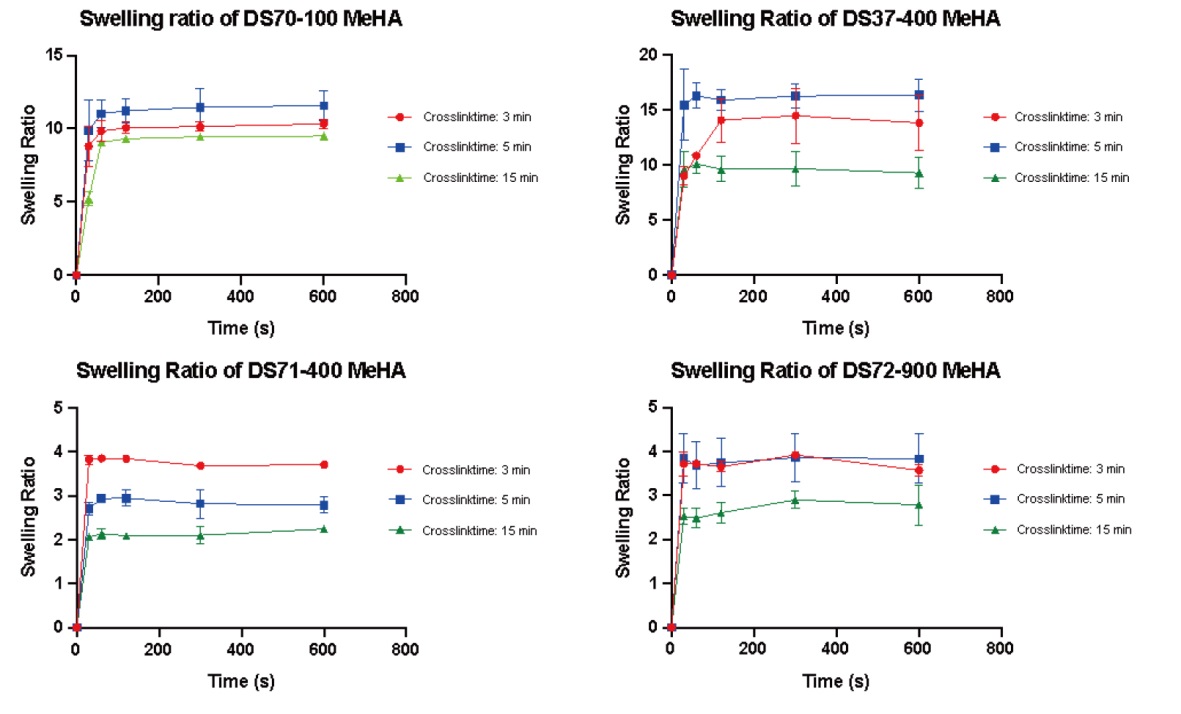
Additional Figure 3. The swelling behaviors of crosslinked MeHA polymers in PBS. MeHA: methacrylated hyaluronic acid; PBS: phosphate-buffered saline. Data are expressed as mean ± SD.
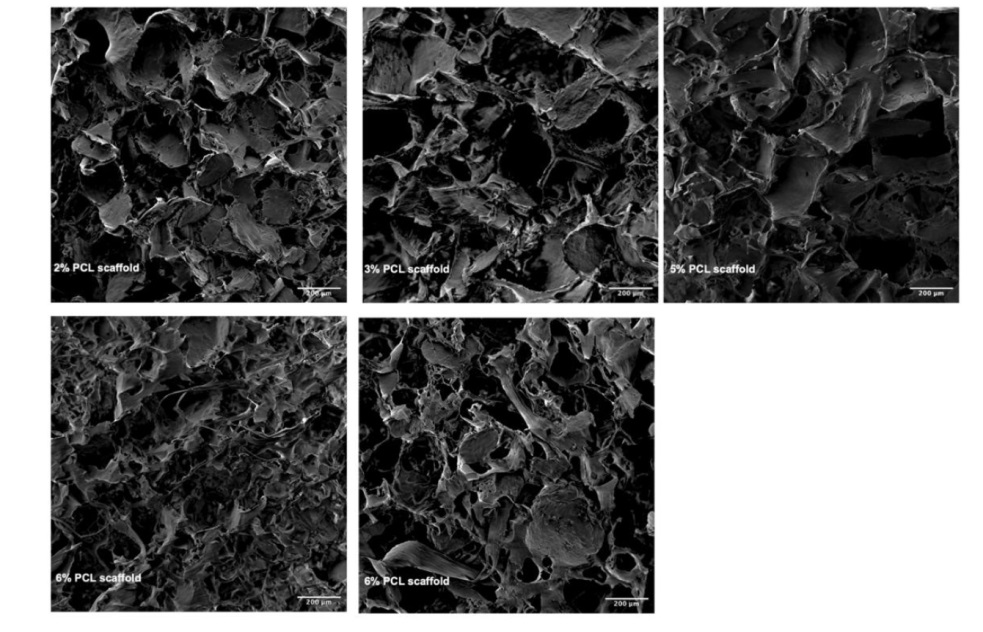
Additional Figure 4. SEM images of the porous PCL scaffolds prepared by using different PCL/NaCl composition. The pores of all scaffolds exhibited non-uniform sizes and shapes. Scale bars: 200 μm. NaCl: sodium chloride; PCL: poly(ε-caprolactone); SEM: scanning electron microscopy.
