Biomaterials Translational ›› 2021, Vol. 2 ›› Issue (3): 248-256.doi: 10.12336/biomatertransl.2021.03.007
• REVIEW • Previous Articles Next Articles
Qingchuan Wang1, Weidan Wang1,2, Yanfang Li3, Weirong Li3, Lili Tan1,*( ), Ke Yang1,*(
), Ke Yang1,*( )
)
Received:2021-06-04
Revised:2021-07-31
Accepted:2021-09-10
Online:2021-09-28
Published:2021-09-28
Contact:
Lili Tan,Ke Yang
E-mail:lltan@imr.ac.cn;kyang@imr.ac.cn
About author:Ke Yang, kyang@imr.ac.cn.
Wang, Q.; Wang, W.; Li, Y.; Li, W.; Tan, L.; Yang, K. Biofunctional magnesium coating of implant materials by physical vapour deposition. Biomater Transl. 2021, 2(3), 248-256.
| Substrate material | Fabrication method | Degradation behaviour | Osteogenic property | Angiogenic property | Antimicrobial property | References |
|---|---|---|---|---|---|---|
| AZ31 | Vapour deposition | Corrosion resistance improved | ﹣ | ﹣ | ﹣ | |
| AZ31 | Vapour deposition | Comparable to the un-coated 6N-Mg | ﹣ | ﹣ | ﹣ | |
| AZ31 | Vapour deposition + hot press and HIP processes | Corrosion resistance improved | ﹣ | ﹣ | ﹣ | |
| Ti6Al4V | Arc ion plating | Continuous release with Mg degradation | Enhanced new bone regenerating ability in vivo | Accelerated blood vessel formation around the scaffold | Strong killing effect of pure Mg film on Staphylococcus aureus | |
| Ti6Al4V | Arc ion plating | Sustained at least for 14 days | Restrained peri-implant osteolysis | ﹣ | Cu addition enhanced the antibacterial property of Mg coatings | |
| Cold-rolled steel plates | Radio-frequency magnetron sputtering | Corrosion rate greatly decreased | ﹣ | ﹣ | ﹣ | |
| Oxidized Si wafer | Physical vapor deposition | Grains remain intact 48 hours after implantation | Thinner fibrous capsule formed than titanium control samples | ﹣ | ﹣ | |
| PEEK | Vapour deposition | Lower degradation rate without galvanic corrosion | ﹣ | The antibacterial rate reached 99% when co-cultured for 12 hours | ﹣ |
Table 1 Summary of the biofunctions of magnesium (Mg) coating on different implant materials.
| Substrate material | Fabrication method | Degradation behaviour | Osteogenic property | Angiogenic property | Antimicrobial property | References |
|---|---|---|---|---|---|---|
| AZ31 | Vapour deposition | Corrosion resistance improved | ﹣ | ﹣ | ﹣ | |
| AZ31 | Vapour deposition | Comparable to the un-coated 6N-Mg | ﹣ | ﹣ | ﹣ | |
| AZ31 | Vapour deposition + hot press and HIP processes | Corrosion resistance improved | ﹣ | ﹣ | ﹣ | |
| Ti6Al4V | Arc ion plating | Continuous release with Mg degradation | Enhanced new bone regenerating ability in vivo | Accelerated blood vessel formation around the scaffold | Strong killing effect of pure Mg film on Staphylococcus aureus | |
| Ti6Al4V | Arc ion plating | Sustained at least for 14 days | Restrained peri-implant osteolysis | ﹣ | Cu addition enhanced the antibacterial property of Mg coatings | |
| Cold-rolled steel plates | Radio-frequency magnetron sputtering | Corrosion rate greatly decreased | ﹣ | ﹣ | ﹣ | |
| Oxidized Si wafer | Physical vapor deposition | Grains remain intact 48 hours after implantation | Thinner fibrous capsule formed than titanium control samples | ﹣ | ﹣ | |
| PEEK | Vapour deposition | Lower degradation rate without galvanic corrosion | ﹣ | The antibacterial rate reached 99% when co-cultured for 12 hours | ﹣ |

Figure 2. (A, B) Surface morphology (A) and cross section (B) of magnesium (Mg)-coated Ti6Al4V alloy. Scale bars: 50 μm.30 Copyright Wiley Periodicals, Inc. Reproduced with permission.
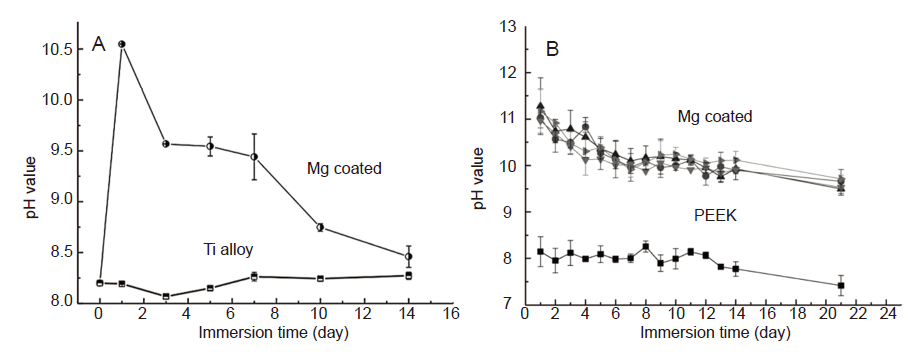
Figure 3. (A) Variation of the pH of immersion solutions after soaking titanium (Ti) alloy with and without magnesium (Mg) coatings. Reproduced from Du et al.30 Copyright Wiley Periodicals, Inc. (B) Variation of the pH of immersion solutions after soaking polyetheretherketone (PEEK) with and without Mg coatings. The upper four lines are all Mg coated. Reprinted from Yu et al.25 Copyright 2018, with permission from Elsevier.
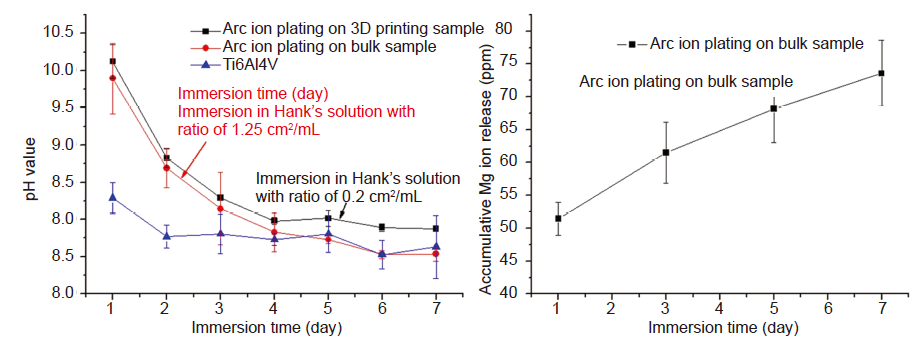
Figure 4. (A, B) pH monitoring (A) and ion release (B) of magnesium (Mg)-coated Ti6Al4V immersed in Hank’s solution for 7 days. Reprinted from Li et al.24
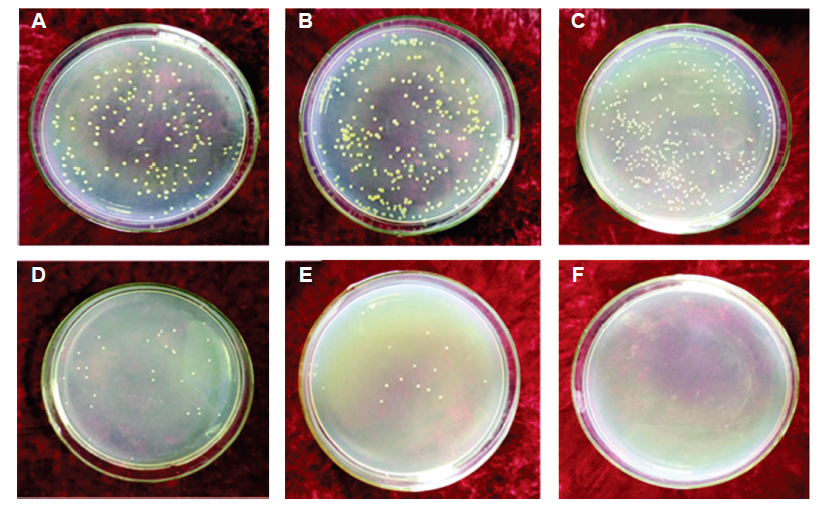
Figure 5. (A-F) Antibacterial effects of Ti6Al4V alloy without (A-C) and with (D-F) magnesium coating, co-cultured with Staphylococcus aureus at 37°C for 6 hours (A, D), 12 hours (B, E) and 24 hours (C, F). Reprinted with permission from Yu et al.31 Copyright © 2017 Acta Metallurgica Sinica.