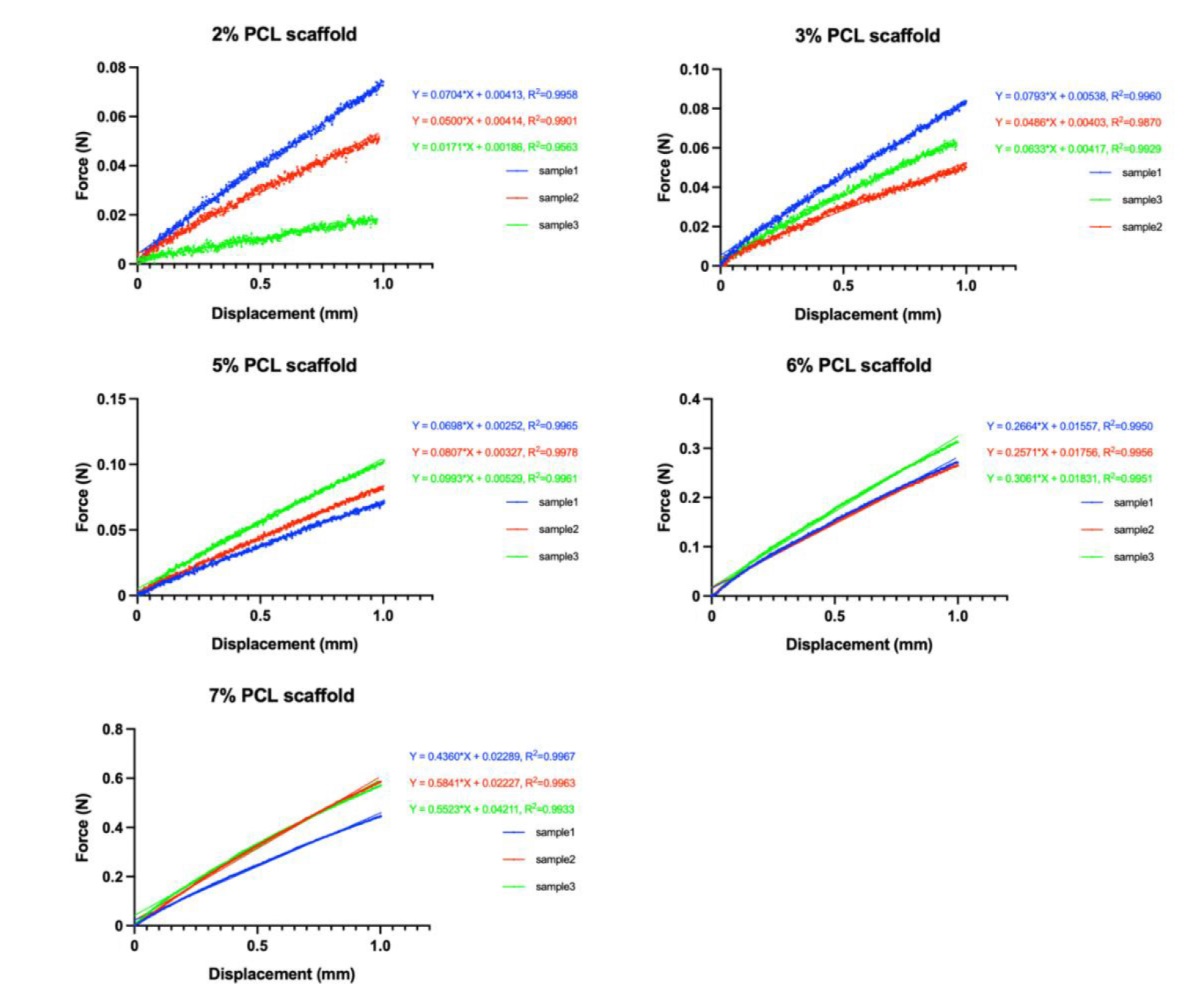
Additional Figure 5. Raw data from tensile elastic modulus tests of PCL scaffolds. The curve fitting is calculated by simple linear regression. PCL: poly(ε-caprolactone).