Figure 6. (A, B) Micro-computed tomographic images of the porous Ti6Al4V with and without magnesium (Mg) coating at 4 and 8 weeks after implantation, where the yellow colour component was the newly-formed bone in these scaffolds. (C) Quantitative results showing the percentage of regenerated bone volume/total volume. Ti: titanium. *P < 0.01, vs. Ti Reprinted from Li et al.24
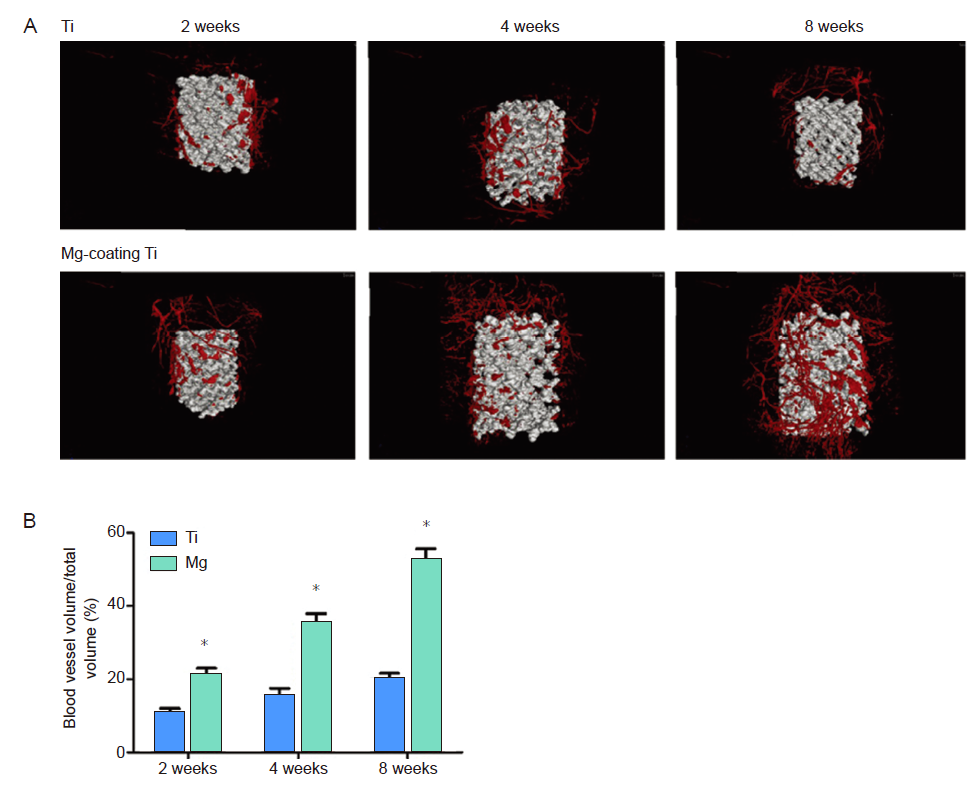
Figure 7. (A) Microangiographic analysis of newly-formed blood vessels around porous Ti6Al4V scaffolds with and without magnesium (Mg) coating. (B) Quantitative results showing blood vessel volume/total volume. *P < 0.05, vs. bare Ti6Al4V scaffold (Ti). Ti: titanium. Reprinted from Gao et al.26
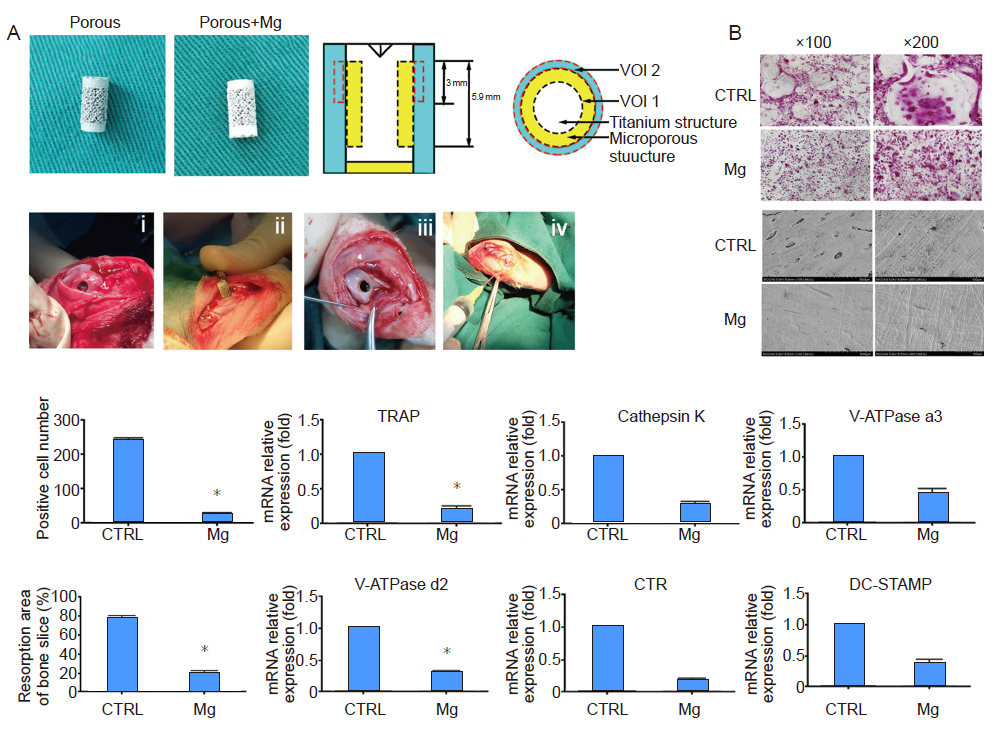
Figure 8. (A) General overview of the implants and implantation process. (B) Magnesium (Mg) inhibited RANKL-induced osteoclastogenesis and bone resorption. *P < 0.05, vs. control (CTRL). CTR: calcitonin receptor; DC-STAMP: dendritic cell-specific transmembrane protein; RANKL: receptor activator of nuclear factor kappa-Β ligand; TRAP: tartrate-resistant acid phosphatase; VOI: volume of interest. Du et al.30 Copyright Wiley-VCH Verlag GmbH & Co. KGaA. Reproduced with permission.
| [1] | Wang, Q. C.; Zhang, B. C.; Ren, Y. B.; Yang, K. Research and application of biomedical nickel-free stainless steels. Jinshu Xuebao. 2017, 53, 1311-1316. |
| [2] | Chen, Q.; Thouas, G. A. Metallic implant biomaterials. Mater Sci Eng RRep. 2015, 87, 1-57. |
| [3] |
Najeeb, S.; Zafar, M. S.; Khurshid, Z.; Siddiqui, F. Applications of polyetheretherketone (PEEK) in oral implantology and prosthodontics. J Prosthodont Res. 2016, 60, 12-19.
doi: 10.1016/j.jpor.2015.10.001 URL |
| [4] |
Drago, C.; Howell, K. Concepts for designing and fabricating metal implant frameworks for hybrid implant prostheses. J Prosthodont. 2012, 21, 413-424.
doi: 10.1111/j.1532-849X.2012.00835.x URL |
| [5] |
Xue, W.; Krishna, B. V.; Bandyopadhyay, A.; Bose, S. Processing and biocompatibility evaluation of laser processed porous titanium. Acta Biomater. 2007, 3, 1007-1018.
doi: 10.1016/j.actbio.2007.05.009 URL |
| [6] | Ratner, B.; Hoffman, A.; Schoen, F.; Lemons, J. Biomaterials science: an introduction to materials in medicine. 2nd ed. Academic Press: 2004. |
| [7] | Asri, R. I. M.; Harun, W. S. W.; Samykano, M.; Lah, N. A. C.; Ghani, S. A. C.; Tarlochan, F, Raza, M. R. Corrosion and surface modification on biocompatible metals: a review. Mater Sci Eng CMater Biol Appl. 2017, 77, 1261-1274. |
| [8] |
Li, Y.; Yang, W.; Li, X.; Zhang, X.; Wang, C.; Meng, X.; Pei, Y.; Fan, X.; Lan, P.; Wang, C.; Li, X.; Guo, Z. Improving osteointegration and osteogenesis of three-dimensional porous Ti6Al4V scaffolds by polydopamine-assisted biomimetic hydroxyapatite coating. ACS Appl Mater Interfaces. 2015, 7, 5715-5724.
doi: 10.1021/acsami.5b00331 URL |
| [9] |
Li, Y.; Jahr, H.; Zhou, J.; Zadpoor, A. A. Additively manufactured biodegradable porous metals. Acta Biomater. 2020, 115, 29-50.
doi: 10.1016/j.actbio.2020.08.018 URL |
| [10] |
Geetha, M.; Singh, A. K.; Asokamani, R.; Gogia, A. K. Ti based biomaterials, the ultimate choice for orthopaedic implants - A review. Prog Mater Sci. 2009, 54, 397-425.
doi: 10.1016/j.pmatsci.2008.06.004 URL |
| [11] | Rafieerad, A. R.; Ashra, M. R.; Mahmoodian, R.; Bushroa, A. R. Surface characterization and corrosion behavior of calcium phosphate-base composite layer on titanium and its alloys via plasma electrolytic oxidation: A review paper. Mater Sci Eng CMater Biol Appl. 2015, 57, 397-413. |
| [12] |
Le, V. Q.; Pourroy, G.; Cochis, A.; Rimondini, L.; Abdel-Fattah, W. I.; Mohammed, H. I.; Carradò, A. Alternative technique for calcium phosphate coating on titanium alloy implants. Biomatter. 2014, 4, e28534.
doi: 10.4161/biom.28534 URL |
| [13] |
Romanò, C. L.; Tsuchiya, H.; Morelli, I.; Battaglia, A. G.; Drago, L. Antibacterial coating of implants: are we missing something? Bone Joint Res. 2019, 8, 199-206.
doi: 10.1302/2046-3758.85.BJR-2018-0316 URL |
| [14] |
Yang, J.; Qin, H.; Chai, Y.; Zhang, P.; Chen, Y.; Yang, K.; Qin, M.; Zhang, Y.; Xia, H.; Ren, L.; Yu, B. Molecular mechanisms of osteogenesis and antibacterial activity of Cu-bearing Ti alloy in a bone defect model with infection in vivo. J Orthop Translat. 2021, 27, 77-89.
doi: 10.1016/j.jot.2020.10.004 URL |
| [15] |
Yang, K.; Zhou, C.; Fan, H.; Fan, Y.; Jiang, Q.; Song, P.; Fan, H.; Chen, Y.; Zhang, X. Bio-Functional design, application and trends in metallic biomaterials. Int J Mol Sci. 2017, 19, 24.
doi: 10.3390/ijms19010024 URL |
| [16] |
Robinson, D. A.; Griffith, R. W.; Shechtman, D.; Evans, R. B.; Conzemius, M. G. In vitro antibacterial properties of magnesium metal against Escherichia coli, Pseudomonas aeruginosa and Staphylococcus aureus. Acta Biomater. 2010, 6, 1869-1877.