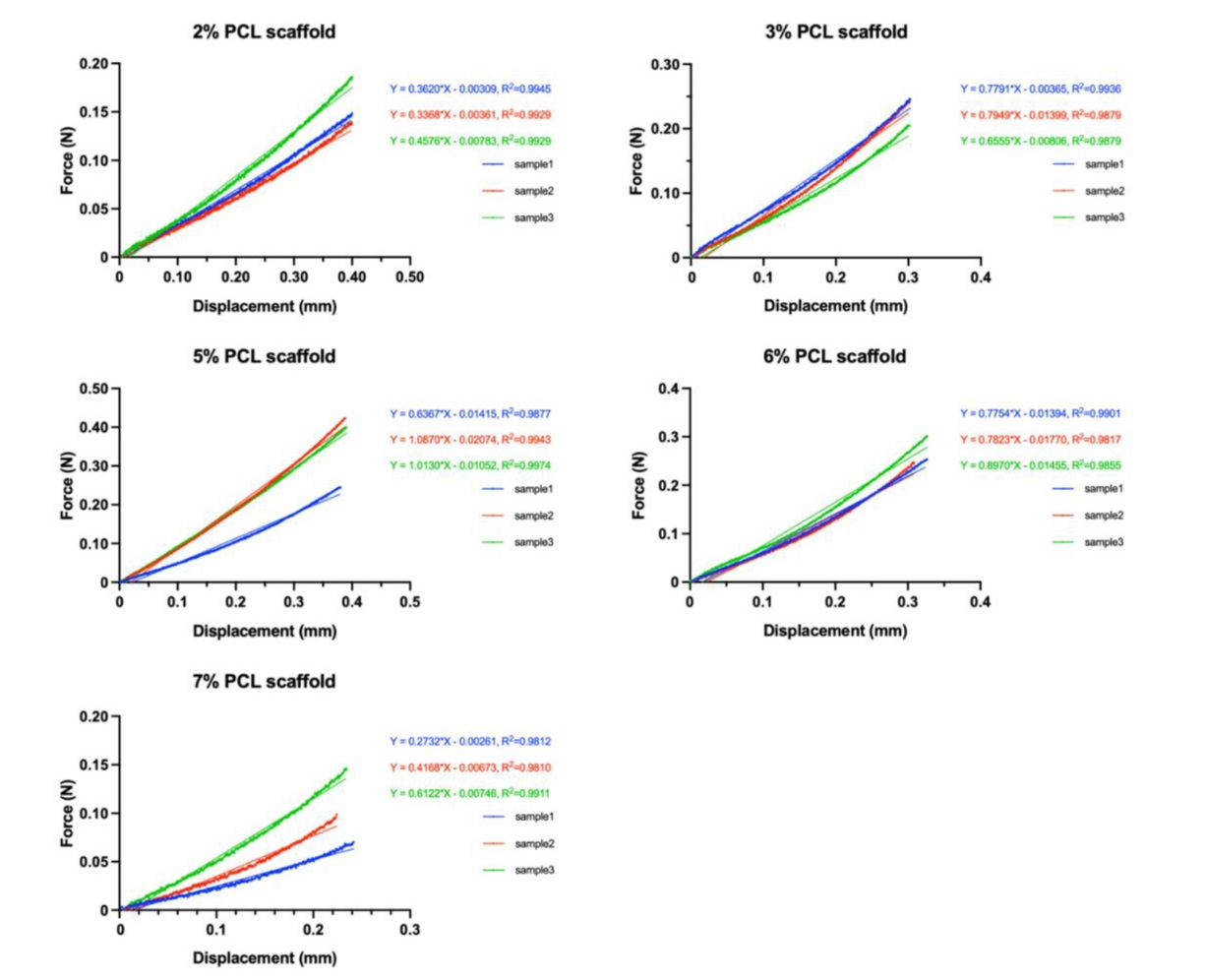
Additional Figure 6. Raw data from compressive elastic modulus tests of PCL scaffolds. The curve fitting is calculated by simple linear regression. PCL: poly(ε-caprolactone).