doi: 10.1016/j.actbio.2009.10.007 URL |
| [17] |
Ren, L.; Lin, X.; Tan, L.; Yang, K. Effect of surface coating on antibacterial behavior of magnesium based metals. Mater Lett. 2011, 65, 3509-3511.
doi: 10.1016/j.matlet.2011.07.109 URL |
| [18] |
Zhai, Z.; Qu, X.; Li, H.; Yang, K.; Wan, P.; Tan, L.; Ouyang, Z.; Liu, X.; Tian, B.; Xiao, F.; Wang, W.; Jiang, C.; Tang, T.; Fan, Q.; Qin, A.; Dai, K. The effect of metallic magnesium degradation products on osteoclast-induced osteolysis and attenuation of NF-κB and NFATc1 signaling. Biomaterials. 2014, 35, 6299-6310.
doi: 10.1016/j.biomaterials.2014.04.044 URL |
| [19] |
Chen, Z.; Mao, X.; Tan, L.; Friis, T.; Wu, C.; Crawford, R.; Xiao, Y. Osteoimmunomodulatory properties of magnesium scaffolds coated with β-tricalcium phosphate. Biomaterials. 2014, 35, 8553-8565.
doi: 10.1016/j.biomaterials.2014.06.038 URL |
| [20] | Wang, J. L.; Xu, J. K.; Hopkins, C.; Chow, D. H.; Qin, L. Biodegradable Magnesium-Based Implants in Orthopedics-A General Review and Perspectives. Adv Sci (Weinh). 2020, 7, 1902443. |
| [21] |
Belluci, M. M.; Giro, G.; Del Barrio, R. A. L.; Pereira, R. M. R.; Marcantonio, E., Jr.; Orrico, S. R. P. Effects of magnesium intake deficiency on bone metabolism and bone tissue around osseointegrated implants. Clin Oral Implants Res. 2011, 22, 716-721.
doi: 10.1111/j.1600-0501.2010.02046.x URL |
| [22] | Lee, M. H.; Bae, I. Y.; Kim, K. J.; Moon, K. M.; Oki, T. Formation mechanism of new corrosion resistance magnesium thin films by PVD method. Surf Coat Technol. 2003,169-170,670-674. |
| [23] |
Salunke, P.; Shanov, V.; Witte, F. High purity biodegradable magnesium coating for implant application. Mater Sci Eng B. 2011, 176, 1711-1717.
doi: 10.1016/j.mseb.2011.07.002 URL |
| [24] |
Li, X.; Gao, P.; Wan, P.; Pei, Y.; Shi, L.; Fan, B.; Shen, C.; Xiao, X.; Yang, K.; Guo, Z. Novel bio-functional magnesium coating on porous Ti6Al4V orthopaedic implants: in vitro and in vivo study. Sci Rep. 2017, 7, 40755.
doi: 10.1038/srep40755 URL |
| [25] | Yu, X.; Ibrahim, M.; Liu, Z.; Yang, H.; Tan, L.; Yang, K. Biofunctional Mg coating on PEEK for improving bioactivity. Bioact Mater. 2018, 3, 139-143. |
| [26] | Gao, P.; Fan, B.; Yu, X.; Liu, W.; Wu, J.; Shi, L.; Yang, D.; Tan, L.; Wan, P.; Hao, Y.; Li, S.; Hou, W.; Yang, K.; Li, X.; Guo, Z. Biofunctional magnesium coated Ti6Al4V scaffold enhances osteogenesis and angiogenesis in vitro and in vivo for orthopedic application. Bioact Mater. 2020, 5, 680-693. |
| [27] |
Yu, X.; Ibrahim, M.; Lu, S.; Yang, H.; Tan, L.; Yang, K. MgCu coating on Ti6Al4V alloy for orthopedic application. Mater Lett. 2018, 233, 35-38.
doi: 10.1016/j.matlet.2018.08.063 URL |
| [28] |
Yamamoto, A.; Watanabe, A.; Sugahara, K.; Fukumoto, S.; Tsubakino, H. Deposition coating of magnesium alloys with pure magnesium. Mater Trans. 2001, 42, 1237-1242.
doi: 10.2320/matertrans.42.1237 URL |
| [29] |
Fukumoto, S.; Sugahara, K.; Yamamoto, A.; Tsubakino, H. Improvement of corrosion resistance and adhesion of coating layer for magnesium alloy coated with high purity magnesium. Mater Trans. 2003, 44, 518-523.
doi: 10.2320/matertrans.44.518 URL |
| [30] | Du, Z.; Yu, X.; Nie, B.; Zhu, Z.; Ibrahim, M.; Yang, K.; Tan, L.; Wang, Y. Effects of magnesium coating on bone-implant interfaces with and without polyether-ether-ketone particle interference: A rabbit model based on porous Ti6Al4V implants. J Biomed Mater Res BAppl Biomater. 2019, 107, 2388-2396. |
| [31] | Yu, X. M.; Tan, L. L.; Liu, Z. Y.; Yang, K.; Zhu, Z. L.; Li, Y. D. Preparation and properties of biological functional magnesium coating on Ti6Al4V substrate. Jinshu Xuebao. 2018, 54, 943-949. |
| [32] |
Ding, Y.; Du, Z.; Zhu, Z.; Yu, X.; Wang, Y. Effect of biodegradable magnesium-copper coatings on bone integration based on the porous structures in a rabbit model. RSC Adv. 2018, 8, 25127-25132.
doi: 10.1039/C8RA03157F URL |
| [33] |
Tsubakino, H.; Yamamoto, A.; Fukumoto, S.; Watanabe, A.; Sugahara, K.; Inoue, H. High-purity magnesium coating on magnesium alloys by vapor deposition technique for improving corrosion resistance. Mater Trans. 2003, 44, 504-510.
doi: 10.2320/matertrans.44.504 URL |
| [34] | International Organization for Standardization. ISO 10993-12:2021. Biological evaluation of medical devices — Part 12: Sample preparation and reference materials. 2021. |
| [35] |
Wang, C.; Lin, K.; Chang, J.; Sun, J. Osteogenesis and angiogenesis induced by porous β-CaSiO(3)/PDLGA composite scaffold via activation of AMPK/ERK1/2 and PI3K/Akt pathways. Biomaterials. 2013, 34, 64-77.
doi: 10.1016/j.biomaterials.2012.09.021 URL |
| [36] |
Yu, Y.; Jin, G.; Xue, Y.; Wang, D.; Liu, X.; Sun, J. Multifunctions of dual Zn/Mg ion co-implanted titanium on osteogenesis, angiogenesis and bacteria inhibition for dental implants. Acta Biomater. 2017, 49, 590-603.
doi: 10.1016/j.actbio.2016.11.067 URL |
| [37] |
Tan, L.; Yu, X.; Wan, P.; Yang, K. Biodegradable materials for bone repairs: a review. J Mater Sci Technol. 2013, 29, 503-513.
doi: 10.1016/j.jmst.2013.03.002 URL |
| [38] |
Hou, P.; Zhao, C.; Cheng, P.; Wu, H.; Ni, J.; Zhang, S.; Lou, T.; Wang, C.; Han, P.; Zhang, X.; Chai, Y. Reduced antibacterial property of metallic magnesium in vivo. Biomed Mater. 2016, 12, 015010.
doi: 10.1088/1748-605X/12/1/015010 URL |
| [39] |
Jin, X.; Gao, L.; Liu, E.; Yu, F.; Shu, X.; Wang, H. Microstructure, corrosion and tribological and antibacterial properties of Ti-Cu coated stainless steel. J Mech Behav Biomed Mater. 2015, 50, 23-32.