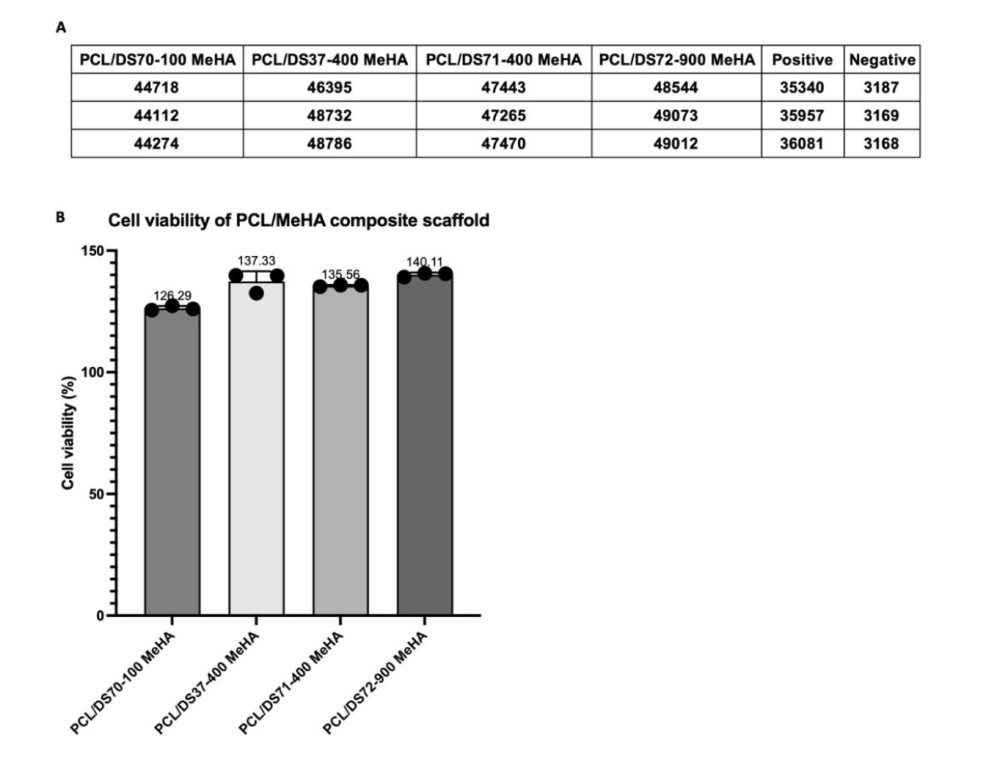
Additional Figure 7. Cell viability indicated the biocompatibility of PCL/MeHA composite scaffold. (A) Raw data of the value obtained from microplate reader. (B) Calculated cell viability of MSC cultured with scaffolds, compared with positive group. Data are expressed as mean ± SD. MeHA: methacrylated hyaluronic acid; MSC: mesenchymal stem cell; PCL: poly(ε-caprolactone).
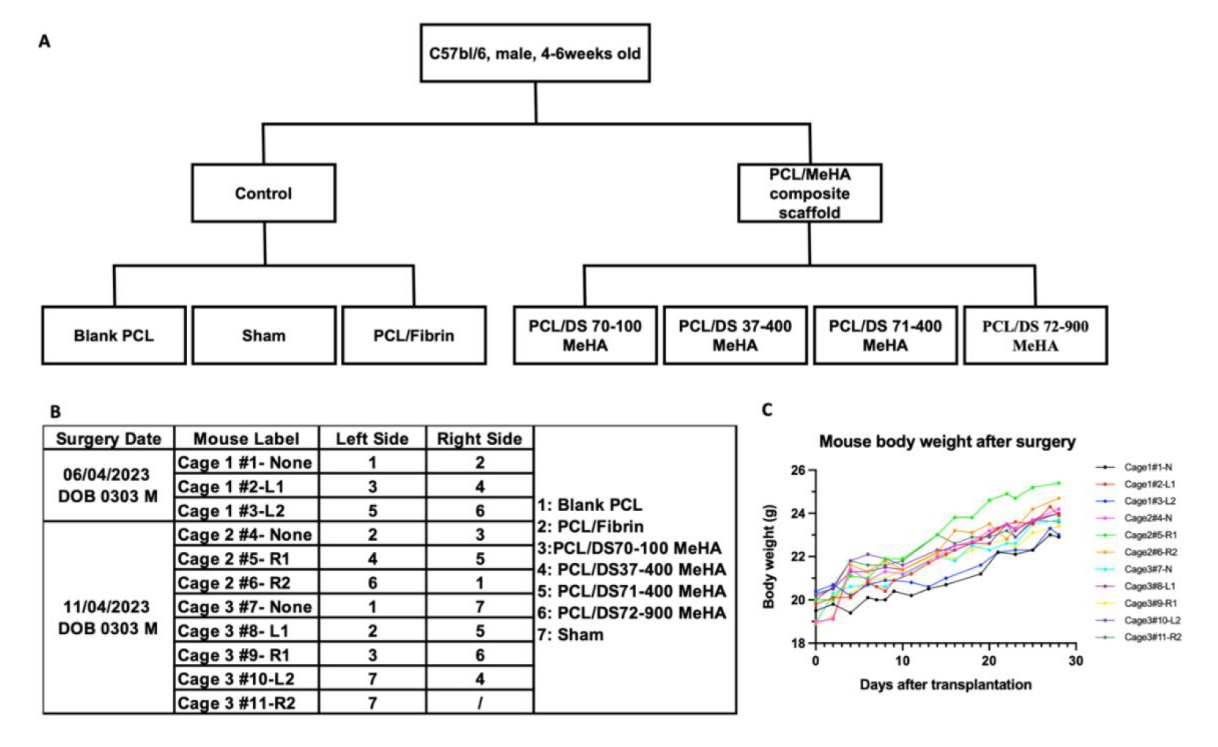
Additional Figure 8. The in vivo experiment design and continuous mice body weight record. (A) Flowchart of animal experiment design. (B) Detailed mice label, transplantation site and the type of the implanted scaffold. (C) Continuous body weight record of mice and the trend during the experiment. All mice were healthy, and their weight were stable. DS: degree of substitution; MeHA: methacrylated hyaluronic acid; PCL: poly(ε-caprolactone).
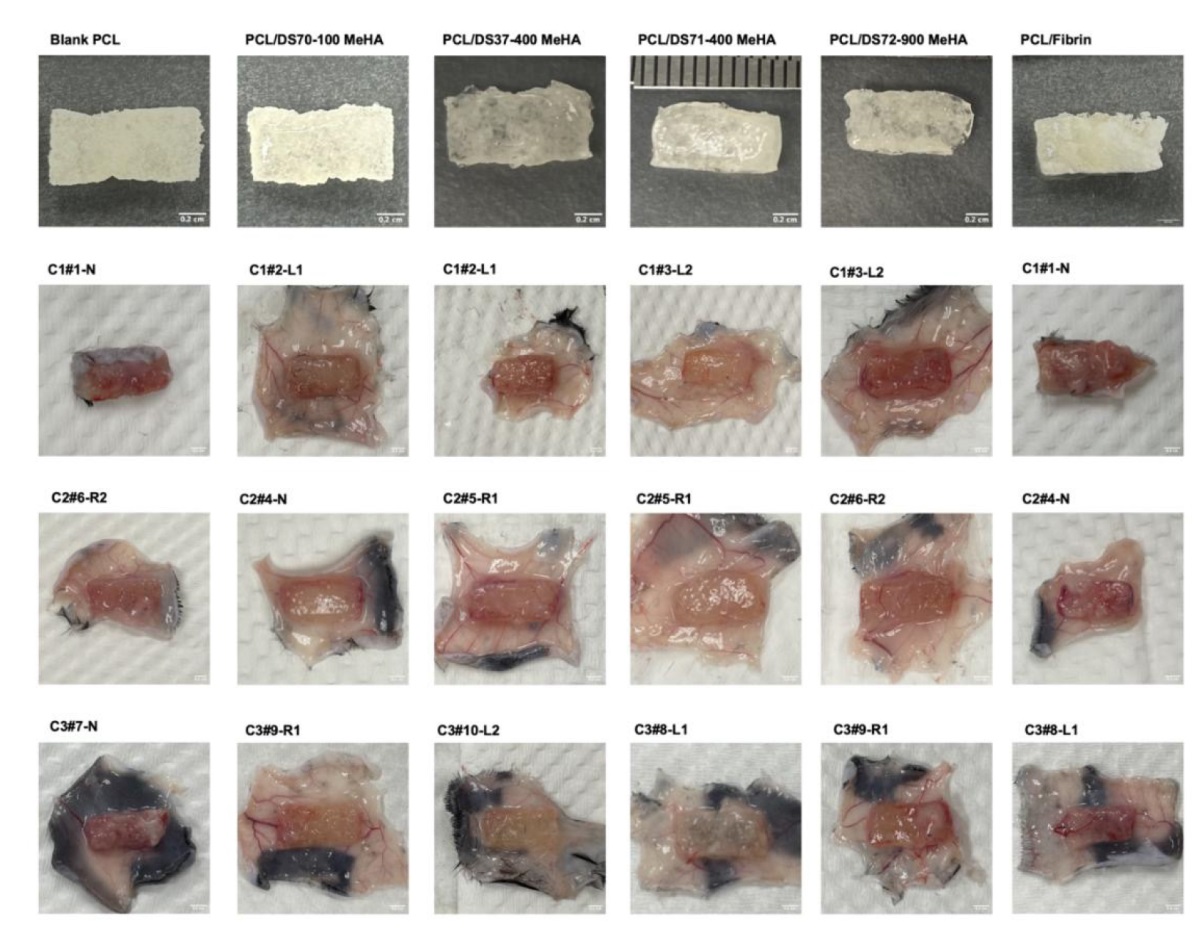
Additional Figure 9. Comparison of angiogenesis situation of all scaffolds before and 4 weeks after transplantation. Images revealed the formation of new blood vessels and fibrotic capsules of different thicknesses around the scaffolds. Notably, the fibrotic capsule around the PCL/DS72–900 MeHA composite scaffolds appeared slightly thicker compared with the others. DS: degree of substitution; MeHA: methacrylated hyaluronic acid; PCL: poly(ε-caprolactone).