doi: 10.1016/j.jmbbm.2015.06.004 URL |
| [40] |
Liu, C.; Fu, X.; Pan, H.; Wan, P.; Wang, L.; Tan, L.; Wang, K.; Zhao, Y.; Yang, K.; Chu, P. K. Biodegradable Mg-Cu alloys with enhanced osteogenesis, angiogenesis, and long-lasting antibacterial effects. Sci Rep. 2016, 6, 27374.
doi: 10.1038/srep27374 URL |
| [41] |
Li, Y.; Liu, L.; Wan, P.; Zhai, Z.; Mao, Z.; Ouyang, Z.; Yu, D.; Sun, Q.; Tan, L.; Ren, L.; Zhu, Z.; Hao, Y.; Qu, X.; Yang, K.; Dai, K. Biodegradable Mg-Cu alloy implants with antibacterial activity for the treatment of osteomyelitis: in vitro and in vivo evaluations. Biomaterials. 2016, 106, 250-263.
doi: 10.1016/j.biomaterials.2016.08.031 URL |
| [42] |
He, Y.; Zhang, Y.; Zhang, J.; Jiang, Y.; Zhou, R. Fabrication and characterization of Ti-13Nb-13Zr alloy with radial porous Ti-HA coatings for bone implants. Mater Lett. 2017, 209, 543-546.
doi: 10.1016/j.matlet.2017.08.098 URL |
| [1] | Yang Zhao, Qing Sun, Bo Huo. Focal adhesion regulates osteogenic differentiation of mesenchymal stem cells and osteoblasts [J]. Biomaterials Translational, 2021, 2(4): 312-322. |
| [2] | Ying Luo, Jue Wang, Michael Tim Yun Ong, Patrick Shu-hang Yung, Jiali Wang, Ling Qin. Update on the research and development of magnesium-based biodegradable implants and their clinical translation in orthopaedics [J]. Biomaterials Translational, 2021, 2(3): 188-196. |
| [3] | Xirui Jing, Qiuyue Ding, Qinxue Wu, Weijie Su, Keda Yu, Yanlin Su, Bing Ye, Qing Gao, Tingfang Sun, Xiaodong Guo. Magnesium-based materials in orthopaedics: material properties and animal models [J]. Biomaterials Translational, 2021, 2(3): 197-213. |
| [4] | Yu Lu, Subodh Deshmukh, Ian Jones, Yu-Lung Chiu. Biodegradable magnesium alloys for orthopaedic applications [J]. Biomaterials Translational, 2021, 2(3): 214-235. |
| [5] | Jialin Niu, Hua Huang, Jia Pei, Zhaohui Jin, Shaokang Guan, Guangyin Yuan. Research and development strategy for biodegradable magnesium-based vascular stents: a review [J]. Biomaterials Translational, 2021, 2(3): 236-247. |
| [6] | Aditya Joshi, George Dias, Mark P. Staiger. In silico modelling of the corrosion of biodegradable magnesium-based biomaterials: modelling approaches, validation and future perspectives [J]. Biomaterials Translational, 2021, 2(3): 257-271. |
| [7] | Jing Long, Bin Teng, Wei Zhang, Long Li, Ming Zhang, Yingqi Chen, Zhenyu Yao, Xiangbo Meng, Xinluan Wang, Ling Qin, Yuxiao Lai. Preclinical evaluation of acute systemic toxicity of magnesium incorporated poly(lactic-co-glycolic acid) porous scaffolds by three-dimensional printing [J]. Biomaterials Translational, 2021, 2(3): 272-284. |
| [8] | Kamolrat Metavarayuth, Esteban Villarreal, Hui Wang, Qian Wang. Surface topography and free energy regulate osteogenesis of stem cells: effects of shape-controlled gold nanoparticles [J]. Biomaterials Translational, 2021, 2(2): 165-173. |
| [9] | Yunsong Shi, Ruijun He, Xiangyu Deng, Zengwu Shao, Davide Deganello, Chunze Yan, Zhidao Xia. Three-dimensional biofabrication of an aragonite-enriched self-hardening bone graft substitute and assessment of its osteogenicity in vitro and in vivo [J]. Biomaterials Translational, 2020, 1(1): 69-81. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||