| 1. | Liu, D.; Bobrovskaya, L.; Zhou, X. F. Cell Therapy for neurological disorders: the perspective of promising cells. Biology (Basel). 2021, 10, 1142. |
| 2. | Hoang, D. M.; Pham, P. T.; Bach, T. Q.; Ngo, A. T. L.; Nguyen, Q. T.; Phan, T. T. K.; Nguyen, G. H.; Le, P. T. T.; Hoang, V. T.; Forsyth, N. R.; Heke, M.; Nguyen, L. T. Stem cell-based therapy for human diseases. Signal Transduct Target Ther. 2022, 7, 272. |
| 3. |
De Pieri, A.; Rochev, Y.; Zeugolis, D. I. Scaffold-free cell-based tissue engineering therapies: advances, shortfalls and forecast. NPJ Regen Med. 2021, 6, 18.
doi: 10.1038/s41536-021-00133-3 |
| 4. | Chan, B. P.; Leong, K. W. Scaffolding in tissue engineering: general approaches and tissue-specific considerations. Eur Spine J. 2008, 17 Suppl 4, 467-479. |
| 5. |
Pina, S.; Ribeiro, V. P.; Marques, C. F.; Maia, F. R.; Silva, T. H.; Reis, R. L.; Oliveira, J. M. Scaffolding strategies for tissue engineering and regenerative medicine applications. Materials (Basel). 2019, 12, 1824.
doi: 10.3390/ma12111824 URL |
| 6. |
Peña, J.; Corrales, T.; Izquierdo-Barba, I.; Doadrio, A. L.; Vallet-Regí, M. Long term degradation of poly(ɛ-caprolactone) films in biologically related fluids. Polym Degrad Stab. 2006, 91, 1424-1432.
doi: 10.1016/j.polymdegradstab.2005.10.016 URL |
| 7. |
Chang, C. Y.; Chan, A. T.; Armstrong, P. A.; Luo, H. C.; Higuchi, T.; Strehin, I. A.; Vakrou, S.; Lin, X.; Brown, S. N.; O’Rourke, B.; Abraham, T. P.; Wahl, R. L.; Steenbergen, C. J.; Elisseeff, J. H.; Abraham, M. R. Hyaluronic acid-human blood hydrogels for stem cell transplantation. Biomaterials. 2012, 33, 8026-8033.
doi: 10.1016/j.biomaterials.2012.07.058 URL |
| 8. |
Mahsa Khatami, S.; Parivar, K.; Naderi Sohi, A.; Soleimani, M.; Hanaee-Ahvaz, H. Acetylated hyaluronic acid effectively enhances chondrogenic differentiation of mesenchymal stem cells seeded on electrospun PCL scaffolds. Tissue Cell. 2020, 65, 101363.
doi: 10.1016/j.tice.2020.101363 URL |
| 9. |
Chang, H.; Zheng, M.; Yu, X.; Than, A.; Seeni, R. Z.; Kang, R.; Tian, J.; Khanh, D. P.; Liu, L.; Chen, P.; Xu, C. A swellable microneedle patch to rapidly extract skin interstitial fluid for timely metabolic analysis. Adv Mater. 2017, 29, 1702243.
doi: 10.1002/adma.v29.37 URL |
| 10. |
Fryhofer, G. W.; Zlotnick, H. M.; Stoeckl, B. D.; Farrell, M. J.; Steinberg, D. R.; Mauck, R. L. Fabrication and maturation of integrated biphasic anatomic mesenchymal stromal cell-laden composite scaffolds for osteochondral repair and joint resurfacing. J Orthop Res. 2021, 39, 2323-2332.
doi: 10.1002/jor.v39.11 URL |
| 11. |
Shin, J.; Lee, J. S.; Lee, C.; Park, H. J.; Yang, K.; Jin, Y.; Ryu, J. H.; Hong, K. S.; Moon, S. H.; Chung, H. M.; Yang, H. S.; Um, S. H.; Oh, J. W.; Kim, D. I.; Lee, H.; Cho, S. W. Tissue adhesive catechol-modified hyaluronic acid hydrogel for effective, minimally invasive cell therapy. Adv Funct Mater. 2015, 25, 3814-3824.
doi: 10.1002/adfm.v25.25 URL |
| 12. | Trujillo-Lemon, M.; Bowman, C. N.; Stansbury, J. W. pH-responsive hydrogel with controlled swelling and degradation rate. 2005 NSTI Nanotechnology Conference and Trade Show - NSTI Nanotech 2005 Technical Proceedings. 2014. |
| 13. | Smink, A. M.; Rodriquez, S.; Li, S.; Ceballos, B.; Corrales, N.; Alexander, M.; Koster, T.; de Haan, B. J.; Lakey, J. R. T.; de Vos, P. Successful islet transplantation into a subcutaneous polycaprolactone scaffold in mice and pigs. Transplantation direct. 2023, 9, e1417. |
| 14. |
Smink, A. M.; Li, S.; Hertsig, D. T.; de Haan, B. J.; Schwab, L.; van Apeldoorn, A. A.; de Koning, E.; Faas, M. M.; Lakey, J. R.; de Vos, P. The efficacy of a prevascularized, retrievable poly(D,L,-lactide-co-ε-caprolactone) subcutaneous scaffold as transplantation site for pancreatic islets. Transplantation. 2017, 101, e112-e119.
doi: 10.1097/TP.0000000000001663 URL |
| 15. |
Intranuovo, F.; Gristina, R.; Brun, F.; Mohammadi, S.; Ceccone, G.; Sardella, E.; Rossi, F.; Tromba, G.; Favia, P. Plasma modification of PCL porous scaffolds fabricated by solvent-casting/particulate-leaching for tissue engineering. Plasma Process Polym. 2014, 11, 184-195.
doi: 10.1002/ppap.v11.2 URL |
| 16. | Cho, Y. S.; Hong, M. W.; Kim, Y. Y.; Cho, Y. S. Assessment of cell proliferation in salt-leaching using powder (SLUP) scaffolds with penetrated macro-pores. J Appl Polym Sci. 2014, 131, 40240. |
| 17. |
Mota, C.; Puppi, D.; Dinucci, D.; Gazzarri, M.; Chiellini, F. Additive manufacturing of star poly(ε-caprolactone) wet-spun scaffolds for bone tissue engineering applications. J Bioact Compat Polym. 2013, 28, 320-340.
doi: 10.1177/0883911513490341 URL |
| 18. | Guarino, V.; Causa, F.; Ambrosio, L. Porosity and mechanical properties relationship in PCL porous scaffolds. J Appl Biomater Biomech. 2007, 5, 149-157. |
| 19. |
Gupta, B.; Patra, S.; Ray, A. R. Preparation of porous polycaprolactone tubular matrix by salt leaching process. J Appl Polym Sci. 2012, 126, 1505-1510.
doi: 10.1002/app.v126.5 URL |
| 20. |
Kinney, S. M.; Ortaleza, K.; Vlahos, A. E.; Sefton, M. V. Degradable methacrylic acid-based synthetic hydrogel for subcutaneous islet transplantation. Biomaterials. 2022, 281, 121342.
doi: 10.1016/j.biomaterials.2021.121342 URL |
| 21. |
Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; Tinevez, J. Y.; White, D. J.; Hartenstein, V.; Eliceiri, K.; Tomancak, P.; Cardona, A. Fiji: an open-source platform for biological-image analysis. Nat Methods. 2012, 9, 676-682.
doi: 10.1038/nmeth.2019 |
| 22. | Schneider, C. A.; Rasband, W. S.; Eliceiri, K. W. NIH image to ImageJ: 25 years of image analysis. Nat Methods. 2012, 9, 671-675. |
| 23. |
Kim, M. H.; Nguyen, H.; Chang, C. Y.; Lin, C. C. Dual functionalization of gelatin for orthogonal and dynamic hydrogel cross-linking. ACS Biomater Sci Eng. 2021, 7, 4196-4208.
doi: 10.1021/acsbiomaterials.1c00709 URL |
| 24. |
Zhang, D.; Chen, Q.; Shi, C.; Chen, M.; Ma, K.; Wan, J.; Liu, R. Dealing with the foreign-body response to implanted biomaterials: strategies and applications of new materials. Adv Funct Mater. 2021, 31, 2007226.
doi: 10.1002/adfm.v31.6 URL |
| 25. |
Anderson, J. M.; Rodriguez, A.; Chang, D. T. Foreign body reaction to biomaterials. Semin Immunol. 2008, 20, 86-100.
doi: 10.1016/j.smim.2007.11.004 URL |
| 26. |
Janmohammadi, M.; Nourbakhsh, M. S.; Bahraminasab, M.; Tayebi, L. Effect of pore characteristics and alkali treatment on the physicochemical and biological properties of a 3D-printed polycaprolactone bone scaffold. ACS Omega. 2023, 8, 7378-7394.
doi: 10.1021/acsomega.2c05571 URL |
| 27. |
Kim, J. W.; Shin, K. H.; Koh, Y. H.; Hah, M. J.; Moon, J.; Kim, H. E. Production of poly(ε-caprolactone)/hydroxyapatite composite scaffolds with a tailored macro/micro-porous structure, high mechanical properties, and excellent bioactivity. Materials (Basel). 2017, 10, 1123.
doi: 10.3390/ma10101123 URL |
| 28. | Ni, Y.; Qi, H.; Zhang, F.; Jiang, S.; Tang, Q.; Cai, W.; Mo, W.; Miron, R. J.; Zhang, Y. Macrophages modulate stiffness-related foreign body responses through plasma membrane deformation. Proc Natl Acad Sci U S A. 2023, 120, e2213837120. |
| 29. |
Sridharan, R.; Cavanagh, B.; Cameron, A. R.; Kelly, D. J.; O’Brien, F. J. Material stiffness influences the polarization state, function and migration mode of macrophages. Acta Biomater. 2019, 89, 47-59.
doi: 10.1016/j.actbio.2019.02.048 URL |
| 30. |
Zhang, C.; Yuan, Y.; Wu, K.; Wang, Y.; Zhu, S.; Shi, J.; Wang, L.; Li, Q.; Zuo, X.; Fan, C.; Chang, C.; Li, J. Driving DNA origami assembly with a terahertz wave. Nano Lett. 2022, 22, 468-475.
doi: 10.1021/acs.nanolett.1c04369 URL |
| 31. |
Zhang, C.; Jing, X.; Guo, L.; Cui, C.; Hou, X.; Zuo, T.; Liu, J.; Shi, J.; Liu, X.; Zuo, X.; Li, J.; Chang, C.; Fan, C.; Wang, L. Remote photothermal control of DNA origami assembly in cellular environments. Nano Lett. 2021, 21, 5834-5841.
doi: 10.1021/acs.nanolett.1c01821 URL |
| 32. |
Pardue, E. L.; Ibrahim, S.; Ramamurthi, A. Role of hyaluronan in angiogenesis and its utility to angiogenic tissue engineering. Organogenesis. 2008, 4, 203-214.
doi: 10.4161/org.4.4.6926 URL |
| 33. |
Zhang, C.; Xia, D.; Liu, J.; Huo, D.; Jiang, X.; Hu, Y. Bypassing the immunosuppression of myeloid-derived suppressor cells by reversing tumor hypoxia using a platelet-inspired platform. Adv Funct Mater. 2020, 30, 2000189.
doi: 10.1002/adfm.v30.22 URL |
| [1] | Jin Yang, Kanwal Fatima, Xiaojun Zhou, Chuanglong He. Meticulously engineered three-dimensional-printed scaffold with microarchitecture and controlled peptide release for enhanced bone regeneration [J]. Biomaterials Translational, 2024, 5(1): 69-83. |
| [2] | Ross H. McWilliam, Wenlong Chang, Zhao Liu, Jiayuan Wang, Fengxuan Han, Richard A. Black, Junxi Wu, Xichun Luo, Bin Li, Wenmiao Shu. Three-dimensional biofabrication of nanosecond laser micromachined nanofibre meshes for tissue engineered scaffolds [J]. Biomaterials Translational, 2023, 4(2): 104-114. |
| [3] | Seyed Ataollah Naghavi, Changning Sun, Mahbubeh Hejazi, Maryam Tamaddon, Jibao Zheng, Leilei Wang, Chenrui Zhang, Swastina Nath Varma, Dichen Li, Mehran Moazen, Ling Wang, Chaozong Liu. On the mechanical aspect of additive manufactured polyether-ether-ketone scaffold for repair of large bone defects [J]. Biomaterials Translational, 2022, 3(2): 142-151. |
| [4] | Panita Maturavongsadit, Weiwei Wu, Jingyu Fan, Igor B. Roninson, Taixing Cui, Qian Wang. Graphene-incorporated hyaluronic acid-based hydrogel as a controlled Senexin A delivery system [J]. Biomaterials Translational, 2022, 3(2): 152-161. |
| [5] | Jing Long, Bin Teng, Wei Zhang, Long Li, Ming Zhang, Yingqi Chen, Zhenyu Yao, Xiangbo Meng, Xinluan Wang, Ling Qin, Yuxiao Lai. Preclinical evaluation of acute systemic toxicity of magnesium incorporated poly(lactic-co-glycolic acid) porous scaffolds by three-dimensional printing [J]. Biomaterials Translational, 2021, 2(3): 272-284. |
| [6] | Maryam Tamaddon, Helena Gilja, Ling Wang, J. Miguel Oliveira, Xiaodan Sun, Rongwei Tan, Chaozong Liu. Osteochondral scaffolds for early treatment of cartilage defects in osteoarthritic joints: from bench to clinic [J]. Biomaterials Translational, 2020, 1(1): 3-17. |
| [7] | Fujian Zhao, Zhen Yang, Lu Liu, Dafu Chen, Longquan Shao, Xiaofeng Chen. Design and evaluation of a novel sub-scaffold dental implant system based on the osteoinduction of micro-nano bioactive glass [J]. Biomaterials Translational, 2020, 1(1): 82-88. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||